Abstract
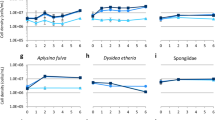
Many marine and freshwater organisms are rocky bottom dwellers, and the mineralogical composition of the substratum is known to potentially condition their biology and ecology. In this work, we propose the use of 3D sponge cellular aggregates, called primmorphs, as suitable models for a multidisciplinary study of the mechanisms which regulate the biological responses triggered by the contact with different inorganic substrata. In our experiments, primmorphs obtained from the marine sponge Petrosia ficiformis (Poiret, 1789) were reared on calcium carbonate or on quartzitic substrata, respectively, and their morphological development was described. In parallel, the quantitative expression levels of two genes, silicatein and heat shock protein 70 (HSP70), were evaluated. The first gene is strictly correlated to spiculogenesis and sponge growth, while the second is an important indicator of stress. The results achieved with this in vitro model clearly demonstrate that quartzitic substrata determine the increase of silicatein gene expression, a lower expression of HSP70 gene, and a remarkable difference in primmorphs morphology compared to the analogous samples grown on marble.





Similar content being viewed by others
References
Aarskog N. K.; Vedeler C. A. Real-time quantitative polymerase chain reaction. A new method that detects both the peripheral myelin protein 22 duplication in Charcot–Marie–Tooth type 1A disease and the peripheral myelin protein 22 deletion in hereditary neuropathy with liability to pressure palsies. Hum. Genet. 107: 494–498; 2000.
Bavestrello G.; Arillo A.; Benatti U.; Cerrano C.; Cattaneo-Vietti R.; Cortesogno L.; Gaggero L.; Giovine M.; Tonetti M.; Sarà M. Quartz dissolution by the sponge Chondrosia reniformis (Porifera, Demospongiae). Nature 378: 374–376; 1995.
Bavestrello G.; Benatti U.; Calcinai B.; Cattaneo-Vietti R.; Cerrano C.; Favre A.; Giovine M.; Lanza S.; Pronzato R.; Sarà M. Body polarity and mineral selectivity in the demosponge Chondrosia reniformis. Biol. Bull. 195: 120–125; 1998.
Bavestrello G.; Benatti U.; Cattaneo-Vietti R.; Cerrano C.; Giovine M. Sponge cell reactivity to various forms of silica. Microsc. Res. Tech. 62: 327–335; 2003.
Bavestrello G.; Cerrano C.; Puce S.; Bianchi C. N.; Calcinai B.; Cattaneo-Vietti R.; Morri C.; Sarà M. Bio-mineralogy as a structuring factor for marine epibenthic communities. Mar. Ecol. Prog. Ser. 193: 241–249; 2000.
Cerrano C.; Bavestrello G.; Arillo A.; Benatti U.; Calcinai B.; Cattaneo-Vietti R.; Cortesogno L.; Gaggero L.; Giovine M.; Puce S.; Sarà M. Organism–quartz interactions in structuring benthic communities: towards a marine bio-mineralogy? Ecol. Lett. 2: 1–3; 1999.
Cerrano C.; Calcinai B.; Di Camillo C. G.; Valisano L.; Bavestrello G. How and why do sponges incorporate foreign material? Strategies in Porifera. In: Custódio M. R.; Hajdu E.; Lôbo-Hajdu G.; Muricy G. (eds) Porifera research: biodiversity. Innovation and Sustainability. Museu Nacional, Rio de Janeiro, pp 239–246; 2007a.
Cerrano C.; Sambolino P.; Calcinai B.; Azzini F.; Bavestrello G. Growth of the massive morph of Cliona nigricans (Schmidt, 1862) (Porifera, Clionaidae). Ital J Zool 74: 13–19; 2007b.
Custodio M. R.; Prokic I.; Steffen R.; Koziol C.; Borojevic R.; Brümmer F.; Nickel M.; Müller W. E. G. Primmorphs generated from dissociated cells from the sponge Suberites domuncula: a model system for studies of cell proliferation and cell death. Mech. Ageing. Dev. 105: 45–59; 1998.
Donaldson K.; Borm P. J. The quartz hazard: a variable entity. Ann. Occup. Hyg. 42: 287–294; 1998.
Efremova S. M.; Margulis B. A.; Guzhova I. V.; Itskovich V. B.; Lauenroth S.; Müller W. E. G.; Schröder H. C. Heat shock protein Hsp70 expression and DNA damage in Baikalian sponges exposed to model pollutants and wastewater from Baikalsk pulp and paper plant. Aquat. Toxicol. 57: 267–280; 2002.
Ehrlich H.; Worch H. Collagen: a huge matrix in glass sponge flexible spicules of the meter-long Hyalonema sieboldi. In: Bauerlein E. (ed) Handbook of biomineralisation. Wiley VCH Verlag GmbH&Co, KGaA, Weinheim, pp 23–41; 2007.
Fubini B. Surface chemistry and quartz hazard. Ann. Occup. Hyg. 42: 521–530; 1998.
Giovine M.; Pozzolini M.; Fenoglio I.; Scarfì S.; Ghiazza M.; Benatti U.; Fubini B. Cristalline silica incubated in ascorbic acid acquires a higher cytotoxic potential. Toxicol. Ind. Health 18: 249–255; 2002.
Groppelli S.; Pennati R.; Scarì G.; Sotgia C.; De Bernardi F. Observations on the settlement of Phallusia mammillata larvae: effects of different lithological substrata. Ital. J. Zool. 70: 321–326; 2003.
Guidetti P.; Bianchi C. N.; Chiantore M.; Schiaparelli S.; Morri C.; Cattaneo-Vietti R. Living on the rocks: substrate mineralogy and the structure of subtidal rocky substrate communities in the Mediterranean Sea. Mar. Ecol. Prog. Ser. 274: 57–68; 2004.
Guidetti P.; Cattaneo-Vietti R. Can mineralogical features influence distribution patterns of fish? A case study in shallow Mediterranean rocky reefs. J. Mar. Biol. Ass. UK 82: 1043–1044; 2002.
Herberts R. J. H.; Hawkins S. J. Effect of rock type on the recruitment and early mortality of the barnacle Chthamalus montagui. J. Exp. Mar. Biol. Ecol. 334: 96–108; 2006.
Koziol C.; Borojevic R.; Steffen R.; Müller W. E. G. Sponges (Porifera) model systems to study the shift from immortal to senescent somatic cells: the telomerase activity in somatic cells. Mech. Ageing. Dev. 100: 107–120; 1998.
Krasko A.; Lorenz B.; Batel R.; Schröder H. C.; Müller I. M.; Müller W. E. G. Expression of silicatein and collagen genes in the marine sponge Suberites domuncula is controlled by silicate and myotrophin. Eur. J. Biochem. 267: 4878–87; 2000.
Maradonna F.; Bavestrello G.; Cardinali M.; Olivotto I.; Cerrano C.; Giovine M.; Carnevali O. Role of substrate on larval development of the freshwater Teleost Pelvicachromis pulcher. Mol. Reprod. Dev. 66: 256–262; 2003.
Moniot F.; Martoja R.; Truchet M.; Fröhlich F. Opal in ascidians: a curious bioaccumulation in the ovary. Mar. Biol. 112: 283–292; 1992.
Müller W. E. G.; Rothenberger M.; Boreiko A.; Tremel W.; Reiber A.; Schröeder H. C. Formation of siliceous spicules in the marine demosponge Suberites domuncula. Cell Tissue Res. 321: 285–297; 2005.
Müller W. E. G.; Wiens M.; Batel R.; Steffen R.; Schröder H. C.; Borojevic R.; Custódio M. R. Establishment of a primary cell culture from a sponge: primmorphs from Suberites domuncula. Mar. Ecol. Prog. Ser. 178: 205–219; 1999.
Pozzolini M.; Sturla L.; Cerrano C.; Bavestrello G.; Camardella L.; Parodi A. M.; Raheli F.; Benatti U.; Müller W. E. G.; Giovine M. Molecular cloning of silicatein gene from the marine sponge Petrosia ficiformis (Porifera, Demospongiae) and development of primmorphs as a model for biosilicification studies. Mar. Biotechnol. 6: 594–603; 2004.
Scarfi S.; Benatti U.; Pozzolini M.; Clavarino E.; Ferrais C.; Magnone M.; Valisano L.; Giovine M. Ascorbic acid-pretreated quartz enhances cyclo-oxygenase-2 expression in RAW 264.7 murine macrophages. FEBS J. 274: 60–73; 2007.
Schiaparelli S.; Guidetti P.; Cattaneo-Vietti R. Can mineralogical features affect the distribution patterns of sessile gastropods? The Vermetidae case in the Mediterranean Sea. J. Mar. Biol. Ass. UK 83: 1267–1268; 2002.
Schröder H. C.; Perovic-Ottstadt S.; Grebenjuk V. A.; Engel S.; Müller I. M.; Müller W. E. G. Biosilica formation in spicules of the sponge Suberites domuncula: synchronous expression of a gene cluster. Genomics 85: 666–678; 2005.
Valisano L.; Bavestrello G.; Giovine M.; Cerrano C. Primmorphs formation dynamics: a screening among Mediterranean sponges. Mar. Biol. 149: 1037–1046; 2006.
Vandesompele J.; De Preter K.; Pattyn F.; Poppe B.; Van Roy N.; De Paepe A.; Speleman F. Accurate normalization of real-time quantitative RT-PCR data by geometric averaging of multiple internal control genes. Genome Biol. 3(7): RESEARCH0034.1–RESEARCH0034.11; 2002.
Zhang X.; Cao X.; Zhang W.; Yu X.; Jin M. Primmorphs from Archaeocytes-dominant cell population of the sponge Hymeniacidon perleve: improved cell proliferation and spiculogenesis. Biotechnol. Bioeng. 84: 583–590; 2003.
Acknowledgments
This work was partially supported by EC fund to MG, by Liguria Region, Area Marine Biotechnology Funds to MG, by Italian Research Ministry Fund (PRIN 2007) to MG, and by University of Genova funds to CC. We thank Laura Negretti for technical support in the SEM analysis of rocks. The experiments of this study comply with the current laws of Italy.
Author information
Authors and Affiliations
Corresponding author
Additional information
Editor: J. Denry Sato
Pozzolini and Valisano equally contributed to this work.
Rights and permissions
About this article
Cite this article
Pozzolini, M., Valisano, L., Cerrano, C. et al. Influence of rocky substrata on three-dimensional sponge cells model development. In Vitro Cell.Dev.Biol.-Animal 46, 140–147 (2010). https://doi.org/10.1007/s11626-009-9253-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11626-009-9253-y