Abstract
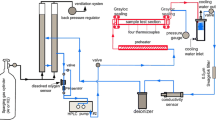
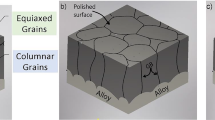
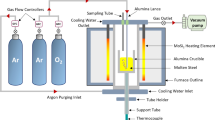
Alloy 617 was exposed to He-CO-CO2 environments with \( P_{\text{CO}} /P_{{{\text{CO}}_{2} }} \) of either 9 or 1320 at temperatures from 1023 K to 1123 K (750 °C to 850 °C) to determine the oxygen diffusion coefficients within the internal oxidation zone of the alloy. The oxygen diffusion coefficients determined based on both intergranular and transgranular oxidation rates were several orders of magnitude greater than those reported in pure nickel and in nickel-based binary alloys, indicating that the rapid internal aluminum oxidation of Alloy 617 was primarily due to enhanced oxygen diffusion along the incoherent Al2O3-alloy interfaces. The range of activation energy values determined for oxygen diffusion associated with the intergranular aluminum oxidation was from 149.6 to 154.7 kJ/mol, and that associated with the transgranular aluminum oxidation was from 244.7 to 283.5 kJ/mol.






Similar content being viewed by others
References
R.E. Mizia: Next Generation Nuclear Plant Intermediate Heat Exchanger Acquisition Strategy, INL/EXT-08-14054, 2008.
J.K. Wright: Next Generation Nuclear Plant Steam Generator and Intermediate Heat Exchanger Materials Research and Development Plan, INL/EXT-08-14107, 2010.
C. Cabet, A. Terlain, P. Lett, L. Guétaz, and J.-M. Gentzbittel: Mater. Corros., 2006, vol. 57, pp. 147–53.
C. Cabet, J. Chapovaloff, F. Rouillard, G. Girardin, D. Kaczorowski, K. Wolski, and M. Pijolat: J. Nucl. Mater., 2008, vol. 375, pp. 173–84.
K. Natesan, A. Purohit, and S.W. Tam: Materials Behavior in HTGR Environments; NUREG/CR-6824, ANL-02/37, Argonne, IL, 2003.
J.K. Wright, L.J. Carroll, C. Cabet, T.M. Lillo, J.K. Benz, J.A. Simpson, W.R. Lloyd, J.A. Chapman, and R.N. Wright: Nucl. Eng. Des., 2012, vol. 251, pp. 252–60.
R. Wright, J. Wright, and C. Cabet: in Comprehensive Nuclear Materials, Elsevier, Amsterdam, 2012, pp. 251–77.
R.N. Wright: Summary of Studies of Aging and Environmental Effects on Inconel 617 and Haynes 230, INL/EXT-06-11750, 2006.
H.J. Christ, D. Schwanke, and T. Uihlein: Oxid. Met., 1988, vol. 30, pp. 1–26.
H.J. Christ, U. Künecke, K. Meyer, and H.G. Sockel: Oxid. Met., 1988, vol. 30, pp. 27–51.
W.J. Quadakkers and H. Schuster: Nucl. Technol., 1984, vol. 66, pp. 383–91.
D. Kumar, R.R. Adharapurapu, T.M. Pollock, and G.S. Was: Metall. Mater. Trans. A, 2011, vol. 42, pp. 1245–65.
R.R. Adharapurapu, D. Kumar, J. Zhu, and T.M. Pollock: Corros. Sci., 2011, vol. 53, pp. 388–98.
W.J. Quadakkers and H. Schuster: Mater. Corros., 1985, vol. 36, pp. 141–50.
C. Cabet and F. Rouillard: J. Nucl. Mater., 2009, vol. 392, pp. 235–42.
C. Cabet and B. Duprey: Nucl. Eng. Des., 2012, vol. 251, pp. 139–45.
Y. Hosoi and S. Abe: Metall. Trans. A, 1975, vol. 6, pp. 1171–78.
H. Yun, P. Ennis, H. Nickel, and H. Schuster: J. Nucl. Mater., 1984, vol. 125, pp. 258–72.
C. Wagner: Zeitschrift Fur Elektrochemie, Berichte Der Bunsengesellschaft Fur Phys. Chemie, 1959, vol. 63, pp. 772–82.
R.A. Rapp: Corrosion, 1965, vol. 21, pp. 382–401.
D.J. Young: High Temperature Oxidation and Corrosion of Metals, 1st ed., Elsevier, Oxford, UK, 2008.
F. Gesmundo and Y. Niu: Oxid. Met., 2003, vol. 60, pp. 347–70.
F. Gesmundo and Y. Niu: Oxid. Met., 2004, vol. 62, pp. 357–74.
F. Maak: Zeitschrift Für Met., 1961, vol. 52, p. 538.
G. Gulsoy and G.S. Was: Metall. Mater. Trans. A, 2014, DOI:10.1007/s11661-014-2629-y.
D.P. Whittle, Y. Shida, G.C. Wood, F.H. Stott, and B.D. Bastow: Philos. Mag. A, 1982, vol. 46, p. 931.
F.H. Stott, G.C. Wood, D.P. Whittle, B.D. Bastow, Y. Shida, and A. Martinez-Villafane: Solid State Ionics, 1984, vol. 12, pp. 365–74.
F.H. Stott and G.C. Wood: Mater. Sci. Technol., 1988, vol. 4, pp. 1072–78.
D. Kumar, C.J. Torbet, and G.S. Was: Meas. Sci. Technol., 2009, vol. 20, p. 95708.
H.C. Yi, S.W. Guan, W.W. Smeltzer, and A. Petric: Acta Metall. Mater., 1994, vol. 42, pp. 981–90.
C.A. Schneider, W.S. Rasband, and K.W. Eliceiri: Nat. Methods, 2012, vol. 9, pp. 671–75.
S.W. Guan and W.W. Smeltzer: Oxid. Met., 1994, vol. 42, pp. 375–91.
J-W. Park and C.J. Altstetter: Metall. Trans. A, 1987, vol. 18, pp. 43–50.
O. Kubaschewski and C.B. Alcock: Materials Thermochemistry, 6th ed., Oxford: Pergamon Press, 1993.
J.A. Nesbitt and R.W. Heckel: Metall. Trans. A, 1987, vol. 18, pp. 2075–86.
C.B. Alcock and P.B. Brown: Met. Sci., 1969, vol. 3, pp. 116–20.
R. Barlow and P.J. Grundy: J. Mater. Sci., 1969, vol. 4, pp. 797–801.
S. Goto, K. Nomaki, and S. Koda: J. Japan Inst. Met., 1967, vol. 31, pp. 600–606.
G.J. Lloyd and J.W. Martin: Met. Sci., 1973, vol. 7, pp. 75–75.
J. Lloyd and J.W. Martin: Met. Sci. J., 1972, vol. 6, pp. 7–11.
R.A. Kerr: Ph.D. Thesis, Ohio State University Columbus, OH, 1972.
S.P. Zholobov and M.D. Malev: Sov. Phys. Tech. Phys., 1971, vol. 16, pp. 488–94.
E.A. Brandes: Smithells Metals Reference Book, 6th ed., Butterworths, London, 1983.
44 S. Garruchet, O. Politano, P. Arnoux, and V. Vignal: Solid State Commun., 2010, vol. 150, pp. 439–42.
H.O. Nam, I.S. Hwang, K.H. Lee, and J.H. Kim: Corros. Sci., 2013, vol. 75, pp. 248–55.
S. Perusin, B. Viguier, D. Monceau, L. Ressier, and E. Andrieu: Acta Mater., 2004, vol. 52, pp. 5375–80.
S. Perusin, D. Monceau, and E. Andrieu: J. Electrochem. Soc., 2005, vol. 152, p. E390.
Y. Shida, F.H. Stott, B.D. Bastow, D.P. Whittle, and G.C. Wood: Oxid. Met., 1982, vol. 18, p. 93.
Acknowledgments
This research was funded by the DOE Office of Nuclear Energy’s Nuclear Energy University Programs under award 00087993. The authors also acknowledge NSF awards DMR-0320740 that made possible the acquisition of the FEI Nova Dual FIB Workstation/SEM at the University of Michigan Electron Microbeam Analysis Laboratory.
Author information
Authors and Affiliations
Corresponding author
Additional information
Manuscript submitted June 30, 2014.
Rights and permissions
About this article
Cite this article
Gulsoy, G., Was, G.S. Enhanced Oxygen Diffusion Within the Internal Oxidation Zone of Alloy 617 in Controlled Impurity Helium Environments from 1023 K to 1123 K (750 °C to 850 °C). Metall Mater Trans A 46, 1628–1638 (2015). https://doi.org/10.1007/s11661-015-2741-7
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11661-015-2741-7