Abstract
Purpose
Accumulating evidence indicates that micro (mi)RNAs play a critical role in carcinogenesis and cancer progression; however, their role in the tumorigenesis of gastric adenocarcinoma remains unclear so the present study investigated this in gastric cancer (GC) tissues and cell lines.
Methods
Human GC specimens (n = 57) and patient-paired non-cancerous specimens were obtained from patients at the First Affiliated Hospital, Henan University of Science and Technology. The AGS and GC9811 gastric cancer cell lines were also used. Expression levels of miR-216b and HDAC8 were examined by quantitative real-time PCR and the expression of HDAC8 was also examined by Western blotting and immunohistochemistry assay. The cell cycle progression was determined by FACS. MiR-216b inhibitor, mimics, and siRNA-HDAC8 transfections were performed to study the loss and gain of function.
Results
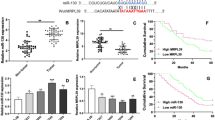
We reported a significantly decreased expression of miR-216b in GC clinical specimens compared with paired non-cancerous tissues. We also observed a significant down-regulation of miR-216b expression in GC cell lines AGS and GC9811 (p < 0.0001). The introduction of miR-216b suppressed GC cell proliferation and cell cycle progression by targeting HDAC8, an oncogene shown to promote malignant tumor development with a potential miR-216b binding site in its 3′ untranslated region. HDAC8 expression was shown to be significantly increased in AGS and GC9811 cell lines (p < 0.0001) and GC tissues compared with controls. Moreover, HDAC8 inhibition suppressed cell cycle progression compared with control groups (22 % ± 1.6 % vs 34 % ± 2.1), indicating that HDAC8 may function as an oncogene in the development of GC. Furthermore, HDAC8 expression was negatively correlated (p < 0.0001), while miR-216b expression was positively correlated with the clinical outcome of GC patients (p < 0.0001).
Discussion
Our data suggest that miR-216b functions as a tumor suppressor in human GC by, at least partially, targeting HDAC8.






Similar content being viewed by others
References
Crew KD, Neugut AI (2006) Epidemiology of gastric cancer. World J Gastroenterol 12(3):354–62
Li X, Zhang Y, Zhang H, Liu X, Gong T, Li M et al (2011) miRNA-223 promotes gastric cancer invasion and metastasis by targeting tumor suppressor EPB41L3. Mol Cancer Res 9(7):824–33
Tie J, Pan Y, Zhao L, Wu K, Liu J, Sun S et al (2010) MiR-218 inhibits invasion and metastasis of gastric cancer by targeting the Robo1 receptor. PLoS Genet 6(3):e1000879
Gravalos C, Jimeno A (2008) HER2 in gastric cancer: a new prognostic factor and a novel therapeutic target. Ann Oncol 19(9):1523–9
Ji Q, Hao X, Meng Y, Zhang M, DeSano J, Fan D et al (2008) Restoration of tumor suppressor miR-34 inhibits human p53-mutant gastric cancer tumorspheres. BMC Cancer 8(1):266
Tamura G (2006) Alterations of tumor suppressor and tumor-related genes in the development and progression of gastric cancer. World J Gastroenterol 12(2):192–8
Lu J, Getz G, Miska EA, Alvarez-Saavedra E, Lamb J, Peck D et al (2005) MicroRNA expression profiles classify human cancers. Nature 435(7043):834–8
Griffiths-Jones S, Grocock RJ, Van Dongen S, Bateman A, Enright AJ (2006) miRBase: microRNA sequences, targets and gene nomenclature. Nucleic Acids Res 34(suppl 1):D140–4
Kumar MS, Lu J, Mercer KL, Golub TR, Jacks T (2007) Impaired microRNA processing enhances cellular transformation and tumorigenesis. Nat Genet 39(5):673–7
Kota J, Chivukula RR, O’Donnell KA, Wentzel EA, Montgomery CL, Hwang H-W et al (2009) Therapeutic microRNA delivery suppresses tumorigenesis in a murine liver cancer model. Cell 137(6):1005–17
He X, Chang Y, Meng F, Wang M, Xie Q, Tang F et al (2012) MicroRNA-375 targets AEG-1 in hepatocellular carcinoma and suppresses liver cancer cell growth in vitro and in vivo. Oncogene 31(28):3357–69
Cheng C-Y, Hwang C-I, Corney DC, Flesken-Nikitin A, Jiang L, Öner GM et al (2014) miR-34 cooperates with p53 in suppression of prostate cancer by joint regulation of stem cell compartment. Cell Reports 6(6):1000–7
Romano G, Acunzo M, Garofalo M, Di Leva G, Cascione L, Zanca C et al (2012) MiR-494 is regulated by ERK1/2 and modulates TRAIL-induced apoptosis in non–small-cell lung cancer through BIM down-regulation. Proc Natl Acad Sci 109(41):16570–5
Yang Q, Jie Z, Ye S, Li Z, Han Z, Wu J et al (2014) Genetic variations in miR-27a gene decrease mature miR-27a level and reduce gastric cancer susceptibility. Oncogene 33(2):193–202
Rokavec M, Öner MG, Li H, Jackstadt R, Jiang L, Lodygin D et al (2014) IL-6R/STAT3/miR-34a feedback loop promotes EMT-mediated colorectal cancer invasion and metastasis. J Clin Invest 124(4):1853
Hossain A, Kuo MT, Saunders GF (2006) Mir-17-5p regulates breast cancer cell proliferation by inhibiting translation of AIB1 mRNA. Mol Cell Biol 26(21):8191–201
Deng M, Liu J, Gu Y, Zheng G, He Z (2013) miR-216b suppresses cell proliferation and invasion by targeting PKCα in nasopharyngeal carcinoma cells. Zhonghua Zhong Liu Za Zhi 35(9):645–50
Deng M, Tang H, Zhou Y, Zhou M, Xiong W, Zheng Y et al (2011) miR-216b suppresses tumor growth and invasion by targeting KRAS in nasopharyngeal carcinoma. J Cell Sci 124(17):2997–3005
Moser MA, Hagelkruys A, Seiser C (2014) Transcription and beyond: the role of mammalian class I lysine deacetylases. Chromosoma 123(1–2):67–78
Witt O, Deubzer HE, Milde T, Oehme I (2009) HDAC family: what are the cancer relevant targets? Cancer Lett 277(1):8–21
Oehme I, Deubzer HE, Wegener D, Pickert D, Linke J-P, Hero B et al (2009) Histone deacetylase 8 in neuroblastoma tumorigenesis. Clin Cancer Res 15(1):91–9
Wang Y, Zheng X, Zhang Z, Zhou J, Zhao G, Yang J et al (2012) MicroRNA-149 inhibits proliferation and cell cycle progression through the targeting of HDAC8 in human gastric cancer. PLoS One 7(10), e41693
Twentyman P, Luscombe M (1987) A study of some variables in a tetrazolium dye (MTT) based assay for cell growth and chemosensitivity. Br J Cancer 56(3):279
Van de Loosdrecht A, Beelen R, Ossenkoppele G, Broekhoven M, Langenhuijsen M (1994) A tetrazolium-based colorimetric MTT assay to quantitate human monocyte mediated cytotoxicity against leukemic cells from cell lines and patients with acute myeloid leukemia. J Immunol Methods 174(1):311–20
Xu X, Zhang Y, Zhang W, Li T, Gao H, Wang Y (2013) MicroRNA-133a functions as a tumor suppressor in gastric cancer. J Biol Regul Homeost Agents 28(4):615–24
Liang J, Liu X, Xue H, Qiu B, Wei B, Sun K (2015) MicroRNA‐103a inhibits gastric cancer cell proliferation, migration and invasion by targeting c‐Myb. Cell Prolif 48(1):78–85
Shen X, Si Y, Yang Z, Wang Q, Yuan J, Zhang X (2015) MicroRNA-542-3p suppresses cell growth of gastric cancer cells via targeting oncogene astrocyte-elevated gene-1. Med Oncol 32(1):1–8
Gu W, Gao T, Shen J, Sun Y, Zheng X, Wang J et al (2014) MicroRNA-183 inhibits apoptosis and promotes proliferation and invasion of gastric cancer cells by targeting PDCD4. International Journal of Clinical and Experimental Medicine 7(9):2519
Zheng X, Dong J, Gong T, Zhang Z, Wang Y, Li Y et al (2015) MicroRNA library-based functional screening identified miR-137 as a suppresser of gastric cancer cell proliferation. J Cancer Res Clin Oncol 141(5):785–95
Li LP, Wu WJ, Sun DY, Xie ZY, Ma YC, Zhao YG (2014) miR-449a and CDK6 in gastric carcinoma. Oncology Letters 8(4):1533–8
Li Z, Yu X, Wang Y, Shen J, Wu WK, Liang J et al (2015) By downregulating TIAM1 expression, microRNA-329 suppresses gastric cancer invasion and growth. Oncotarget 6(19):17559–69
Han X, Chen Y, Yao N, Liu H, Wang Z (2015) MicroRNA let-7b suppresses human gastric cancer malignancy by targeting ING1. Cancer Gene Ther 22(3):122–9
Bannister AJ, Kouzarides T (2011) Regulation of chromatin by histone modifications. Cell Res 21(3):381–95
Haberland M, Montgomery RL, Olson EN (2009) The many roles of histone deacetylases in development and physiology: implications for disease and therapy. Nat Rev Genet 10(1):32–42
Oehme I, Deubzer HE, Lodrini M, Milde T, Witt O (2009) Targeting of HDAC8 and investigational inhibitors in neuroblastoma. Expert Opin Investig Drugs 18(11):1605–17
Park SY, Jun J, Jeong KJ, Heo HJ, Sohn JS, Lee HY et al (2011) Histone deacetylases 1, 6 and 8 are critical for invasion in breast cancer. Oncol Rep 25(6):1677–81
Abbas A, Gupta S (2008) The role of histone deacetylases in prostate cancer. Epigenetics 3(6):300–9
Lin CC, Papadopoulos K (2007) Novel targeted therapies for advanced esophageal cancer. Dis Esophagus 20(5):365–71
Nakagawa M, Oda Y, Eguchi T, Aishima S-I, Yao T, Hosoi F et al (2007) Expression profile of class I histone deacetylases in human cancer tissues. Oncol Rep 18(4):769–74
Hsieh T-H, Hsu C-Y, Tsai C-F, Long C-Y, Wu C-H, Wu D-C, et al (2015) HDAC inhibitors target HDAC5, upregulate MicroRNA-125a-5p, and induce apoptosis in breast cancer cells. Mol Ther 23(4):656–66
Chen D-Q, Huang J-Y, Feng B, Pan B-Z, De W, Wang R et al (2014) Histone deacetylase 1/Sp1/MicroRNA-200b signaling accounts for maintenance of cancer stem-like cells in human lung adenocarcinoma. PLoS One 9(10):e109578
Seol HS, Akiyama Y, Shimada S, Lee HJ, Im Kim T, Chun SM et al (2014) Epigenetic silencing of microRNA-373 to epithelial-mesenchymal transition in non-small cell lung cancer through IRAK2 and LAMP1 axes. Cancer Lett 353(2):232–41
Chen D-Q, Pan B-Z, Huang J-Y, Zhang K, Cui S-Y, De W et al (2014) HDAC 1/4-mediated silencing of microRNA-200b promotes chemoresistance in human lung adenocarcinoma cells. Oncotarget 5(10):3333–49
Author Contributions
Y.W. performed data analyses and wrote the manuscript, initiated the project, designed the experiments, and interpreted the data. P.X. and ZG.S. performed cell culture and cell transfection. SY.S. and XJ.Z. performed qRT-PCR analysis. XS.F. and SG.G performed the cell cycle and proliferation experiments in vitro.
Conflict of Interest
Dr. Ying Wang, Dr. Po Xu, Dr. Jun Yao, Dr. Ruina Yang, Dr. Zhenguo Shi, Dr. Xiaojuan Zhu, Dr. Xiaoshan Feng and Dr. Shegan Gao have no conflicts of interest to disclose.
Funding Statement
This study was supported by the National Natural Science Foundation of China (No. 81301763 and No. 81572849) and Henan provincial key scientific and technological projects (No. 142102310473).
Ethics Statement
Written informed consent was obtained from patients before obtaining tissue samples. The procedures used in this study were approved by the Institutional Review Board of the First Affiliated Hospital, Henan University of Science and Technology and conformed to the Helsinki Declaration and to local legislation.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Ying Wang and Po Xu contributed equally to this work.
This article has been retracted by agreement of the corresponding author, the First Affiliated Hospital of Henan University of Science and Technology, Luoyang, China, and the Editor-in-Chief. Errors were identified in some of the published figures, and the authors could not provide a satisfactory explanation owing to loss of the primary data.
An erratum to this article is available at http://dx.doi.org/10.1007/s11523-016-0470-5.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplementary Table 1
Relationship between clinical parameters and miR-216b (mean ± SE) expression in primary gastric adenocarcinoma. One-way ANOVA analysis, n = 3 (DOCX 15 kb)
Supplementary Table 2
Relationship between clinical parameters and HDAC8 (mean ± SE) expression in primary gastric adenocarcinoma. One-way ANOVA analysis, n = 3 (DOCX 20 kb)
Supplementary Table 3
Antibodies used in Western blotting assay (DOCX 17 kb)
Supplementary Fig. 1
A. Schematic of Luc-HDAC8 3′UTR and Luc-HDAC8 3′MutUTR constructs. Luc-HDAC8 3′UTR and Luc-HDAC8 3′Mut UTR were cloned into a pmirGLO plasmid downstream of the firefly luciferase coding region between PmeI and XbaI sites. B. Wild-type and mutant HDAC8-3′UTR containing the putative binding site of miR-216b were cloned into psiCHECK-2 vector. HDAC8-3′UTR was amplified from genomic DNA of GC9811. C. Lane 3,4: Recombinant plasmids of HDAC8-1; lane 7,8: Results of enzyme digestion of recombinant plasmids of HDAC8-1. Results showed that HDAC8-1 have been successfully inserted into the vectors.(M: DL2000 DNA Marker; HDAC8-1 bands: 1283 bp; Vectors bands: 6.1 Kb). D. M: 1 kb DNA Ladder Marker. Lane 1: amplification of mutHDAC8F1/R1 (negative control without Taq enzyme). E. Sequencing results of WT-HDAC8 and MT –HDAC8 (WT: wild type; MT: mutated type). (GIF 69 kb)
Supplementary Fig. 2
Expression of HDAC8 in GC cells and the normal gastric epithelial cells were examined by Western blotting and shown as mean ± SE (normalized to tubulin). (GIF 7 kb)
About this article
Cite this article
Wang, Y., Xu, P., Yao, J. et al. RETRACTED ARTICLE: MicroRNA-216b is Down-Regulated in Human Gastric Adenocarcinoma and Inhibits Proliferation and Cell Cycle Progression by Targeting Oncogene HDAC8. Targ Oncol 11, 197–207 (2016). https://doi.org/10.1007/s11523-015-0390-9
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11523-015-0390-9