Abstract
Background
In a phase II study, we showed that temozolomide (TMZ) was tolerable and active in heavily pre-treated patients with advanced colorectal cancer (CRC) and MGMT methylation. A schedule of dose-dense TMZ may have enhanced activity due to the higher cumulative dose and induction of MGMT depletion, even in resistant tumors.
Methods
Thirty-two patients with chemorefractory MGMT-methylated CRC were treated with TMZ at a daily dose of 75 mg/m2 for 21 consecutive days every 4 weeks, for up to six cycles or until the occurrence of progressive disease/unacceptable toxicity. The primary endpoint was treatment activity in terms of objective response rate (ORR). MGMT protein expression was tested by immunohistochemistry (IHC) on two pooled cohorts: patients from the previous study of standard-dose TMZ and those from the current investigation.
Results
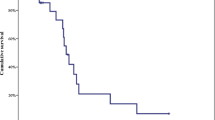
From November 2013 to December 2014, 32 patients were treated at Fondazione IRCCS Istituto Nazionale dei Tumori. We observed only three episodes of grade 3 asthenia and no significant myelotoxicity. The ORR was 16 % (all partial responses occurring in RAS-BRAF-mutated tumors). Median progression-free survival (PFS) and overall survival (OS) were 2.3 and 6.7 months, respectively. Patients with MGMT-low expression by IHC had a significantly higher ORR (p < 0.0001) and PFS (p = 0.001) compared to those with MGMT-high expression, while no difference was observed in OS.
Conclusions
Our data confirm the encouraging activity of TMZ in chemorefractory CRC patients selected for MGMT silencing, even in the RAS-BRAF-mutated population. The role of MGMT IHC as a biomarker for improving patient selection warrants further prospective confirmation.



Similar content being viewed by others
References
Shen L, Kondo Y, Rosner GL, Xiao L, Hernandez NS, Vilaythong J et al (2005) MGMT promoter methylation and field defect in sporadic colorectal cancer. J Natl Cancer Inst 97:1330–8
Esteller M, Risques RA, Toyota M, Capella G, Moreno V, Peinado MA et al (2001) Promoter hypermethylation of the DNA repair gene O(6)-methylguanine-DNA methyltransferase is associated with the presence of G: C to A: T transition mutations in p53 in human colorectal tumorigenesis. Cancer Res 61:4689–92
Esteller M, Herman JG (2004) Generating mutations but providing chemosensitivity: the role of O6-methylguanine DNA methyltransferase in human cancer. Oncogene 23:1–8
Hegi ME, Diserens AC, Gorlia T, Hamou MF, de Tribolet N, Weller M et al (2005) MGMT gene silencing and benefit from temozolomide in glioblastoma. N Engl J Med 352:997–1003
Amatu A, Sartore-Bianchi A, Moutinho C, Belotti A, Bencardino K, Chirico G et al (2013) Promoter CpG island hypermethylation of the DNA repair enzyme MGMT predicts clinical response to dacarbazine in a phase II study for metastatic colorectal cancer. Clin Cancer Res 19:2265–72
Hochhauser D, Glynne-Jones R, Potter V, Grávalos C, Doyle TJ, Pathiraja K et al (2013) A phase II study of temozolomide in patients with advanced aerodigestive tract and colorectal cancers and methylation of the O6-methylguanine-DNA methyltransferase promoter. Mol Cancer Ther 12:809–18
Inno A, Fanetti G, Di Bartolomeo M, Gori S, Maggi C, Cirillo M et al (2014) Role of MGMT as biomarker in colorectal cancer. World J Clin Cases 2:835–9
Pietrantonio F, Perrone F, de Braud F, Castano A, Maggi C, Bossi I et al (2014) Activity of temozolomide in patients with advanced chemorefractory colorectal cancer and MGMT promoter methylation. Ann Oncol 25:404–8
Tolcher AW, Gerson SL, Denis L, Geyer C, Hammond LA, Patnaik A et al (2003) Marked inactivation of O6-alkylguanine-DNA alkyl-transferase activity with protracted temozolomide schedules. Br J Cancer 88:1004–11
Chinot OL, Barrié M, Fuentes S, Eudes N, Lancelot S, Metellus P et al (2007) Correlation between O6-methylguanine-DNA methyltransferase and survival in inoperable newly diagnosed glioblastoma patients treated with neoadjuvant temozolomide. J Clin Oncol 25:1470–5
Di Bartolomeo M, Pietrantonio F, Perrone F, Dotti KF, Lampis A, Bertan C, Italian Trials in Medical Oncology Group et al (2014) Lack of KRAS, NRAS, BRAF and TP53 mutations improves outcome of elderly metastatic colorectal cancer patients treated with cetuximab, oxaliplatin and UFT. Target Oncol 9:155–62
Simon R (1989) Optimal two-stage design for phase II clinical trials. Control Clin Trials 10:1–10
National cancer Institute: Common Terminology Criteria for Adverse Events (CTCAE) v.3.0. Available from: http://ctep.cancer.gov/protocolDevelopment/electronic_applications/docs/ctcaev3.pdf
Hsu CY, Lin SC, Ho HL, Chang-Chien YC, Hsu SP, Yen YS et al (2013) Exclusion of histiocytes/endothelial cells and using endothelial cells as internal reference are crucial for interpretation of MGMT immunohistochemistry in glioblastoma. Am J Surg Pathol 37:264–71
Preusser M, Charles Janzer R, Felsberg J, Reifenberger G, Hamou MF et al (2008) Anti-O6-methylguanine-methyltransferase (MGMT) immunohistochemistry in glioblastoma multiforme: observer variability and lack of association with patient survival impede its use as clinical biomarker. Brain Pathol 18:520–32
Capper D, Mittelbronn M, Meyermann R, Schittenhelm J (2008) Pitfalls in the assessment of MGMT expression and in its correlation with survival in diffuse astrocytomas: proposal of a feasible immunohistochemical approach. Acta Neuropathol 115:249–59
Brell M, Tortosa A, Verger E, Gil JM, Viñolas N, Villá S et al (2005) Prognostic significance of O6-methylguanine-DNA methyltransferase determined by promoter hypermethylation and immunohistochemical expression in anaplastic gliomas. Clin Cancer Res 11:5167–74
Amatu A, Bencardino K, Barault L, Cassingena A, Bonazzina E, Ghezzi S et al (2015) Phase II study of temozolomide (TMZ) in metastatic colorectal cancer (mCRC) patients molecularly selected by MGMT promoter hypermethylation [abstract]. J Clin Oncol 33(Suppl 3):583
Esteller M, Toyota M, Sanchez-Cespedes M, Capella G, Peinado MA, Watkins DN et al (2000) Inactivation of the DNA repair gene O6-methylguanine-DNA methyltransferase by promoter hypermethylation is associated with G to A mutations in K-ras in colorectal tumorigenesis. Cancer Res 60:2368–71
Sato A, Sunayama J, Matsuda K, Seino S, Suzuki K, Watanabe E et al (2011) MEK-ERK signaling dictates DNA-repair gene MGMT expression and temozolomide resistance of stem-like glioblastoma cells via the MDM2-p53 axis. Stem Cells 29:1942–51
Tosoni A, Franceschi E, Ermani M, Bertorelle R, Bonaldi L, Blatt V et al (2008) Temozolomide three weeks on and one week off as first line therapy for patients with recurrent or progressive low grade gliomas. J Neuro-Oncol 89:179–85
Miyazaki M, Nishihara H, Terasaka S, Kobayashi H, Yamaguchi S, Ito T et al (2014) Immunohistochemical evaluation of O6 -methylguanine DNA methyltransferase (MGMT) expression in 117 cases of glioblastoma. Neuropathology 34:268–76
Brell M, Ibáñez J, Tortosa A (2011) O6-Methylguanine-DNA methyltransferase protein expression by immunohistochemistry in brain and non-brain systemic tumours: systematic review and meta-analysis of correlation with methylation-specific polymerase chain reaction. BMC Cancer 11:35
Barault L, Amatu A, Bleeker FE, Moutinho C, Falcomatà C, Fiano V, et al. Digital PCR quantification of MGMT methylation refines prediction of clinical benefit from alkylating agents in glioblastoma and metastatic colorectal cancer. Ann Oncol. 2015. doi:10.1093/annonc/mdv272
Fine RL, Gulati AP, Krantz BA, Moss RA, Schreibman S, Tsushima DA et al (2013) Capecitabine and temozolomide (CAPTEM) for metastatic, well-differentiated neuroendocrine cancers: The Pancreas Center at Columbia University experience. Cancer Chemother Pharmacol 71:663–70
Reynés G, Balañá C, Gallego O, Iglesias L, Pérez P, García JL (2014) A phase I study of irinotecan in combination with metronomic temozolomide in patients with recurrent glioblastoma. Anti-Cancer Drugs 25:717–22
Ku GY, Krol G, Ilson DH (2007) Successful treatment of leptomeningeal disease in colorectal cancer with a regimen of bevacizumab, temozolomide, and irinotecan. J Clin Oncol 25:e14–6
Acknowledgments
The authors thank all patients included in this study and their families.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
None.
Conflict of Interest
FP, FdB, MM, CM, RI, KFD, FP, ET AND MC declared no conflict of interest.
Rights and permissions
About this article
Cite this article
Pietrantonio, F., de Braud, F., Milione, M. et al. Dose-Dense Temozolomide in Patients with MGMT-Silenced Chemorefractory Colorectal Cancer. Targ Oncol 11, 337–343 (2016). https://doi.org/10.1007/s11523-015-0397-2
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11523-015-0397-2