Summary
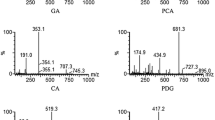
A rapid, sensitive, and selective liquid chromatography-tandem mass spectrometry was developed for the simultaneous determination of lycorine and galanthamine, two major constituents in Lycoris radiata extract, in rat plasma. Liquid-liquid extraction with ethyl ether was carried out using diphenhydramine as the internal standard. The two bioactive alkaloids were separated on a Zorbax SB-C18 reserved-phase column (150 mm × 4.6 mm, i.d., 5 μm) by gradient elution using a mobile phase consisting of methanol with 0.1% formic acid (A) and water with 0.1% formic acid (B) at a flow rate of 0.6 mL/min. All analytes showed good linearity over a wide concentration range (r 2>0.99) and the lower limit of quantification was 3.00 ng/mL for each analyte. The average extraction recovery of the analytes from rat plasma was more than 82.15%, and the intra-day and inter-day accuracy and precision of the assay were less than 12.6%. The validated method was successfully applied to monitoring the concentrations and pharmacokinetic studies of two Amaryllidaceous alkaloids in rat plasma after an oral administration of Lycoris radiata extract.
Similar content being viewed by others
References
Wang L, Zhang X, Yin Z, et al. Two new Amaryllidaceae alkaloids from the bulbs of Lycoris radiate. Chem Pharm Bull, 2009, 57(6):610–611
Hao B, Shen S, Zhao Q. Cytotoxic and antimalarial Amaryllidaceae alkaloids from the bulbs of Lycoris radiate. Molecules, 2013, 18(3):2458–2468
Jin Z. Amaryllidaceae and Sceletium alkaloids. Nat Prod Rep, 2007, 24(4): 886–905
Sener B, Orhan I, Satayavivad J. Antimalarial activity screening of some alkaloids and the plant extracts from Amaryllidaceae. Phytother Res, 2003, 17(10):1220–1223
Szlávik L, Gyuris A, Minárovits J, et al. Alkaloids from Leucojum vernum and antiretroviral activity of Amaryllidaceae alkaloids. Planta Med, 2004, 70(9):871–873
Liu XS, Jiang J, Jiao XY, et al. Lycorine induces apoptosis and down-regulation of Mcl-1 in human leukemia cells. Cancer Lett, 2009, 274(1):16–24
Thomsen T, Kewitz H. Selective inhibition of human acetylcholinesterase by galanthamine in vitro and in vivo. Life Sci, 1990, 46(21):1553–1558
Tariot PN, Solomon PR, Morris JC, et al. A 5-month, randomized, placebo-controlled trial of galantamine in AD. The Galantamine USA-10 Study Group. Neurology, 2000, 54(12): 2269–2276
Azevedo Marques L, Giera M, Lingeman H, et al. Analysis of acetylcholinesterase inhibitors: bioanalysis, degradation and metabolism. Biomed Chromatogr, 2011, 25(1–2):278–299
Hwang YC, Chu JJ, Yang PL, et al. Rapid identification of inhibitors that interfere with poliovirus replication using a cell-based assay. Antiviral Res, 2008, 77(3):232–236
Ghosal S, Singh SK, Kumar Y, et al. The role of ungeremine in the growth-inhibiting and cytotoxic effects of lycorine: evidence and speculation. Planta Med, 1988, 54(2):114–116
Shakirov R, Telezhenetskaya MV, Bessonova IA, et al. Alkaloids, plants, structures, properties. Chem Natl Comp, 1996, 32(3):596–675
Nirogi RV, Kandikere VN, Mudigonda K, et al. Quantitative determination of galantamine in human plasma by sensitive liquid chromatography-tandem mass spectrometry using loratadine as an internal standard. J Chromatogr Sci, 2007, 45(2):97–103
Verhaeghe T, Diels L, de Vries R, et al. Development and validation of a liquid chromatographic-tandem mass spectrometric method for the determination of galantamine in human heparinised plasma. J Chromatogr B Analyt Technol Biomed Life Sci, 2003, 789(2):337–346
Noetzli M, Ansermot N, Dobrinas M, et al. Simultaneous determination of antidementia drugs in human plasma: Procedure transfer from HPLC-MS to UPLC-MS/MS. J Pharm Biomed Anal, 2012, 64–65:16–25
Li X, Xiong YF, Jiang LH, et al. Extraction of galanthamine and lycorine from Lycoris Herb. with one-step method. Huagong Jinzhan (Chinese), 2008, 27:904–912
US DHHS, FDA, CDER. Guidance for Industry: Bioequivalence Guidance. US Department of Health and Human Services, Food and Drug Administration, Center for Veterinary Medicine, (2006). Available from: http://www/fda.gov/cder/guidance/index.htm.
Li MK, Zhang YQ, Dong ZR, et al. Determination of galanthamine, lycoramine and lycorine in Lycoris radiate by high performance liquid chromatography. J Instr Anal, 2012, 31(8): 957–961
Li X, Xiong HR, Jiang LH, et al. Determination of galanthamine and lycorine in Lycoris Herb. by HPLC. Chin J Appl Chem (Chinese), 2010, 27(11):1362–1364
Zhao QY, Verhaeghe T, Truyen L. Pharmacokinetics and safety of galantamine in subjects with hepatic impairment and healthy volunteers. J Clin Pharm, 2002, 42(4):428–436
Author information
Authors and Affiliations
Corresponding author
Additional information
Both authors contributed equally to this work.
This project was supported by the Natural Science Foundation of Fujian Province of China (No. 2013J01382).
Rights and permissions
About this article
Cite this article
Zhou, X., Liu, Yb., Huang, S. et al. An LC-MS/MS method for the simultaneous determination of lycorine and galanthamine in rat plasma and its application to pharmacokinetic study of Lycoris radiata extract in rats. J. Huazhong Univ. Sci. Technol. [Med. Sci.] 34, 861–868 (2014). https://doi.org/10.1007/s11596-014-1365-2
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11596-014-1365-2