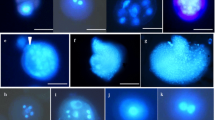
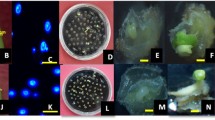
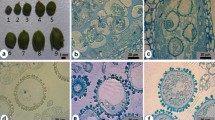
Abstract
Brassica nigra is generally regarded as a recalcitrant species for microspore culture among Brassica crops. Conditions for reliable induction of microspore embryogenesis of B. nigra were studied in this context. Flower bud length and microspore developmental stage were correlated with further embryogenesis. The optimal bud size range was 2.0–2.5 mm for the highest proportion of totipotent, late uninucleate microspore and the highest frequency of microspore embryogenesis. Treatment of a short heat shock by incubating the microspore culture at 32°C for 24 h was suitable for the microspore survival, sustained cell divisions, and further induced embryogenesis. Subsequently, the use of NLN medium with the addition of 13% sucrose and 0.1% activated charcoal (AC) provided the optimal conditions for the development of microspore-derived embryos (MDEs). The early cotyledonary (EC) stage embryos cultured on MS medium fortified with 4.6 μM zeatin (ZT) and 0.12 μM indole-3-acetic acid (IAA) resulted in the most efficient rates of plantlet regeneration. The ploidy levels of regenerated plants of B. nigra were determined by flow cytometry, revealing that 50.6% were diploid. The results enable the advancement of breeding programs and genetic studies in B. nigra.





Similar content being viewed by others
References
Abraha E, Bechyne M, Klima M, Vyvadilova M (2008) Analysis of factors affecting embryogenesis in microspore cultures of Brassica carinata. Agric Trop Subtrop 41:53–60
Acanda Y, Prado MJ, González MV, Rey M (2013) Somatic embryogenesis from stamen filaments in grapevine (Vitis vinifera L. cv. Mencía): changes in ploidy level and nuclear DNA content. In Vitro Cell Dev Biol Plant. doi:10.1007/s11627-013-9499-7
Ahmadi B, Alizadeh K, Teixeira da Silva JA (2012) Enhanced regeneration of haploid plantlets from microspores of Brassica napus L. using bleomycin, PCIB, and phytohormones. Plant Cell Tissue Organ Cult 109:525–533
Ali MM, Mian MAK, Custers JBM, Khuram MMH (2008) Microspore culture and the performance of microspore derived doubled haploid in Brassica juncea (L.). Bangladesh J Agric Res 33:571–578
Asif M, Eudes F, Goyal A, Amundsen E, Randhawa H, Spaner D (2013) Organelle antioxidants improve microspore embryogenesis in wheat and triticale. In Vitro Cell Dev Biol Plant. doi:10.1007/s11627-013-9514-z
Belmonte MF, Ambrose SJ, Ross ARS, Abrams SR, Stasolla C (2006) Improved development of microspore-derived embryo cultures of Brassica napus cv. Topaz following changes in glutathione metabolism. Physiol Plant 127:690–700
Bhowmik P, Dirpaul J, Polowick P, Ferrie AMR (2011) A high throughput Brassica napus microspore culture system: influence of percoll gradient separation and bud selection on embryogenesis. Plant Cell Tissue Organ Cult 106:359–362
Chanana NP, Dhawan V, Bhojwani SS (2005) Morphogenesis in isolated microspore cultures of Brassica juncea. Plant Cell Tissue Organ Cult 83:169–177
Dias JS (1999) Effect of activated charcoal on Brassica oleracea microspore culture embryogenesis. Euphytica 108:65–69
Feher A, Pasternak TP, Dudits D (2003) Transition of somatic plant cells to an embryogenic state. Plant Cell Tissue Organ Cult 74:201–228
Ferrie AMR, Caswell L (2010) Isolated microspore culture techniques and recent progress for haploid and doubled haploid plant production. Plant Cell Tissue Organ Cult 104:301–309
Gamborg OL, Miller RA, Ojima K (1968) Nutrient requirements of suspension cultures of soybean root cells. Exp Cell Res 50:151–158
Gland A, Lichter R, Schweiger HG (1988) Genetic and exogenous factors affecting embryogenesis in isolated microspore cultures of Brassica napus L. J Plant Physiol 132:613–617
Gu HH, Zhang DQ, Zhou WJ (2004) Effect of medium renovation and colchicine treatment on embryogenesis of isolated microspores of Brassica rapa ssp. chinensis. Acta Agron Sin 30:78–81
Guo YD, Pulli S (1996) High-frequency embryogenesis in Brassica campestris microspore culture. Plant Cell Tissue Organ Cult 46:219–225
Haddadi P, Moieni A, Karimzadeh G, Abdollahi MR (2008) Effects of gibberellin, abscisic acid and embryo desiccation on normal plantlet regeneration, secondary embryogenesis and callogenesis in microspore of Brassica napus L. cv. PF704. Int J Plant Prod 2:153–162
Henderson CAP, Pauls KP (1992) The use of haploidy to develop plants that express several recessive traits using light-seeded canola (Brassica napus) as an example. Theor Appl Genet 83:476–479
Kernan Z, Ferrie AMR (2006) Microspore embryogenesis and the development of a double haploidy protocol for cow cockle (Saponaria vaccaria). Plant Cell Rep 25:274–280
Lemonnier-Le PC, Chatelet C, Kloareg B, Potin P (2001) Carrageenan oligosaccharides enhance stress-induced microspore embryogenesis in Brassica oleracea var. italica. Plant Sci 160:1211–1220
Lichter R (1982) Induction of haploid plants from isolated pollen of Brassica napus. Plant Breed 105:427–434
Lionneton E, Beuret W, Delaitre C, Ochatt S, Rancillac M (2001) Improved microspore culture and doubled-haploid plant regeneration in the brown condiment mustard (Brassica juncea). Plant Cell Rep 20:126–130
Murashige T, Skoog F (1962) A revised media for rapid growth and bioassays with tobacco tissue culture. Physiol Plant 15:473–479
Prem D, Gupta K, Sarkar G, Agnihotri A (2008) Activated charcoal induced high frequency microspore embryogenesis and efficient doubled haploid production in Brassica juncea. Plant Cell Tissue Organ Cult 93:269–282
Reynolds TL (1997) Pollen embryogenesis. Plant Mol Biol 33:1–10
Takahata Y, Keller WA (1991) High frequency embryogenesis and plant regeneration in isolated microspore culture of Brassica oleracea L. Plant Sci 74:235–242
Takahira J, Cousin A, Nelson MN, Cowling WA (2011) Improvement in efficiency of microspore culture to produce doubled haploid canola (Brassica napus L.) by flow cytometry. Plant Cell Tissue Organ Cult 104:51–59
Taylor JD, Conway J, Roberts SJ, Astley D, Vicente JG (2002) Sources and origin of resistance to Xanthomonas campestris pv. campestris in Brassica genomes. Phytopathology 92:105–111
Tian H, Yao CY, Sun MX (2004) High frequency conversion of microspore-derived embryos of Brassica napus cv. Topas by supplemental calcium and vitamins. Plant Cell Tissue Organ Cult 76:159–165
Wakui K, Takahata Y, Kaizuma N (1994) Effect of abscisic acid and high osmoticum concentration on the induction of desiccation tolerance in microspore-derived embryos of Chinese cabbage (Brassica campestris L.). Breed Sci 44:29–34
Wan GL, Naeem MS, Geng XX, Xu L, Li B, Jilani G, Zhou WJ (2011) Optimization of microspore embryogenesis and plant regeneration protocols for Brassica napus. Int J Agric Biol 13:83–88
Wang TT, Li HX, Zhang J, Ouyang B, Lu Y, Ye Z (2009) Initiation and development of microspore embryogenesis in recalcitrant purple flowering stalk (Brassica campestris ssp. chinensis var. purpurea Hort.) genotypes. Sci Hortic 121:419–424
Westman AL, Kresovich S, Dickson MH (1999) Regional variation in Brassica nigra and other weedy crucifers for disease reaction to Alternaria brassicicola and Xanthomonas campestris pv. campestris. Euphytica 106:253–259
Winarto B, Teixeira da Silva JA (2011) Microspore culture protocol for Indonesian Brassica oleracea. Plant Cell Tissue Organ Cult 107:305–315
Wu JH, Zhang XL, Nie YC, Jin SX, Liang SG (2004) Factors affecting somatic embryogenesis and plant regeneration from a range of recalcitrant genotypes of Chinese cottons (Gossypium hirsutum L.). In Vitro Cell Dev Biol Plant 40:371–375
Xu L, Najeeb U, Tang GX, Gu HH, Zhang GQ, He Y, Zhou WJ (2007) Haploid and doubled haploid technology. In: Gupta SK, Delseny M, Kader JC (eds) Advances in botanical research: Rapeseed breeding, pp 181–216
Zhang GQ, He Y, Xu L, Tang GX, Zhou WJ (2006a) Genetic analyses of agronomic and seed quality traits of doubled haploid population in Brassica napus through microspore culture. Euphytica 149:169–177
Zhang GQ, Zhang DQ, Tang GX, He Y, Zhou WJ (2006b) Plant development from microspore-derived embryos in oilseed rape as affected by chilling, desiccation and cotyledon excision. Biol Plant 50:180–186
Zhang W, Fu Q, Dai X, Bao M (2008) The culture of isolated microspores of ornamental kale (Brassica oleracea var. acephala) and the importance of genotype to embryo regeneration. Sci Hortic 117:69–72
Zhou WJ, Tang GX, Hagberg P (2002) Efficient production of doubled haploid plants by immediate colchicine treatment of isolated microspores in winter Brassica napus. Plant Growth Regul 37:185–192
Acknowledgments
This work was financially supported by the Natural Science Foundation of China (31201629), Zhejiang Province for Key Agriculture Development Projects (2012C12903), Science and Technology Department of Zhejiang Province, and Zhejiang Academy of Agricultural Sciences.
Author information
Authors and Affiliations
Corresponding author
Additional information
Editor: John W. Forster
Rights and permissions
About this article
Cite this article
Gu, H., Sheng, X., Zhao, Z. et al. Initiation and development of microspore embryogenesis and plant regeneration of Brassica nigra . In Vitro Cell.Dev.Biol.-Plant 50, 534–540 (2014). https://doi.org/10.1007/s11627-014-9612-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11627-014-9612-6