Abstract
Several bacterial and host-related factors concur in causing Helicobacter pylori eradication failure. We ascertained the role of bacterial virulence genes (cagA, vacA), clarithromycin resistance [ClaR, 23S ribosomal RNA (rRNA) mutations], host polymorphism of CYP2C19 (polyphosphoinositide, PPI, metabolism) and of the cytokines IL-1B-31C>T, IL-1RN VNTR, IFN-γ+874A>T, TNF-α-1031T>C, TNF-α-857C>T, TNF-α-376G>A, TNF-α-308G>A, TNF-α-238G>A, IL-10-1082A>G, IL-10-819C>T, IL-10-592C>A, IL-12A+6686G>A, IL-12B+15485A>C. Two groups of H. pylori-infected and H. pylori-treated patients were retrospectively identified: 45 not eradicated and 57 eradicated. Treatment failure was significantly correlated with ClaR (all resistant strains in non-eradicated patients); with TNF-α-238, IL10-819, IL10-592, IL-12B+15485 single nucleotide polymorphism (SNP); with IL10 ATA/ATA haplotype; and with antral inflammatory grade. On considering ClaS-infected patients only, logistic regression analysis (eradication = dependent; TNF-α-238, IL12B + 15485 genotypes, IL10 ATA/ATA as present or absent, antral gastritis grade = covariates) confirmed as significantly correlated with eradication antral gastritis grade only (Exp(B) = 6.48; 95% CI, 1.2–35.01). In conclusion, the bacterial determinant causing triple therapy failure is clarithromycin resistant, being virulence genes not involved. The host related factors that favor eradication are those linked to inflammation: a higher inflammatory infiltrate in the mucosa, possibly favored by genotypes able to down regulate the anti-inflammatory cytokine response, enhance the chance of eradication success.



Similar content being viewed by others
Abbreviations
- ClaR :
-
Clarithromycin resistant
- ClaS :
-
Clarithromycin sensitive
- EM:
-
extensive metabolizer
- IM:
-
intermediate metabolizer
- MGB:
-
DNA minor groove binder
- PAI:
-
pathogenicity island
- PCR:
-
polymerase chain reaction
- PM:
-
poor metabolizer
- PPI:
-
proton pump inhibitor
- RFLP:
-
restriction fragment length polymorphism
- rRNA:
-
ribosomal RNA
- SNP:
-
single nucleotide polymorphism
- UBT:
-
urea breath test
- UM:
-
ultrarapid metabolizer
- VNTR:
-
variable number of tandem repeats
- 6-FAM:
-
6-carboxyfluorescin
References
Dunn BE, Cohen H, Blaser MJ. Helicobacter pylori. Clin Microbiol Rev 1997;10:720–741.
Brown ML. Helicobacter pylori: epidemiology and routes of transmission. Epidemiol Rev 2000;22:283–297.
Montecucco C, Rappuoli R. Living dangerously: how Helicobacter pylori survives in the human stomach. Nat Rev Mol Cell Biol 2001;2:457–466.
Suerbaum S, Michetti P. Helicobacter pylori infection. New Engl J Med 2002;347:1175–1186.
Malfertheiner P, Megraud F, O’morain C, Bazzoli F, El-Omar E, Graham D, Hunt R, Rokkas T, Vakil N, Kuipers EJ. Current concepts in the management of Helicobacter pylori infection—the Maastricht III consensus report. Gut 2007;56:772–781.
Blaser MJ, Chyou PH, Nomura A. Age at establishment of Helicobacter pylori infection and gastric adenocarcinoma, gastric ulcer, and duodenal ulcer risk. Cancer Res 1995;55:562–565.
Blaser MJ, Perez-Perez GI, Kleanthous H, Cover TL, Peek RM, Chyou PH, Stemmermann GN, Nomura A. Infection with Helicobacter pylori strains possessing cagA is associated with an increased risk of developing adenocarcinoma of the stomach. Cancer Res 1995;55:2111–2115.
Parsonnet J, Friedman GD, Orentreich N, Vogelman H. Risk for gastric cancer in people with CagA positive or CagA negative Helicobacter pylori infection. Gut 1997;40:297–301.
Atherton JC, Peek RM, Tham KT, Cover TL, Blaser MJ. Clinical and pathological importance of heterogeneity in vacA, the vacuolating cytotoxin gene of Helicobacter pylori. Gastroenterology 1997;112:92–99.
Basso D, Navaglia F, Brigato L, Piva MG, Toma A, Greco E, Di Mario F, Galeotti F, Roveroni G, Corsini A, Plebani M. Analysis of Helicobacter pylori vacA and cagA genotypes and serum antibody profile in benign and malignant gastroduodenal diseases. Gut 1998;43:182–186.
Zambon C-F, Navaglia F, Basso D, Rugge M, Plebani M. Helicobacter pylori babA2, cagA, and s1 vacA genes work synergistically in causing intestinal metaplasia. J Clin Pathol 2003;56:287–291.
Covacci A, Rappuoli R. Tyrosine-phosphorylated bacterial proteins: Trojan horses for the host cell. J Exp Med 2000;191:587–592.
Papini E, Zoratti M, Cover TL. In search of the Helicobacter pylori VacA mechanism of action. Toxicon 2001;39:1757–1767.
Basso D, Scrigner M, Toma A, Navaglia F, Di Mario F, Rugge M, Plebani M. Helicobacter pylori infection enhances mucosal interleukin-1 beta, interleukin-6, and the soluble receptor of interleukin-2. Int J Clin Lab Res 1996;26:207–210.
Lindholm C, Quiding-Jarbrink M, Lonroth H, Hamlet A, Svennerholm A-M. Local cytokine response in Helicobacter pylori-infected subjects. Infect Immun 1998;66:5964–5971.
Bontems P, Robert F, Van Gossum A, Cadranel S, Mascart F. Helicobacter pylori modulation of gastric and duodenal mucosal T cell cytokine secretion in children compared with adults. Helicobacter 2003;8:216–226.
Beales ILP, Calam J. Interleukin 1β and tumour necrosis factor a inhibit acid secretion in cultured rabbit parietal cells by multiple pathways. Gut 1998;42:227–234.
Takashima M, Furuta T, Hanai H, Sugimura H, Kaneko E. Effects of Helicobacter pylori infection on gastric acid secretion and serum gastrin levels in Mongolian gerbils. Gut 2001;48:765–773.
El-Omar EM, Carrington M, Chow W-H, McColl KE, Bream JH, Young HA, Herrera J, Lissowska J, Yuan CC, Rothman N, Lanyon G, Martin M, Fraumeni JF Jr, Rabkin CS. Interleukin-1 polymorphisms associated with increased risk of gastric cancer. Nature 2000;404:398–402 (Erratum in: Nature 2001;412:99).
Machado JC, Pharoah P, Sousa S, Carvalho R, Oliveira C, Figueiredo C, Amorim A, Seruca R, Caldas C, Carneiro F, Sobrinho-Simoes M. Interleukin 1B and interleukin 1RN polymorphisms are associated with increased risk of gastric carcinoma. Gastroenterology 2001;121:823–829.
Figueiredo C, Machado JC, Pharoah P, Seruca R, Sousa S, Carvalho R, Capelinha AF, Quint W, Caldas C, van Doorn LJ, Carneiro F, Sobrinho-Simoes M. Helicobacter pylori and interleukin 1 genotyping: an opportunity to identify high-risk individuals for gastric carcinoma. J Natl Cancer Inst 2002;94:1680–16877.
Zambon C-F, Basso D, Navaglia F, Germano G, Gallo N, Milazzo M, Greco E, Fogar P, Mazza S, Di Mario F, Basso G, Rugge M, Plebani M. Helicobacter pylori virulence genes and host IL-1RN and IL-1 beta genes interplay in favouring the development of peptic ulcer and intestinal metaplasia. Cytokine 2002;18:242–251.
Zambon C-F, Basso D, Navaglia F, Falda A, Belluco C, Fogar P, Greco E, Gallo N, Farinati F, Cardin R, Rugge M, Di Mario F, Plebani M. Increased risk of noncardia gastric cancer associated with proinflammatory cytokine gene polymorphisms. Gastroenterology 2004;126:382–383.
Zambon C-F, Basso D, Navaglia F, Belluco C, Falda A, Fogar P, Greco E, Gallo N, Rugge M, Di Mario F, Plebani M. Pro- and anti-inflammatory cytokines gene polymorphisms and Helicobacter pylori infection: interactions influence outcome. Cytokine 2005;29:141–152.
Kunstmann E, Epplen C, Elitok E, Harder M, Suerbaum S, Peitz U, Schmiegel W, Epplen JT. Helicobacter pylori infection and polymorphisms in the tumor necrosis factor region. Electrophoresis 1999;20:1756–1761.
Yea SS, Yang Y-I, Jang WH, Lee YJ, Bae HS, Paik KH. Association between TNF-α promoter polymorphism and Helicobacter pylori cagA subtype infection. J Clin Pathol 2001;54:703–706.
Machado JC, Figueiredo C, Canedo P, Pharoah P, Carvalho R, Nabais S, Castro Alves C, Campos ML, Van Doorn LJ, Caldas C, Seruca R, Carneiro F, Sobrinho-Simoes M. A proinflammatory genetic profile increases the risk for chronic atrophic gastritis and gastric carcinoma. Gastroenterology 2003;125:364–371.
Thye T, Burchard GD, Nilius M, Muller-Myhsok B, Horstmann RD. Genomewide linkage analysis identifies polymorphisms in the human interferon-g receptor affecting Helicobacter pylori infection. Am J Hum Genet 2003;72:448–453.
El-Omar EM, Rabkin CS, Gammon MD, Vaughan TL, Risch HA, Schoenberg JB, Stanford JL, Mayne ST, Goedert J, Blot WJ, Fraumeni JF Jr, Chow WH. Increased risk of noncardia gastric cancer associated with proinflammatory cytokine gene polymorphisms. Gastroenterology 2003;124:1193–1201.
Navaglia F, Basso D, Zambon C-F, Ponzano E, Caenazzo L, Gallo N, Falda A, Belluco C, Fogar P, Greco E, Di Mario F, Rugge M, Plebani M. Interleukin 12 gene polymorphisms enhance gastric cancer risk in H. pylori infected individuals. J Med Genet 2005;42:503–510.
Ford AC, Delaney BC, Forman D, Moayyedi P. Eradication therapy for peptic ulcer disease in Helicobacter pylori positive patients. Cochrane Database Syst Rev 2006;2:CD003840.
Farinha P, Gascoyne RD. Helicobacter pylori and MALT lymphoma. Gastroenterology 2005;128:1579–1605.
Malfertheiner P, Megraud F, O’Morain C, Hungin AP, Jones R, Axon A, Graham DJ, Tytgat G, European Helicobacter Pylori Study Group (EHPSG). Current concepts in the management of Helicobacter pylori infection-The Maastricht 2—2000 consensus report. Aliment Pharmacol Ther 2002;16:167–180.
Della Monica P, Lavagna A, Masoero G, Lombardo LC. Effectiveness of Helicobacter pylori eradication treatments in a primary care setting in Italy. Aliment Pharmacol Ther 2002;16:1269–1275.
Calvet X, Ducons J, Bujanda L, Bory F, Montserrat A, Gisbert JP, Hp Study Group of the Asociacion Espanola de Gastroenterologia. Seven versus ten days of rabeprazole triple therapy for Helicobader pylori eradication: a multicenter randomized trial. Am J Gastroenterol 2005;100:1696–1701.
Mégraud F, Lamouliatte H. The treatment of refractory Helicobacter pylori infections. Aliment Pharmacol Ther 2003;17:1333–1343.
van Doorn L-J, Debet-Ossenkopp YJ, Marais A, Sanna R, Megraud F, Kusters JG, Quint WGV. Rapid detection, by PCR and reverse hybridization, of mutations in the Helicobacter pylori 23S rRNA gene, associated with macrolide resistance. Antimicrob Agents Chemother 1999;43:1779–1782.
van Doorn L-J; Glupczynski Y, Kusters JG, Megraud F, Midolo P, Maggi-Solcà, Queiroz DMM, Nouhan N, Stet E, Quint WGV. Accurate prediction of macrolide resistance in Helicobacter pylori by a PCR line probe assay for detection of mutations in the 23S rRNA gene: multicenter validation study. Antimicrob Agents Chemother 2001;45:1500–1504.
Furuta T, Shirai N, Sugimoto M, Nakamura A, Hishida A, Ishizaki T. Influence of CYP2C19 pharmacogenetic polymorphism on proton pump inhibitor-based therapies. Drug Metab Pharmacokinet 2005;20:153–167.
Sim SC, Risinger C, Dahl M-L, Aklillu E, Christensen M, Bertilsson L, Ingelman-Sundberg M. A novel CYP2C19 gene variant causes ultrarapid drug metabolism relevant for the drug response to proton pump inhibitors and antidepressants. Clin Pharmacol Ther 2006;79:103–113.
Schneider S, Roessli D, Excoffier L. Arlequin ver. 2.000: a software for population genetics data analysis. Switzerland: Genetics and Biometry Laboratory, University of Geneva.
Cavallaro LG, Egan B, O’Morain C, Di Mario F. Treatment of Helicobacter pylori infection. Helicobacter 2006;11:36–39.
Pilotto A, Rassu M, Leandro G, Franceschi M, Di Mario. Prevalence of Helicobacter pylori resistance to antibiotics in Northeast Italy: a multicenter study. Dig Liver Dis 2000;32:763–768.
Toracchio S, Marzio L. Primary and secondary Helicobacter pylori resistance of Helicobacter pylori strains isolated in central Italy during the years 1998-2002. Dig Liver Dis 2003;35:541–545.
De Francesco V, Margiotta M, Zullo A, Hassan C, Valle ND, Burattini O, Cea U, Stoppino G, Amoruso A, Stella F, Morini S, Panella C, Ierardi E. Primary clarithromycin resistance in Italy assessed on Helicobacter pylori DNA sequences by TaqMan real-time polymerase chain reaction. Aliment Pharmacol Ther 2006;23:429–435.
Megraud F. H. pylori antibiotic resistance: prevalence, importance, and advances in testing. Gut 2004;53:1374–1384.
van Doorn L-J, Schneeberger PM, Nouhan N, Plaisier AP, Quint WGV, de Boer WA. Importance of Helicobacter pylori cagA and vacA status for the efficacy of antibiotic treatment. Gut 2000;46:321–326.
Suzuki T, Matsuo K, Sawaki A, Ito H, Hirose K, Wakai K, Sato S, Nakamura T, Yamao K, Ueda R, Tajima K. Systematic review and meta-analysis: importance of CagA status for successful eradication of Helicobacter pylori infection. Aliment Pharmacol Ther 2006;24:273–280.
Rudi J, Reuther S, Sieg A, Hoerner M, Stremmel W. Relevance of underlying disease and bacterial vacA and cagA status on the efficacy of Helicobacter pylori eradication. Digestion 2002;65:11–15.
De Francesco V, Zullo A, Margiotta M, Marangi S, Burattini O, Berloco P, Russo F, Barone M, Di Leo A, Minenna MF, Stoppino V, Morini S, Panella C, Francavilla A, Ierardi E. Sequential treatment for Helicobacter pylori does not share the risk factors of triple therapy failure. Aliment Pharmacol Ther 2004;19:407–414.
Kurzawski M, Gawronska-Szklarz B, Wrzesniewska J, Siuda A, Starzynska T, Drozdzik M. Effect of CYP2C19*17 gene variant on Helicobacter pylori eradication in peptic ulcer patients. Eur J Clin Pharmacol 2006;62:877–880.
Borody T, Ren Z, Pang G, Clancy R. Impaired host immunity contributes to Helicobacter pylori eradication failure. Am J Gastroenterol 2002;97:3032–3037.
Author information
Authors and Affiliations
Corresponding author
Additional information
DISCUSSION
Kimberly M. Dalal, M.D. (Travis AFB, CA): I would like to commend Dr. Zambon and Dr. Pedrazzoli and their colleagues for their work and thank them for submitting their manuscript to me for comment.
They examined the issue of H. pylori eradication failure, which is clinically important as this may lead to not only peptic ulcers but also gastric adenocarcinoma or MALToma in a subset of patients. They ascertained the association between triple therapy efficacy and various methods of failure, including clarithromycin‑resistance genes, bacterial virulence genes, and host polymorphisms of PPI metabolism as well as various cytokines. Treatment failure was noted to be significantly correlated with clarithromycin resistance and antral gastritis grade. Moreover, inflammation seemed to favor eradication. I have two questions.
You demonstrated that severe antral inflammatory grade is correlated with a higher rate of successful therapy, and you also showed that patients bearing the ATA/ATA genotype who are low IL‑10 producers and develop more severe inflammation were found to be at a higher frequency in the eradicated than the non‑eradicated group. How do you reconcile this role of inflammation with eradication in some patients but also a higher risk of cancer development in others?
Sergio Pedrazzoli, M.D. (Padova, Italy): We found that more inflammation was better and was due to the onset of cytokines locally. Now, there are enough patient studies to tell you what happens with cancers in these patients. On the other hand, the ATA haplotype is significantly associated with lower production of interleukin 10, and this may explain why the result was different in this kind of patient.
Dr. Dalal:My second question is: you mentioned that bacterial resistance to amoxicillin among Italian patients is low while clarithromycin resistance is more prevalent. How will your data that you have shown today change your approach to patient management in Italy?
Dr. Pedrazzoli: The findings from the Maastricht III Consensus Report indicated that you have to change therapy when you have a prevalence higher than 20%. You need to know if the patient is resistant and then change therapy. We in Italy are in the range lower than 20%. But when you have a prevalence that is higher, you need to look for resistance. Otherwise you treat the patient, observe the patient, and if the patient does not recover, you can change therapy.
Jonathan F. Critchlow, M.D. (Boston, MA): You had a high level of resistant patients. You had a 45% resistance rate.
Dr. Pedrazzoli: Sorry, this was a mistake. I am not the specialist on the matter because he was not able to come here. When preparing the discussion, I posed the same question. Allow me to explain why.
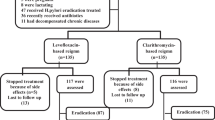
It is a selection bias because we selected from among 800 patients a group of patients who responded and a group of patients who did not respond and compared the pattern of patient characteristics and H. pylori characteristics that would influence the response. But our response rate to triple therapy is about 80‑85%, as is usually observed.
Rights and permissions
About this article
Cite this article
Zambon, CF., Fasolo, M., Basso, D. et al. Clarithromycin Resistance, Tumor Necrosis Factor Alpha Gene Polymorphism and Mucosal Inflammation Affect H. pylori Eradication Success. J Gastrointest Surg 11, 1506–1514 (2007). https://doi.org/10.1007/s11605-007-0246-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11605-007-0246-4