Abstract
Background
Treatment decisions for colorectal cancer vary based on lymph node status. While some histopathological features of the primary tumor correlate with lymph node spread, the relative influences of these risk factors are not well quantified.
Objective
This study aims to systematically review published studies relating histopathological features of primary colorectal cancer to the presence of lymph node metastases and to determine how reliable certain factors might be at predicting nodal metastasis when only the primary lesion is available for study.
Data Sources
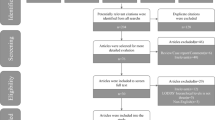
Inclusive literature search using EMBASE and Ovid MEDLINE databases plus manual reference checks of all articles correlating lymphatic spread with colorectal cancer (any T stage) from 1984 to mid-2008 was performed.
Study Selection
This search generated two levels of screening utilized on 602 citations, yielding 123 articles for full review. Data reported from 76 articles were chosen.
Main Outcome Measures
The relative influence of each histopathological feature on the likelihood of lymphatic metastases was determined. Fixed-effects meta-analysis was performed, and results were reported as Mantel–Haenszel odds ratios (OR).
Results
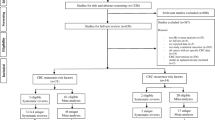
Of 42 histopathological features analyzed, only 40.4% were reported in >2 articles. The positive predictive values for the top quartile of most frequently reported risk factors were 25.5–86.4%. Among the commonly reported histopathological findings, lymphatic invasion (OR, 8.62) significantly outperformed tumor depth (T2 vs. T1; OR, 2.62) and overall differentiation (OR, 2.38) in predicting nodal spread. For the rectal cancer subset, risk factors differed from the overall colorectal group in predictive ability; poor differentiation at the invasive front (OR, 6.08) and tumor budding (OR, 5.82) were the most predictive.
Limitations
This literature search is limited by the small number of studies examining only rectal cancers and the potential changes in histological and/or surgical techniques over the study period.
Conclusions
No single histopathological feature of colorectal cancer reliably predicted lymph node metastases. Several risk factors that correlate highly with nodal disease are not routine components of standard pathology reports. Until further research establishes histopathological or molecular patterns for predicting lymph node spread, caution should be exercised when basing treatment decisions solely on these factors.



Similar content being viewed by others
References
Smith FM, Reynolds JV, Miller N, Stephens RB, Kennedy MJ. Pathological and molecular predictors of the response of rectal cancer to neoadjuvant radiochemotherapy. Eur J Surg Oncol. 2006;32:55–64.
Kuremsky JG, Tepper JE, McLeod HL. Biomarkers for response to neoadjuvant chemoradiation for rectal cancer. Int J Radiat Oncol Biol Phys. 2009;74:673–88.
Walther A, Johnstone E, Swanton C, Midgley R, Tomlinson I, Kerr D. Genetic prognostic and predictive markers in colorectal cancer. Nat Rev Cancer. 2009;9:489–99.
Negri FV, Campanini N, Camisa R, et al. Biological predictive factors in rectal cancer treated with preoperative radiotherapy or radiochemotherapy. Br J Cancer. 2008;98:143–7.
Liersch T, Langer C, Ghadimi BM, et al. Lymph node status and TS gene expression are prognostic markers in stage II/III rectal cancer after neoadjuvant fluorouracil-based chemoradiotherapy. J Clin Oncol. 2006;24:4062–8.
Smith AJ, Driman DK, Spithoff K, et al. Guideline for optimization of colorectal cancer surgery and pathology. J Surg Oncol. 2010;101:5–12.
Kim YW, Kim NK, Min BS, Lee KY, Sohn SK, Cho CH. The influence of the number of retrieved lymph nodes on staging and survival in patients with stage II and III rectal cancer undergoing tumor-specific mesorectal excision. Ann Surg. 2009;249:965–72.
Chang GJ, Rodriguez-Bigas MA, Skibber JM, Moyer VA. Lymph node evaluation and survival after curative resection of colon cancer: systematic review. J Natl Cancer Inst. 2007;99:433–41.
Sakuragi M, Togashi K, Konishi F, et al. Predictive factors for lymph node metastasis in T1 stage colorectal carcinomas. Dis Colon Rectum. 2003;46:1626–32.
You YN, Baxter NN, Stewart A, Nelson H. Is the increasing rate of local excision for stage I rectal cancer in the United States justified?: a nationwide cohort study from the National Cancer Database. Ann Surg. 2007;245:726–33.
Ricciardi R, Madoff RD, Rothenberger DA, Baxter NN. Population-based analyses of lymph node metastases in colorectal cancer. Clin Gastroenterol Hepatol. 2006;4:1522–7.
Tulchinsky H, Shmueli E, Figer A, Klausner JM, Rabau M. An interval >7 weeks between neoadjuvant therapy and surgery improves pathologic complete response and disease-free survival in patients with locally advanced rectal cancer. Ann Surg Oncol. 2008;15:2661–7.
Joseph NE, Sigurdson ER, Hanlon AL, et al. Accuracy of determining nodal negativity in colorectal cancer on the basis of the number of nodes retrieved on resection. Ann Surg Oncol. 2003;10:213–8.
Gopaul D, Belliveau P, Vuong T, et al. Outcome of local excision of rectal carcinoma. Dis Colon Rectum. 2004;47:1780–8.
Madbouly KM, Remzi FH, Erkek BA, et al. Recurrence after transanal excision for T1 rectal cancer: should we be concerned? Dis Colon Rectum. 2005;48:719–21.
Mellgren A, Sirivongs P, Rothenberger DA, Madoff RD, García-Aguilar J. Is local excision adequate therapy for early rectal cancer? Dis Colon Rectum. 2000;43:1064–71; discussion 1071–4.
Paty PB, Nash GM, Baron P, et al. Long-term results of local excision for rectal cancer. Ann Surg. 2002;236:522–30.
Endreseth BH, Myrvold HE, Romundstad P, et al. Transanal excision vs. major surgery for T1 rectal cancer. Dis Colon Rectum. 2005;48:1380–8.
Bentrem DJ, Okabe S, Wong WD, et al. T1 adenocarcinoma of the rectum. Transanal excision or radical surgery? Ann Surg. 2005;242:472–9.
Nascimbeni R, Burgart LJ, Nivatvongs S, Larson DR. Risk of lymph node metastasis in T1 carcinoma of the colon and rectum. Dis Colon Rectum. 2002;45:200–6.
Nash GM, Weiser MR, Guillem JG, et al. Long-term survival after transanal excision of T1 rectal cancer. Dis Colon Rectum. 2009;52:577–82.
Christoforidis D, Cho HM, Dixon MR, Mellgren AF, Madoff RD, Finne CO. Transanal Endoscopic Microsurgery Versus Conventional Transanal Excision for Patients With Early Rectal Cancer. Ann Surg. 2009;249:776.
Garcia-Aguilar J, Mellgren A, Sirivongs P, Buie D, Madoff RD, Rothenberger DA. Local excision of rectal cancer without adjuvant therapy: a word of caution. Ann Surg. 2000;231:345–51.
Aoki R, Tanaka S, Haruma K, et al. MUC-1 expression as a predictor of the curative endoscopic treatment of submucosally invasive colorectal carcinoma. Dis Colon Rectum. 1998;41:1262–72.
Masaki T, Muto T. Predictive value of histology at the invasive margin in the prognosis of early invasive colorectal carcinoma. J Gastroenterol. 2000;35:195–200.
Masaki T, Matsuoka H, Sugiyama M, et al. Budding as a useful determinant of the optimal treatment for T1 rectal carcinomas. Hepatogastroenterology. 2003;50:388–91.
Park YJ, Kim WH, Paeng SS, Park JG. Histoclinical analysis of early colorectal cancer. World J Surg. 2000;24:1029–35.
Ishikawa Y, Akishima-Fukasawa Y, Ito K, et al. Histopathologic determinants of regional lymph node metastasis in early colorectal cancer. Cancer. 2008;112:924–33.
Kurokawa S, Arimura Y, Yamamoto H, et al. Tumour matrilysin expression predicts metastatic potential of stage I (pT1) colon and rectal cancers. Gut. 2005;54:1751–8.
Masaki T, Goto A, Sugiyama M, et al. Possible contribution of CD44 variant 6 and nuclear beta-catenin expression to the formation of budding tumor cells in patients with T1 colorectal carcinoma. Cancer. 2001;92:2539–46.
Saleh HA, Jackson H, Banerjee M. Immunohistochemical expression of bcl-2 and p53 oncoproteins: correlation with Ki67 proliferation index and prognostic histopathologic parameters in colorectal neoplasia. Appl Immunohistochem Mol Morphol. 2000;8:175–82.
Guzińska-Ustymowicz K. MMP-9 and cathepsin B expression in tumor budding as an indicator of a more aggressive phenotype of colorectal cancer (CRC). Anticancer Res. 2006;26:1589–94.
Kaneko I, Tanaka S, Oka S, et al. Immunohistochemical molecular markers as predictors of curability of endoscopically resected submucosal colorectal cancer. World J Gastroenterol. 2007;13:3829–35.
Oh-e H, Tanaka S, Kitadai Y, Shimamoto F, Yoshihara M, Haruma K. Cathepsin D expression as a possible predictor of lymph node metastasis in submucosal colorectal cancer. Eur J Cancer. 2001;37:180–8.
Elpek GO, Gelen T, Karpuzoglu G, Karpuzoglu T, Aksoy NH, Keles N. Clinicopathologic evaluation of CDw75 antigen expression in colorectal adenocarcinomas. Pathol Oncol Res. 2002;8:175–82.
Sitzler PJ, Seow-Choen F, Ho YH, Leong AP. Lymph node involvement and tumor depth in rectal cancers: an analysis of 805 patients. Dis Colon Rectum. 1997;40:1472–6.
Shinto E, Jass JR, Tsuda H, et al. Differential prognostic significance of morphologic invasive markers in colorectal cancer: tumor budding and cytoplasmic podia. Dis Colon Rectum. 2006;49:1422–30.
Goldstein NS, Hart J. Histologic features associated with lymph node metastasis in stage T1 and superficial T2 rectal adenocarcinomas in abdominoperineal resection specimens. Identifying a subset of patients for whom treatment with adjuvant therapy or completion abdominoperineal resection should be considered after local excision. Am J Clin Pathol. 1999;111:51–8.
Hase K, Shatney CH, Mochizuki H, et al. Long-term results of curative resection of “minimally invasive” colorectal cancer. Dis Colon Rectum. 1995;38:19–26.
Kaneko I, Tanaka S, Oka S, et al. Lymphatic vessel density at the site of deepest penetration as a predictor of lymph node metastasis in submucosal colorectal cancer. Dis Colon Rectum. 2007;50:13–21.
Kojima M, Shiokawa A, Ohike N, et al. Clinical significance of nuclear morphometry at the invasive front of T1 colorectal cancer and relation to expression of VEGF-A and VEGF-C. Oncology. 2005;68:230–8.
Masaki T, Matsuoka H, Sugiyama M, Abe N, Sakamoto A, Atomi Y. Actual number of tumor budding as a new tool for the individualization of treatment of T1 colorectal carcinomas. J Gastroenterol Hepatol. 2006;21:1115–21.
Okuyama T, Oya M, Yamaguchi M. Budding (sprouting) as a useful prognostic marker in colorectal mucinous carcinoma. Jpn J Clin Oncol. 2002;32:412–6.
Sohn DK, Chang HJ, Park JW, et al. Histopathological risk factors for lymph node metastasis in submucosal invasive colorectal carcinoma of pedunculated or semipedunculated type. J Clin Pathol. 2007;60:912–5.
Ueno H, Mochizuki H, Shinto E, Hashiguchi Y, Hase K, Talbot IC. Histologic indices in biopsy specimens for estimating the probability of extended local spread in patients with rectal carcinoma. Cancer. 2002;94:2882–91.
Ueno H, Mochizuki H, Hashiguchi Y, et al. Risk factors for an adverse outcome in early invasive colorectal carcinoma. Gastroenterology. 2004;127:385–94.
Brodsky JT, Richard GK, Cohen AM, Minsky BD. Variables correlated with the risk of lymph node metastasis in early rectal cancer. Cancer. 1992;69:322–6.
Huddy SP, Husband EM, Cook MG, Gibbs NM, Marks CG, Heald RJ. Lymph node metastases in early rectal cancer. Br J Surg. 1993;80:1457–8.
Kikuchi R, Takano M, Takagi K, et al. Management of early invasive colorectal cancer. Risk of recurrence and clinical guidelines. Dis Colon Rectum. 1995;38:1286–95.
Minsky BD, Rich T, Recht A, Harvey W, Mies C. Selection criteria for local excision with or without adjuvant radiation therapy for rectal cancer. Cancer. 1989;63:1421–9.
Nelson JC, Nimr AN, Thomford NR. Criteria for the selection of ‘early’ carcinomas of the rectum. Are they valid? Arch Surg. 1987;122:533–6.
Tanaka S, Yokota T, Saito D, Okamoto S, Oguro Y, Yoshida S. Clinicopathologic features of early rectal carcinoma and indications for endoscopic treatment. Dis Colon Rectum. 1995;38:959–63.
Arai T, Akiyama Y, Yamamura A, et al. Allelotype analysis of early colorectal cancers with lymph node metastasis. Int J Cancer. 1998;79:418–23.
Blumberg D, Paty PB, Guillem JG, et al. All patients with small intramural rectal cancers are at risk for lymph node metastasis. Dis Colon Rectum. 1999;42:881–5.
Masaki T, Sugiyama M, Atomi Y, et al. The indication of local excision for T2 rectal carcinomas. Am J Surg. 2001;181:133–7.
Fang WL, Chang SC, Lin JK, et al. Metastatic potential in T1 and T2 colorectal cancer. Hepatogastroenterology. 2005;52:1688–91.
Maeda K, Yashiro M, Nishihara T, et al. Correlation between vascular endothelial growth factor C expression and lymph node metastasis in T1 carcinoma of the colon and rectum. Surg Today. 2003;33:736–9.
Okabe S, Shia J, Nash G, et al. Lymph node metastasis in T1 adenocarcinoma of the colon and rectum. J Gastrointest Surg. 2004;8:1032–9; discussion 1039–40.
Shimomura T, Ishiguro S, Konishi H, et al. New indication for endoscopic treatment of colorectal carcinoma with submucosal invasion. J Gastroenterol Hepatol. 2004;19:48–55.
Suzuki T, Sadahiro S, Mukoyama S, et al. Risk of lymph node and distant metastases in patients with early invasive colorectal cancer classified as Haggitt’s level 4 invasion: image analysis of submucosal layer invasion. Dis Colon Rectum. 2003;46:203–8.
Yamamoto S, Watanabe M, Hasegawa H, et al. The risk of lymph node metastasis in T1 colorectal carcinoma. Hepatogastroenterology. 2004;51:998–1000.
Abe A, Fukui H, Fujii S, et al. Involvement of cyclooxygenase-2 and vascular endothelial growth factor in vascularization and lymph node metastasis of colorectal cancers with submucosal invasion. J Gastroenterol Hepatol. 2007;22:1071–7.
Chok KS, Law WL. Prognostic factors affecting survival and recurrence of patients with pT1 and pT2 colorectal cancer. World J Surg. 2007;31:1485–90.
Masaki T, Matsuoka H, Sugiyama M, et al. Tumor budding and evidence-based treatment of T2 rectal carcinomas. J Surg Oncol. 2005;92:59–63.
Rasheed S, Bowley DM, Aziz O, et al. Can depth of tumour invasion predict lymph node positivity in patients undergoing resection for early rectal cancer? A comparative study between T1 and T2 cancers. Colorectal Dis. 2008;10:231–8.
Wang HS, Liang WY, Lin TC, et al. Curative resection of T1 colorectal carcinoma: risk of lymph node metastasis and long-term prognosis. Dis Colon Rectum. 2005;48:1182–92.
Yasuda K, Inomata M, Shiromizu A, Shiraishi N, Higashi H, Kitano S. Risk factors for occult lymph node metastasis of colorectal cancer invading the submucosa and indications for endoscopic mucosal resection. Dis Colon Rectum. 2007;50:1370–6.
Garinis GA, Manolis EN, Spanakis NE, Patrinos GP, Peros G, Menounos PG. High frequency of concomitant nm23-H1 and E-cadherin transcriptional inactivation in primary non-inheriting colorectal carcinomas. J Mol Med. 2003;81:256–63.
Guzińska-Ustymowicz K, Chetnik A, Kemona A. Effects of changes at the site of E-cadherin expression as an indicator of colon cancer aggressiveness. Rocz Akad Med Bialymst. 2004;49 Suppl 1:70–2.
Pan W, Terai T, Abe S, et al. Location of early colorectal cancers at fold-top may reduce the risk of lymph node metastasis. Dis Colon Rectum. 2006;49:579–87.
Sakashita M, Aoyama N, Minami R, et al. Glut1 expression in T1 and T2 stage colorectal carcinomas: its relationship to clinicopathological features. Eur J Cancer. 2001;37:204–9.
Lenander C, Habermann JK, Ost A, et al. Laminin-5 gamma 2 chain expression correlates with unfavorable prognosis in colon carcinomas. Anal Cell Pathol. 2001;22:201–9.
Kubo H, Miki C, Kusunoki M. Evaluation of genetic mutations of tumor suppresser genes in colorectal cancer patients. Hepatogastroenterology. 2004;51:114–7.
Kazama S, Watanabe T, Ajioka Y, Kanazawa T, Nagawa H. Tumour budding at the deepest invasive margin correlates with lymph node metastasis in submucosal colorectal cancer detected by anticytokeratin antibody CAM5.2. Br J Cancer. 2006;94:293-8.
Ishikawa F, Saito N, Koda K, et al. Nuclear morphometric analysis of T2 lesions of the rectum—a simple, reproducible method for predicting malignancy potential. Am J Surg. 2002;183:686–91.
Makino M, Yamane N, Taniguchi T, Honboh T, Kurayoshi K, Kaibara N. p53 as an indicator of lymph node metastases in invasive early colorectal cancer. Anticancer Res. 2000;20:2055–9.
Sory A, Minamoto T, Ohta T, et al. Does p53 overexpression cause metastases in early invasive colorectal adenocarcinoma? Eur J Surg. 1997;163:685–92.
Starzynska T, Bromley M, Ghosh A, Stern PL. Prognostic significance of p53 overexpression in gastric and colorectal carcinoma. Br J Cancer. 1992;66:558–62.
Kasakura Y, Fujii M, Mochizuki F, Kobayashi M, Yamagata M. Clinicopathological analyses of advanced colorectal cancers of different sizes—especially those of 20 mm or less in diameter. Hepatogastroenterology. 2001;48:696–701.
Fujiya M, Watari J, Ashida T, et al. Reduced expression of syndecan-1 affects metastatic potential and clinical outcome in patients with colorectal cancer. Jpn J Cancer Res. 2001;92:1074–81.
Guzinska-Ustymowicz K, Kemona A. Transforming growth factor beta can be a parameter of aggressiveness of pT1 colorectal cancer. World J Gastroenterol. 2005;11:1193–5.
Xiong B, Gong LL, Zhang F, Hu MB, Yuan HY. TGF beta1 expression and angiogenesis in colorectal cancer tissue. World J Gastroenterol. 2002;8:496–8.
Famulski W, Guzińska-Ustymowicz K, Sulkowska M, et al. Tumour budding intensity in relation to cathepsin D expression and some clinicopathological features of colorectal cancer. Folia Histochem Cytobiol. 2001;39 Suppl 2:171–2.
Hase K, Shatney C, Johnson D, Trollope M, Vierra M. Prognostic value of tumor “budding” in patients with colorectal cancer. Dis Colon Rectum. 1993;36:627–35.
Okuyama T, Nakamura T, Yamaguchi M. Budding is useful to select high-risk patients in stage II well-differentiated or moderately differentiated colon adenocarcinoma. Dis Colon Rectum. 2003;46:1400–6.
Guzińska-Ustymowicz K. The role of tumour budding at the front of invasion and recurrence of rectal carcinoma. Anticancer Res. 2005;25:1269–72.
Hörkkö TT, Klintrup K, Mäkinen JM, et al. Budding invasive margin and prognosis in colorectal cancer—no direct association with beta-catenin expression. Eur J Cancer. 2006;42:964–71.
Choi HJ, Park KJ, Shin JS, Roh MS, Kwon HC, Lee HS. Tumor budding as a prognostic marker in stage-III rectal carcinoma. Int J Colorectal Dis. 2007;22:863–8.
Kanazawa H, Mitomi H, Nishiyama Y, et al. Tumour budding at invasive margins and outcome in colorectal cancer. Colorectal Dis. 2008;10:41–7.
Fried GM, Hreno A, Duguid WP, Hampson LG. Rational management of malignant colon polyps based on long-term follow-up. Surgery. 1984;96:815–22.
Acknowledgement
The authors thank Howard Monroe, Ph.D., of Genesis Concepts & Consultants, for providing statistical support and meta-analysis under AFMSA/SG5M South Contract No. FA7014-09-D-0008, Brooks City-Base, TX, USA.
Author information
Authors and Affiliations
Corresponding author
Additional information
This study was supported exclusively using institutional funds.
Rights and permissions
About this article
Cite this article
Glasgow, S.C., Bleier, J.I.S., Burgart, L.J. et al. Meta-analysis of Histopathological Features of Primary Colorectal Cancers that Predict Lymph Node Metastases. J Gastrointest Surg 16, 1019–1028 (2012). https://doi.org/10.1007/s11605-012-1827-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11605-012-1827-4