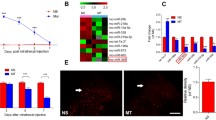
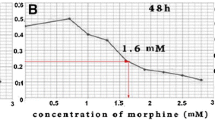
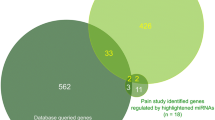
Abstract
MicroRNAs (miRNA), a class of ~22-nucleotide RNA molecules, are important gene regulators that bind to the target sites of mRNAs to inhibit the gene expressions either through translational inhibition or mRNA destabilization. There are growing evidences that miRNAs have played several regulatory roles in opioid pharmacology. Like other research fields such as cancer biology, the area where numerous miRNAs are found to be involved in gene regulation, we assume that in opioid studies including research fields of drug additions and opioid receptor regulation, there may be more miRNAs waiting to be discovered. This review will summarize our current knowledge of miRNA functions on opioids biology and briefly describe future research directions of miRNAs related to opioids.


Similar content being viewed by others
Abbreviations
- Ago2:
-
Argonaute 2
- BDNF:
-
Brain-derived neurotrophic factor
- CNS:
-
Central nervous system
- CREB:
-
cAMP-response element binding protein
- CYP2D6:
-
cytochrome P450 2D6
- Drd2:
-
Dopamine 2 receptor
- Drd3:
-
dopamine D3 receptor
- ERK:
-
Extracellular signal-regulated kinase
- FGF-2:
-
Fibroblast growth factor-2
- FosB:
-
FBJ murine osteosarcoma viral oncogene homolog B
- GPCR:
-
G protein-coupled receptor
- HEK:
-
Human embryonic kidney
- HIV-1:
-
Human immunodeficiency virus type 1
- MeCP2:
-
Methyl CpG binding protein 2
- MOR:
-
Mu opioid receptor
- NeuroD:
-
Neurogenic differentiation 1
- PKA:
-
Protein kinase A
- PKC:
-
Protein kinase C
References
Albulescu R, Neagu M, Albulescu L, Tanase C (2011) Tissular and soluble miRNAs for diagnostic and therapy improvement in digestive tract cancers. Expert Rev Mol Diagn 11(1):101–120. doi:10.1586/erm.10.106
Ali S, Drendel AL, Kircher J, Beno S (2010) Pain management of musculoskeletal injuries in children: current state and future directions. Pediatr Emerg Care 26(7):518–524. doi:10.1097/PEC.0b013e3181e5c02b
Amir RE, Van den Veyver IB, Wan M, Tran CQ, Francke U, Zoghbi HY (1999) Rett syndrome is caused by mutations in X-linked MECP2, encoding methyl-CpG-binding protein 2. Nat Genet 23(2):185–188. doi:10.1038/13810
Amir RE, Sutton VR, Van den Veyver IB (2005) Newborn screening and prenatal diagnosis for Rett syndrome: implications for therapy. J Child Neurol 20(9):779–783
Artavanis-Tsakonas S, Rand MD, Lake RJ (1999) Notch signaling: cell fate control and signal integration in development. Science 284(5415):770–776
Azaryan AV, Clock BJ, Cox BM (1996a) Mu opioid receptor mRNA in nucleus accumbens is elevated following dopamine receptor activation. Neurochem Res 21(11):1411–1415
Azaryan AV, Coughlin LJ, Buzas B, Clock BJ, Cox BM (1996b) Effect of chronic cocaine treatment on mu- and delta-opioid receptor mRNA levels in dopaminergically innervated brain regions. J Neurochem 66(2):443–448
Bartel DP (2004) MicroRNAs: genomics, biogenesis, mechanism, and function. Cell 116(2):281–297. doi:10.1016/S0092-8674(04)00045-5
Bell JE, Arango JC, Anthony IC (2006) Neurobiology of multiple insults: HIV-1-associated brain disorders in those who use illicit drugs. J Neuroimmune Pharmacol 1(2):182–191. doi:10.1007/s11481-006-9018-2
Brodsky M, Elliott K, Hynansky A, Jenab S, Inturrisi CE (1995) Quantitation of mu-opioid receptor (MOR-1) mRNA in selected regions of the rat CNS. Neuroreport 6(5):725–729
Chahrour M, Jung SY, Shaw C, Zhou X, Wong ST, Qin J, Zoghbi HY (2008) MeCP2, a key contributor to neurological disease, activates and represses transcription. Science 320(5880):1224–1229. doi:10.1126/science.1153252
Chen CZ, Li L, Lodish HF, Bartel DP (2004) MicroRNAs modulate hematopoietic lineage differentiation. Science 303(5654):83–86. doi:10.1126/science.1091903
Chhabra R, Adlakha YK, Hariharan M, Scaria V, Saini N (2009) Upregulation of miR-23a-27a-24-2 cluster induces caspase-dependent and -independent apoptosis in human embryonic kidney cells. PLoS One 4(6):e5848. doi:10.1371/journal.pone.0005848
Chhabra R, Dubey R, Saini N (2010) Cooperative and individualistic functions of the microRNAs in the miR-23a 27a 24–2 cluster and its implication in human diseases. Mol Cancer 9:232. doi:10.1186/1476-4598-9-232
Dave RS, Khalili K (2010) Morphine treatment of human monocyte-derived macrophages induces differential miRNA and protein expression: impact on inflammation and oxidative stress in the central nervous system. J Cell Biochem 110(4):834–845. doi:10.1002/jcb.22592
Deng JV, Rodriguiz RM, Hutchinson AN, Kim IH, Wetsel WC, West AE (2010) MeCP2 in the nucleus accumbens contributes to neural and behavioral responses to psychostimulants. Nat Neurosci 13(9):1128–1136. doi:10.1038/nn.2614
Donahoe RM, Vlahov D (1998) Opiates as potential cofactors in progression of HIV-1 infections to AIDS. J Neuroimmunol 83(1–2):77–87
Dreyer JL (2010) New insights into the roles of microRNAs in drug addiction and neuroplasticity. Genome Med 2(12):92. doi:10.1186/gm213
Drugs.com (2011) Codeine Information from Drugs.com. Web site from Drugs.com. http://www.drugs.com/monograph/codeine.html. Accessed October 18 2011
Duttaroy A, Yoburn BC (1995) The effect of intrinsic efficacy on opioid tolerance. Anesthesiology 82(5):1226–1236
Duursma AM, Kedde M, Schrier M, le Sage C, Agami R (2008) miR-148 targets human DNMT3b protein coding region. RNA 14(5):872–877. doi:10.1261/rna.972008
Edbauer D, Neilson JR, Foster KA, Wang CF, Seeburg DP, Batterton MN, Tada T, Dolan BM, Sharp PA, Sheng M (2010) Regulation of synaptic structure and function by FMRP-associated microRNAs miR-125b and miR-132. Neuron 65(3):373–384. doi:10.1016/j.neuron.2010.01.005
Feng J, Nestler EJ (2010) MeCP2 and drug addiction. Nat Neurosci 13(9):1039–1041. doi:10.1038/nn0910-1039
Fitting S, Xu R, Bull C, Buch SK, El-Hage N, Nath A, Knapp PE, Hauser KF (2010) Interactive comorbidity between opioid drug abuse and HIV-1 Tat: chronic exposure augments spine loss and sublethal dendritic pathology in striatal neurons. Am J Pathol 177(3):1397–1410. doi:10.2353/ajpath.2010.090945
Flores CM, Mogil JS (2001) The pharmacogenetics of analgesia: toward a genetically-based approach to pain management. Pharmacogenomics 2(3):177–194. doi:10.1517/14622416.2.3.177
Gao FB (2010) Context-dependent functions of specific microRNAs in neuronal development. Neural Dev 5:25. doi:10.1186/1749-8104-5-25
Gasche Y, Daali Y, Fathi M, Chiappe A, Cottini S, Dayer P, Desmeules J (2004) Codeine intoxication associated with ultrarapid CYP2D6 metabolism. N Engl J Med 351(27):2827–2831. doi:10.1056/NEJMoa041888
Gonzalez-Scarano F, Martin-Garcia J (2005) The neuropathogenesis of AIDS. Nat Rev Immunol 5(1):69–81. doi:10.1038/nri1527
Gottlieb GS, Heath L, Nickle DC, Wong KG, Leach SE, Jacobs B, Gezahegne S, van’t Wout AB, Jacobson LP, Margolick JB, Mullins JI (2008) HIV-1 variation before seroconversion in men who have sex with men: analysis of acute/early HIV infection in the multicenter AIDS cohort study. J Infect Dis 197(7):1011–1015. doi:10.1086/529206
Guo CJ, Li Y, Tian S, Wang X, Douglas SD, Ho WZ (2002) Morphine enhances HIV infection of human blood mononuclear phagocytes through modulation of beta-chemokines and CCR5 receptor. J Investig Med 50(6):435–442
Hassan MQ, Gordon JA, Beloti MM, Croce CM, van Wijnen AJ, Stein JL, Stein GS, Lian JB (2010) A network connecting Runx2, SATB2, and the miR-23a 27a 24–2 cluster regulates the osteoblast differentiation program. Proc Natl Acad Sci U S A 107(46):19879–19884. doi:10.1073/pnas.1007698107
Hauser KF, El-Hage N, Stiene-Martin A, Maragos WF, Nath A, Persidsky Y, Volsky DJ, Knapp PE (2007) HIV-1 neuropathogenesis: glial mechanisms revealed through substance abuse. J Neurochem 100(3):567–586. doi:10.1111/j.1471-4159.2006.04227.x
He Y, Yang C, Kirkmire CM, Wang ZJ (2010) Regulation of opioid tolerance by let-7 family microRNA targeting the mu opioid receptor. J Neurosci 30(30):10251–10258. doi:10.1523/JNEUROSCI.2419-10.2010
Ho WZ, Guo CJ, Yuan CS, Douglas SD, Moss J (2003) Methylnaltrexone antagonizes opioid-mediated enhancement of HIV infection of human blood mononuclear phagocytes. J Pharmacol Exp Ther 307(3):1158–1162. doi:10.1124/jpet.103.056697
Hollander JA, Im HI, Amelio AL, Kocerha J, Bali P, Lu Q, Willoughby D, Wahlestedt C, Conkright MD, Kenny PJ (2010) Striatal microRNA controls cocaine intake through CREB signalling. Nature 466(7303):197–202. doi:10.1038/nature09202
Homan JW, Steele AD, Martinand-Mari C, Rogers TJ, Henderson EE, Charubala R, Pfleiderer W, Reichenbach NL, Suhadolnik RJ (2002) Inhibition of morphine-potentiated HIV-1 replication in peripheral blood mononuclear cells with the nuclease-resistant 2-5A agonist analog, 2-5A(N6B). J Acquir Immune Defic Syndr 30(1):9–20
Ingelman-Sundberg M (2005) Genetic polymorphisms of cytochrome P450 2D6 (CYP2D6): clinical consequences, evolutionary aspects and functional diversity. Pharmacogenomics J 5(1):6–13. doi:10.1038/sj.tpj.6500285
Jeal W, Benfield P (1997) Transdermal fentanyl. A review of its pharmacological properties and therapeutic efficacy in pain control. Drugs 53(1):109–138
Jeffrey GP, MacQuillan G, Chua F, Galhenage S, Bull J, Young E, Hulse G, O’Neil G (2007) Hepatitis C virus eradication in intravenous drug users maintained with subcutaneous naltrexone implants. Hepatology 45(1):111–117. doi:10.1002/hep. 21470
Kalow W (2001) Pharmacogenetics, pharmacogenomics, and pharmacobiology. Clin Pharmacol Ther 70(1):1–4. doi:10.1067/mcp.2001.116714
Kawashima H, Numakawa T, Kumamaru E, Adachi N, Mizuno H, Ninomiya M, Kunugi H, Hashido K (2010) Glucocorticoid attenuates brain-derived neurotrophic factor-dependent upregulation of glutamate receptors via the suppression of microRNA-132 expression. Neuroscience 165(4):1301–1311. doi:10.1016/j.neuroscience.2009.11.057
Keele BF, Giorgi EE, Salazar-Gonzalez JF, Decker JM, Pham KT, Salazar MG, Sun C, Grayson T, Wang S, Li H, Wei X, Jiang C, Kirchherr JL, Gao F, Anderson JA, Ping LH, Swanstrom R, Tomaras GD, Blattner WA, Goepfert PA, Kilby JM, Saag MS, Delwart EL, Busch MP, Cohen MS, Montefiori DC, Haynes BF, Gaschen B, Athreya GS, Lee HY, Wood N, Seoighe C, Perelson AS, Bhattacharya T, Korber BT, Hahn BH, Shaw GM (2008) Identification and characterization of transmitted and early founder virus envelopes in primary HIV-1 infection. Proc Natl Acad Sci U S A 105(21):7552–7557. doi:10.1073/pnas.0802203105
Kim E, Clark AL, Kiss A, Hahn JW, Wesselschmidt R, Coscia CJ, Belcheva MM (2006) Mu- and kappa-opioids induce the differentiation of embryonic stem cells to neural progenitors. J Biol Chem 281(44):33749–33760. doi:10.1074/jbc.M603862200
Kim J, Inoue K, Ishii J, Vanti WB, Voronov SV, Murchison E, Hannon G, Abeliovich A (2007) A MicroRNA feedback circuit in midbrain dopamine neurons. Science 317(5842):1220–1224. doi:10.1126/science.1140481
Kim VN, Han J, Siomi MC (2009) Biogenesis of small RNAs in animals. Nat Rev Mol Cell Biol 10(2):126–139. doi:10.1038/nrm2632
Kimura H, Kawasaki H, Taira K (2004) Mouse microRNA-23b regulates expression of Hes1 gene in P19 cells. Nucleic Acids Symp Ser (Oxf) 48:213–214. doi:10.1093/nass/48.1.213
Klein ME, Lioy DT, Ma L, Impey S, Mandel G, Goodman RH (2007) Homeostatic regulation of MeCP2 expression by a CREB-induced microRNA. Nat Neurosci 10(12):1513–1514. doi:10.1038/nn2010
Kozomara A, Griffiths-Jones S (2011) miRBase: integrating microRNA annotation and deep-sequencing data. Nucleic Acids Res 39(Database issue):D152–D157. doi:10.1093/nar/gkq1027
Kusuda R, Cadetti F, Ravanelli MI, Sousa TA, Zanon S, De Lucca FL, Lucas G (2011) Differential expression of microRNAs in mouse pain models. Mol Pain 7:17. doi:10.1186/1744-8069-7-17
Lagos-Quintana M, Rauhut R, Yalcin A, Meyer J, Lendeckel W, Tuschl T (2002) Identification of tissue-specific microRNAs from mouse. Curr Biol 12(9):735–739. doi:10.1016/S0960-9822(02)00809-6
Lai EC (2002) Micro RNAs are complementary to 3′ UTR sequence motifs that mediate negative post-transcriptional regulation. Nat Genet 30(4):363–364. doi:10.1038/ng865
Lai EC, Burks C, Posakony JW (1998) The K box, a conserved 3′ UTR sequence motif, negatively regulates accumulation of enhancer of split complex transcripts. Development 125(20):4077–4088
Lai EC, Tam B, Rubin GM (2005) Pervasive regulation of Drosophila Notch target genes by GY-box-, Brd-box-, and K-box-class microRNAs. Genes Dev 19(9):1067–1080. doi:10.1101/gad.1291905
Law PY, Wong YH, Loh HH (2000) Molecular mechanisms and regulation of opioid receptor signaling. Annu Rev Pharmacol Toxicol 40:389–430
Lee YS, Dutta A (2006) MicroRNAs: small but potent oncogenes or tumor suppressors. Curr Opin Investig Drugs 7(6):560–564
Lee RC, Feinbaum RL, Ambros V (1993) The C. elegans heterochronic gene lin-4 encodes small RNAs with antisense complementarity to lin-14. Cell 75(5):843–854. doi:10.1016/0092-8674(93)90529-Y
Lee HJ, Palkovits M, Young WS 3rd (2006) miR-7b, a microRNA up-regulated in the hypothalamus after chronic hyperosmolar stimulation, inhibits Fos translation. Proc Natl Acad Sci U S A 103(42):15669–15674. doi:10.1073/pnas.0605781103
Lehmann U, Hasemeier B, Christgen M, Muller M, Romermann D, Langer F, Kreipe H (2008) Epigenetic inactivation of microRNA gene hsa-mir-9-1 in human breast cancer. J Pathol 214(1):17–24. doi:10.1002/path.2251
Lewis JD, Meehan RR, Henzel WJ, Maurer-Fogy I, Jeppesen P, Klein F, Bird A (1992) Purification, sequence, and cellular localization of a novel chromosomal protein that binds to methylated DNA. Cell 69(6):905–914. doi:10.1016/0092-8674(92)90610-O
Lewis BP, Burge CB, Bartel DP (2005) Conserved seed pairing, often flanked by adenosines, indicates that thousands of human genes are microRNA targets. Cell 120(1):15–20. doi:10.1016/j.cell.2004.12.035
Li Y, Wang X, Tian S, Guo CJ, Douglas SD, Ho WZ (2002) Methadone enhances human immunodeficiency virus infection of human immune cells. J Infect Dis 185(1):118–122. doi:10.1086/338011
Lin Q, Gao Z, Alarcon RM, Ye J, Yun Z (2009) A role of miR-27 in the regulation of adipogenesis. FEBS J 276(8):2348–2358
Liu H, Kohane IS (2009) Tissue and process specific microRNA-mRNA co-expression in mammalian development and malignancy. PLoS One 4(5):e5436. doi:10.1371/journal.pone.0005436
Liu L, Walker EA, Kissane S, Khan I, Murray PI, Rauz S, Wallace GR (2011) Gene Expression and miR Profiles of Human Corneal Fibroblasts in Response to Dexamethasone. Invest Ophthalmol Vis Sci 52(10):7282–7288. doi:10.1007/s11033-010-0343-4
Macey TA, Bobeck EN, Hegarty DM, Aicher SA, Ingram SL, Morgan MM (2009) Extracellular signal-regulated kinase 1/2 activation counteracts morphine tolerance in the periaqueductal gray of the rat. J Pharmacol Exp Ther 331(2):412–418. doi:10.1124/jpet.109.152157
Madadi P, Ciszkowski C, Gaedigk A, Leeder JS, Teitelbaum R, Chitayat D, Koren G (2011) Genetic transmission of cytochrome P450 2D6 (CYP2D6) ultrarapid metabolism: implications for breastfeeding women taking codeine. Curr Drug Saf 6(1):36–39
Makeyev EV, Zhang J, Carrasco MA, Maniatis T (2007) The MicroRNA miR-124 promotes neuronal differentiation by triggering brain-specific alternative pre-mRNA splicing. Mol Cell 27(3):435–448. doi:10.1016/j.molcel.2007.07.015
Mansour A, Fox CA, Akil H, Watson SJ (1995) Opioid-receptor mRNA expression in the rat CNS: anatomical and functional implications. Trends Neurosci 18(1):22–29
Mayes S, Ferrone M (2006) Fentanyl HCl patient-controlled iontophoretic transdermal system for the management of acute postoperative pain. Ann Pharmacother 40(12):2178–2186. doi:10.1345/aph.1H135
McGregor RA, Choi MS (2011) microRNAs in the regulation of adipogenesis and obesity. Curr Mol Med 11(4):304–316
Mogensen TH, Melchjorsen J, Larsen CS, Paludan SR (2010) Innate immune recognition and activation during HIV infection. Retrovirology 7:54. doi:10.1186/1742-4690-7-54
Nair MP, Schwartz SA, Polasani R, Hou J, Sweet A, Chadha KC (1997) Immunoregulatory effects of morphine on human lymphocytes. Clin Diagn Lab Immunol 4(2):127–132
Neves G, Cooke SF, Bliss TV (2008) Synaptic plasticity, memory and the hippocampus: a neural network approach to causality. Nat Rev Neurosci 9(1):65–75. doi:10.1038/nrn2303
Nudelman AS, DiRocco DP, Lambert TJ, Garelick MG, Le J, Nathanson NM, Storm DR (2010) Neuronal activity rapidly induces transcription of the CREB-regulated microRNA-132, in vivo. Hippocampus 20(4):492–498. doi:10.1002/hipo.20646
O’Connell RM, Taganov KD, Boldin MP, Cheng G, Baltimore D (2007) MicroRNA-155 is induced during the macrophage inflammatory response. Proc Natl Acad Sci U S A 104(5):1604–1609. doi:10.1073/pnas.0610731104
Ohno M, Natsume A, Kondo Y, Iwamizu H, Motomura K, Toda H, Ito M, Kato T, Wakabayashi T (2009) The modulation of microRNAs by type I IFN through the activation of signal transducers and activators of transcription 3 in human glioma. Mol Cancer Res 7(12):2022–2030. doi:10.1158/1541-7786.MCR-09-0319
Pan YX, Xu J, Mahurter L, Bolan E, Xu M, Pasternak GW (2001) Generation of the mu opioid receptor (MOR-1) protein by three new splice variants of the Oprm gene. Proc Natl Acad Sci U S A 98(24):14084–14089
Pedersen IM, Cheng G, Wieland S, Volinia S, Croce CM, Chisari FV, David M (2007) Interferon modulation of cellular microRNAs as an antiviral mechanism. Nature 449(7164):919–922. doi:10.1038/nature06205
Pergolizzi J, Boger RH, Budd K, Dahan A, Erdine S, Hans G, Kress HG, Langford R, Likar R, Raffa RB, Sacerdote P (2008) Opioids and the management of chronic severe pain in the elderly: consensus statement of an International Expert Panel with focus on the six clinically most often used World Health Organization Step III opioids (buprenorphine, fentanyl, hydromorphone, methadone, morphine, oxycodone). Pain Pract 8(4):287–313. doi:10.1111/j.1533-2500.2008.00204.x
Perry AK, Chen G, Zheng D, Tang H, Cheng G (2005) The host type I interferon response to viral and bacterial infections. Cell Res 15(6):407–422. doi:10.1038/sj.cr.7290309
Rodrigues AC, Li X, Radecki L, Pan YZ, Winter JC, Huang M, Yu AM (2011) MicroRNA expression is differentially altered by xenobiotic drugs in different human cell lines. Biopharm Drug Dispos 32(6):355–367. doi:10.1002/bdd.764
Rusinov V, Baev V, Minkov IN, Tabler M (2005) MicroInspector: a web tool for detection of miRNA binding sites in an RNA sequence. Nucleic Acids Res 33(Web Server issue):W696–W700. doi:10.1093/nar/gki364
Sanchez-Simon FM, Zhang XX, Loh HH, Law PY, Rodriguez RE (2010) Morphine regulates dopaminergic neuron differentiation via miR-133b. Mol Pharmacol 78(5):935–942. doi:10.1124/mol.110.066837
Sato F, Tsuchiya S, Meltzer SJ, Shimizu K (2011) MicroRNAs and epigenetics. FEBS J 278(10):1598–1609. doi:10.1111/j.1742-4658.2011.08089.x
Schaefer A, Im HI, Veno MT, Fowler CD, Min A, Intrator A, Kjems J, Kenny PJ, O’Carroll D, Greengard P (2010) Argonaute 2 in dopamine 2 receptor-expressing neurons regulates cocaine addiction. J Exp Med 207(9):1843–1851. doi:10.1084/jem.20100451
Schratt GM, Tuebing F, Nigh EA, Kane CG, Sabatini ME, Kiebler M, Greenberg ME (2006) A brain-specific microRNA regulates dendritic spine development. Nature 439(7074):283–289. doi:10.1038/nature04367
Schwartz C, Catez P, Rohr O, Lecestre D, Aunis D, Schaeffer E (2000) Functional interactions between C/EBP, Sp1, and COUP-TF regulate human immunodeficiency virus type 1 gene transcription in human brain cells. J Virol 74(1):65–73
Somel M, Guo S, Fu N, Yan Z, Hu HY, Xu Y, Yuan Y, Ning Z, Hu Y, Menzel C, Hu H, Lachmann M, Zeng R, Chen W, Khaitovich P (2010) MicroRNA, mRNA, and protein expression link development and aging in human and macaque brain. Genome Res 20(9):1207–1218. doi:10.1101/gr.106849.110
Tuttle DL, Harrison JK, Anders C, Sleasman JW, Goodenow MM (1998) Expression of CCR5 increases during monocyte differentiation and directly mediates macrophage susceptibility to infection by human immunodeficiency virus type 1. J Virol 72(6):4962–4969
Unterwald EM, Horne-King J, Kreek MJ (1992) Chronic cocaine alters brain mu opioid receptors. Brain Res 584(1–2):314–318
Unterwald EM, Rubenfeld JM, Kreek MJ (1994) Repeated cocaine administration upregulates kappa and mu, but not delta, opioid receptors. Neuroreport 5(13):1613–1616
Unterwald EM, Rubenfeld JM, Imai Y, Wang JB, Uhl GR, Kreek MJ (1995) Chronic opioid antagonist administration upregulates mu opioid receptor binding without altering mu opioid receptor mRNA levels. Brain Res Mol Brain Res 33(2):351–355. doi:10.1016/0169-328X(95)00143-G
Valencia-Sanchez MA, Liu J, Hannon GJ, Parker R (2006) Control of translation and mRNA degradation by miRNAs and siRNAs. Genes Dev 20(5):515–524. doi:10.1101/gad.1399806
Vallejo R, de Leon-Casasola O, Benyamin R (2004) Opioid therapy and immunosuppression: a review. Am J Ther 11(5):354–365
van Jaarsveld MT, Helleman J, Berns EM, Wiemer EA (2010) MicroRNAs in ovarian cancer biology and therapy resistance. Int J Biochem Cell Biol 42(8):1282–1290. doi:10.1016/j.biocel.2010.01.014
van Rooij E (2011) The art of microRNA research. Circ Res 108(2):219–234. doi:10.1161/circresaha.110.227496
Visvanathan J, Lee S, Lee B, Lee JW, Lee SK (2007) The microRNA miR-124 antagonizes the anti-neural REST/SCP1 pathway during embryonic CNS development. Genes Dev 21(7):744–749. doi:10.1101/gad.1519107
Wang X (2008) miRDB: a microRNA target prediction and functional annotation database with a wiki interface. RNA 14(6):1012–1017. doi:10.1261/rna.965408
Wang X, El Naqa IM (2008) Prediction of both conserved and nonconserved microRNA targets in animals. Bioinformatics 24(3):325–332. doi:10.1093/bioinformatics/btm595
Wang J, Gabuzda D (2006) Reconstitution of human immunodeficiency virus-induced neurodegeneration using isolated populations of human neurons, astrocytes, and microglia and neuroprotection mediated by insulin-like growth factors. J Neurovirol 12(6):472–491. doi:10.1080/13550280601039659
Wang X, Ye L, Hou W, Zhou Y, Wang YJ, Metzger DS, Ho WZ (2009) Cellular microRNA expression correlates with susceptibility of monocytes/macrophages to HIV-1 infection. Blood 113(3):671–674. doi:10.1182/blood-2008-09-175000
Wang X, Li C, Ju S, Wang Y, Wang H, Zhong R (2011a) Myeloma cell adhesion to bone marrow stromal cells confers drug resistance by microRNA-21 up-regulation. Leuk Lymphoma 52(10):1991–1998. doi:10.3109/10428194.2011.591004
Wang X, Ye L, Zhou Y, Liu MQ, Zhou DJ, Ho WZ (2011b) Inhibition of anti-HIV microRNA expression: a mechanism for opioid-mediated enhancement of HIV infection of monocytes. Am J Pathol 178(1):41–47. doi:10.1016/j.ajpath.2010.11.042
Wayman GA, Davare M, Ando H, Fortin D, Varlamova O, Cheng HY, Marks D, Obrietan K, Soderling TR, Goodman RH, Impey S (2008) An activity-regulated microRNA controls dendritic plasticity by down-regulating p250GAP. Proc Natl Acad Sci U S A 105(26):9093–9098. doi:10.1073/pnas.0803072105
Wibrand K, Panja D, Tiron A, Ofte ML, Skaftnesmo KO, Lee CS, Pena JT, Tuschl T, Bramham CR (2010) Differential regulation of mature and precursor microRNA expression by NMDA and metabotropic glutamate receptor activation during LTP in the adult dentate gyrus in vivo. Eur J Neurosci 31(4):636–645. doi:10.1111/j.1460-9568.2010.07112.x
Wu Q, Law PY, Wei LN, Loh HH (2008) Post-transcriptional regulation of mouse mu opioid receptor (MOR1) via its 3′ untranslated region: a role for microRNA23b. FASEB J 22(12):4085–4095. doi:10.1096/fj.08-108175
Wu Q, Zhang L, Law PY, Wei LN, Loh HH (2009) Long-term morphine treatment decreases the association of mu-opioid receptor (MOR1) mRNA with polysomes through miRNA23b. Mol Pharmacol 75(4):744–750. doi:10.1124/mol.108.053462
Yang B, Lin H, Xiao J, Lu Y, Luo X, Li B, Zhang Y, Xu C, Bai Y, Wang H, Chen G, Wang Z (2007) The muscle-specific microRNA miR-1 regulates cardiac arrhythmogenic potential by targeting GJA1 and KCNJ2. Nat Med 13(4):486–491. doi:10.1038/nm1569
Yu JY, Chung KH, Deo M, Thompson RC, Turner DL (2008) MicroRNA miR-124 regulates neurite outgrowth during neuronal differentiation. Exp Cell Res 314(14):2618–2633. doi:10.1016/j.yexcr.2008.06.002
Zheng H, Chu J, Zeng Y, Loh HH, Law PY (2010a) Yin Yang 1 phosphorylation contributes to the differential effects of mu-opioid receptor agonists on microRNA-190 expression. J Biol Chem 285(29):21994–22002. doi:10.1074/jbc.M110.112607
Zheng H, Zeng Y, Chu J, Kam AY, Loh HH, Law PY (2010b) Modulations of NeuroD activity contribute to the differential effects of morphine and fentanyl on dendritic spine stability. J Neurosci 30(24):8102–8110. doi:10.1523/JNEUROSCI.6069-09.2010
Zheng H, Zeng Y, Zhang X, Chu J, Loh HH, Law PY (2010c) mu-Opioid receptor agonists differentially regulate the expression of miR-190 and NeuroD. Mol Pharmacol 77(1):102–109. doi:10.1124/mol.109.060848
Acknowledgements
This work was supported by the National Institutes of Health National Institutes of Drug Abuse [Grants DA000564, DA001583, DA011806, K05-DA070554, DA011190, DA013926]; and by the A & F Stark Fund of the Minnesota Medical Foundation. The authors declare no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hwang, C.K., Wagley, Y., Law, PY. et al. MicroRNAs in Opioid Pharmacology. J Neuroimmune Pharmacol 7, 808–819 (2012). https://doi.org/10.1007/s11481-011-9323-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11481-011-9323-2