Abstract
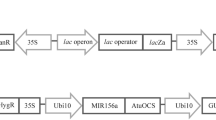
A fast regenerating Agrobacterium tumefaciens-mediated transformation protocol for Bacopa monnieri (L.) Wettst. was developed as a model system for heterologous expression of terpenoid indole alkaloid pathway genes from Catharanthus roseus (L.) G. Don. The direct regeneration of shoots from leaf explants co-cultured with A. tumefaciens resulted in the integration of a tryptophan decarboxylase (tdc) and strictosidine synthase (str) cassette (<hpt-<Tdc2-<Str-gus>) in the regenerated progeny. The highest transformation efficiency (83.88%) was achieved when leaf explants were infected on the adaxial laminar surface by manual pricking with 48- to 72-h-old suspensions (OD600 = 0.5–0.6) of A. tumefaciens strain LBA1119 (carrying the binary vector pMOG22). The heterologous expression of tryptophan decarboxylase and strictosidine synthase genes that are otherwise not present in B. monnieri plants was confirmed through semi-quantitative PCR and metabolite quantification assays. The entire protocol duration from co-cultivation through regeneration of transgenic plants to their establishment in the glass house took 40–45 d. The developed B. monnieri model can be used to test expression cassettes carrying genes for plant secondary metabolic pathway engineering, especially those genes that are expressed in differentiated cell, tissue, or organs.





Similar content being viewed by others
References
Abiri R, Valdiani A, Maziah M, Shaharuddin NA, Sahebi M, Yusof ZNB (2015) A critical review of the concept of transgenic plants: insights into pharmaceutical biotechnology and molecular farming. Curr Issues Mol Biol 18:21–42
Birch RG (1997) Plant transformations: problems and strategies for practical applications. Annu Rev Plant Physiol Plant Mol Biol 48:297–326
Canel C, Lopes-Cardoso MI, Whitmer S, van der Fits L, Pasquali G, van der Heijden R, Hoge JHC, Verpoorte R (1998) Effects of over-expression of strictosidine synthase and tryptophan decarboxylase on alkaloid production by cell cultures of Catharanthus roseus. Planta 205:414–419
Darokar MP, Khanuja SPS, Gupta AK, Kumar S, Gupta MM, Singh AK, Shasany AK, Raju BV, Mishra PK, Rehman L, Kalra A, Verma RK, Mishra S, Gangwar A, Misra HO, Chauhan HS, Singh S, Bahl JR, Kumar B, Lal RK, Singh AK, Krishna K, Bansal RP, Tandon S, Dhawan OP (2007) CIM-Jagriti a variety of brahmi (Bacopa monnieri) with high herbage and bacoside—a yield potential. J Med Arom Pl Sci 29:90–93
Farré G, Twyman RM, Christou P, Capell T, Zhu C (2015) Knowledge-driven approaches for engineering complex metabolic pathways in plants. Curr Opin Biotech 32:54–60
Gandhi SG, Mahajan V, Bedi YS (2015) Changing trends in biotechnology of secondary metabolism in medicinal and aromatic plants. Planta 241:303–317
Jefferson RA, Kavanagh TA, Bevan MW (1987) GUS fusions: beta-glucuronidase as a sensitive and versatile gene fusion marker in higher plants. EMBO J 6:3901–3907
Khanuja SPS, Shasany AK, Darokar MP, Kumar S (1999) Rapid isolation of DNA from dry and fresh samples of plants producing large amounts of secondary metabolites and essential oils. Plant Mol Biol Rep 17:1–7
Komori T, Imayama T, Kato N, Ishida Y, Ueki J, Komari T (2007) Current status of binary vectors and super binary vectors. Plant Physiol 145:1155–1160
Kumari U, Vishwakarma RK, Gupta N, Ruby SMV, Khan BM (2015) Efficient shoots regeneration and genetic transformation of Bacopa monnieri. Physiol Mol Biol Plants 21:261–267
Ladics GS, Bartholomaeus A, Bregitzer P, Doerrer NG, Gray A, Holzhauser T, Jordan M, Keese P, Kok E, Macdonald P, Parrot W, Privalle L, Raybould A, Rhee SY, Rice E, Romeis J, Vaughn J, Wal JM, Glenn K (2015) Genetic basis and detection of unintended effects in genetically modified crop plants. Transgenic Res 24:587–603
Mahender A, Mallesham B, Srinivas K, Kumar GK, Rao KV, Rajesh Y, Zhang P, Sadanandam A (2012) A rapid and efficient method for in vitro shoot organogenesis and production of transgenic Bacopa monnieri L. mediated by Agrobacterium tumefaciens. In Vitro Cell Dev Biol Plant 48:153–159
Majumdar S, Garai S, Jha S (2011) Genetic transformation of Bacopa monnieri by wild type strains of Agrobacterium rhizogenes stimulates production of bacopa saponins in transformed calluses and plants. Plant Cell Rep 30:941–954
Mathew J, Paul J, Nandhu MS, Paulose CS (2010) Bacopa monnieri And Bacoside-a for ameliorating epilepsy associated behavioral deficits. Fitoterapia 81:315–322
Menassa R, Ahmad A, Joensuu JJ (2012) Transient expression using Agroinfiltration and its applications in molecular farming. In: Wang A, Ma S (eds) Molecular farming in plants: recent advances and future prospects. Springer, London, pp 183–198
Mitsuhara I, Ugaki M, Hirochika H, Ohshima M, Murakami T, Gotoh Y, Katayose Y, Nakamura S, Honkura R, Nishimiya S, Ueno K, Mochizuki A, Tanimoto H, Tsugawa H, Otsuki Y, Ohashi Y (1996) Efficient promoter cassettes for enhanced expression of foreign genes in dicotyledonous and monocotyledonous plants. Pl Cell Physiol 37:49–59
Murashige T, Skoog F (1962) A revised medium for rapid growth and bioassays with tobacco tissue cultures. Physiol Plant 15:473–497
Newell CA (2000) Plant transformation technology: developments and applications. Mol Biotechnol 16:53–65
Nisha KK, Seetha K, Rajmohan K, Purushothama MG (2003) Agrobacterium tumefaciens-mediated transformation of Brahmi [Bacopa monniera (L.) Wettst.], a popular medicinal herb of India. Curr Sci 85:85–89
Rahman LU, Verma PC, Singh D, Gupta MM, Banerjee S (2002) Bacoside production by suspension cultures of Bacopa monnieri (L.) Pennell. Biotech Lett 24:1427–1429
Ramesh M, Karthikeyan A, Vijayakumar K, Largia MJV, Pandian SK (2011) Agrobacterium-mediated transformation of pharmaceutically important Indian medicinal herb Bacopa monnieri (L.). J Med Plant Res 5:2316–2321
Roepke J, Salim V, Wu M, Thamm AMK, Murata J, Ploss K, Boland W, De Luca V (2010) Vinca drug components accumulate exclusively in leaf exudates of Madagascar periwinkle. PNAS USA 107:15287–15292
Sainsbury F, Lomonossoff GP (2014) Transient expressions of synthetic biology in plants. Curr Opin Plant Bio 19:1–7
Sharma KK, Bhatnagar MP, Thorpe TA (2005) Genetic transformation technology: status and problems. In Vitro Cell Dev Biol Plant 41:102–112
Shrivastava N, Rajani M (1999) Multiple shoot regeneration and tissue culture studies on Bacopa monnieri (L.) Pennell. Plant Cell Rep 18:919–923
Sivaramakrishna C, Rao CV, Trimurtulu G (2005) Triterpenoid glycosides from Bacopa monniera. Phytochem 66:2719–2728
St-Pierre B, Vazquez-Flota FA, De Luca V (1999) Multicellular compartmentation of Catharanthus roseus alkaloid biosynthesis predicts intercellular translocation of a pathway intermediate. Plant Cell 11:887–900
Tiwari V, Tiwari KN, Singh BD (2001) Comparative studies of cytokinins on in vitro propagation of Bacopa monnieri. Plant Cell Tissue Organ Cult 66:9–16
Tiwari V, Tiwari KN, Singh BD (2006) Shoot bud regeneration from different explants of Bacopa monniera (L.) Wettst. by trimethoprim and bavistin. Plant Cell Rep 25:629–635
Trick HN, Finer JJ (1997) SAAT: sonicated-assisted Agrobacterium-mediated transformation. Transgenic Res 6:329–336
Vancanneyt G, Schmidt R, O’Connor-Sanchez A, Willmitzer L, Rocha-Sosa M (1990) Construction of an intron-containing marker gene: splicing of the intron in transgenic plants and its use in monitoring early events in Agrobacterium-mediated plant transformation. Mol Gen Genet 220:245–250
Verma P, Mathur AK (2011) Agrobacterium tumefaciens mediated transgenic plant production via direct shoot bud organogenesis from pre-plasmolyzed leaf explants of Catharanthus roseus. Biotechnol Lett 33:1053–1060
Verma P, Mathur AK, Srivastava A, Mathur A (2012) Emerging trends in research on spatial and temporal organization of terpenoid indole alkaloid pathway in Catharanthus roseus: a literature update. Protoplasma 249:255–268
Verma P, Sharma A, Khan SA, Shanker K, Mathur AK (2015a) Over-expression of Catharanthus roseus tryptophan decarboxylase and strictosidine synthase in rol gene integrated transgenic cell suspensions of Vinca minor. Protoplasma 252:373–381
Verma P, Mathur AK, Khan SA, Verma N, Sharma A (2015b) Transgenic studies for modulating terpenoid indole alkaloids pathway in Catharanthus roseus: present status and future options. Phytochem Rev DOI. doi:10.1007/s11101-015-9447-8
Vijayakumar M, Vijayakumar R, Stephen R (2010) In vitro propagation of Bacopa monnieri L.-A multipurpose medicinal plant. Ind J Sci Tech 3:781–786
Yadav S, Sharma P, Srivastava A, Desai P, Shrivastava N (2014) Strain specific Agrobacterium-mediated genetic transformation of Bacopa monnieri. J Genet Eng Biotechnol 12:89–94
Zarate R, Verpoorte R (2007) Strategies for the genetic modification of the medicinal plant Catharanthus roseus (L.) G. Don. Phytochem Rev 6:475–491
Zhao L, Sander GW, Shanks JV (2013) Perspectives of the metabolic engineering of terpenoid indole alkaloids in Catharanthus roseus hairy roots. Adv Biochem Eng Biotechnol 134:23–54
Acknowledgements
The authors gratefully acknowledge the Council of Scientific and Industrial Research (CSIR), New Delhi, and Director, CSIR-CIMAP, Lucknow, for providing the facility and financial support to carry out this work. AS is highly thankful to the Department of Science and Technology (DST), Gov. of India, for providing an INSPIRE fellowship. NV thanks the University Grants Commission, New Delhi, for awarding her the UGC-Rajiv Gandhi National Fellowship to conduct part of this investigation. We also thank Prof. Johan Memelink of Leiden University for providing the Agrobacterium tumefaciens strain LBA1119 with construct (<hpt-<Tdc2-<Str-gus>).
Author information
Authors and Affiliations
Corresponding author
Additional information
Editor: Todd Jones
Rights and permissions
About this article
Cite this article
Sharma, A., Verma, N., Verma, P. et al. Optimization of a Bacopa monnieri-based genetic transformation model for testing the expression efficiency of pathway gene constructs of medicinal crops. In Vitro Cell.Dev.Biol.-Plant 53, 22–32 (2017). https://doi.org/10.1007/s11627-017-9804-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11627-017-9804-y