Abstract
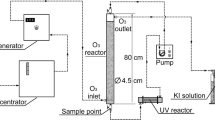
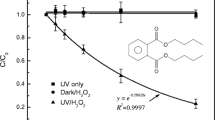
Hydroxylated polyhalodiphenyl ethers (HO-PXDEs) have attracted considerable scientific interest as examples of emerging aquatic pollutants. However, a comprehensive assessment of disposal methods for this particular pollutant was seldom investigated. This study examined the UV light degradation of HO-PXDEs, using 2′-HO-2, 3′, 4, 5′-tetrabromodiphenyl ether (2′-HO-BDE-68) as a case study. The results showed that UV light was superior to visible light and electron beam irradiation for producing a high degradation rate of 2′-HO-BDE-68. At low concentrations of HO-BDE, the degradation rate was not obviously improved with decreasing initial concentration. The degradation efficiency was also found to be better in alkaline solutions. In a UV/H2O2 system, the hydroxyl radical provided by H2O2 was shown to enhance the degradation efficiency. The main photolysis products of 2′-HO-BDE-68 were identified, and the possible photodegradation pathways were proposed. 1, 3, 8-Tribromodibenzo-p-dioxin was one of the photoproducts, which indicates that secondary pollution must also be considered with the UV photolysis process.







Similar content being viewed by others
Reference
Arnoldsson K, Andersson PL, Haglund P (2012) Photochemical formation of polybrominated dibenzo-p-dioxins from environmentally abundant hydroxylated polybrominated diphenyl ethers. Environmental science & technology 46:7567–7574. doi:10.1021/es301256x
Ayodhya D, Venkatesham M, Santoshi Kumari A, Reddy GB, Ramakrishna D, Veerabhadram G (2015) Photocatalytic degradation of dye pollutants under solar, visible and UV lights using green synthesised CuS nanoparticles. J Exp Nanosci 11:418–432. doi:10.1080/17458080.2015.1070312
Bastos PM, Eriksson J, Bergman A (2009) Photochemical decomposition of dissolved hydroxylated polybrominated diphenyl ethers under various aqueous conditions. Chemosphere 77:791–797. doi:10.1016/j.chemosphere.2009.08.013
Birnbaum L (2003) Health effects of polybrominated dibenzo-p-dioxins (PBDDs) and dibenzofurans (PBDFs). Environ Int 29:855–860. doi:10.1016/s0160-4120(03)00106-5
Choi JW et al (2003) Polybrominated dibenzo-p-dioxins, dibenzofurans, and diphenyl ethers in Japanese human adipose tissue. Am Chem Soc 37:817–821
Feo ML et al (2014) Advanced UV/H(2)O(2) oxidation of deca-bromo diphenyl ether in sediments. Sci Total Environ 479-480:17–20. doi:10.1016/j.scitotenv.2014.01.091
Frangos P et al (2016) Improvement of the degradation of pesticide deethylatrazine by combining UV photolysis with electrochemical generation of hydrogen peroxide. Chem Eng J 291:215–224. doi:10.1016/j.cej.2016.01.089
Hamers T et al (2006) In vitro profiling of the endocrine-disrupting potency of brominated flame retardants. Toxicol Sci 92:157–173
He X et al (2012) Efficient removal of microcystin-LR by UV-C/H(2)O(2) in synthetic and natural water samples. Water Res 46:1501–1510. doi:10.1016/j.watres.2011.11.009
Hendriks HS, Fernandes ECA, Bergman Å, van den Berg M, Westerink RH (2010) PCB-47, PBDE-47, and 6-OH-PBDE-47 differentially modulate human GABAA and α4β2 nicotinic acetylcholine receptors. Toxicol Sci 118:635–642
Illes E, Takacs E, Dombi A, Gajda-Schrantz K, Racz G, Gonter K, Wojnarovits L (2013) Hydroxyl radical induced degradation of ibuprofen. Sci Total Environ 447:286–292. doi:10.1016/j.scitotenv.2013.01.007
Kliegman S, Eustis SN, Arnold WA, McNeill K (2013) Experimental and theoretical insights into the involvement of radicals in triclosan phototransformation. Environmental science & technology 47:6756–6763
Lai Y, Lu M, Lin S, Cai Z (2012) Glucuronidation of hydroxylated polybrominated diphenyl ethers and their modulation of estrogen UDP-glucuronosyltransferases. Chemosphere 86:727–734. doi:10.1016/j.chemosphere.2011.10.047
Liu N, Sijak S, Zheng M, Tang L, Xu G, Wu MH (2015) Aquatic photolysis of florfenicol and thiamphenicol under direct UV irradiation, UV/H2O2 and UV/Fe(II) processes. Chem Eng J 260:826–834. doi:10.1016/j.cej.2014.09.055
Marsh G, Stenutz R, Bergman Å (2003) Synthesis of hydroxylated and methoxylated polybrominated diphenyl ethers—natural products and potential polybrominated diphenyl ether metabolites. Eur J Org Chem 2003:2566–2576. doi:10.1002/ejoc.200300081
Nayak S, O'Donnell SE, Sales CM, Tikekar RV (2016) Fructose accelerates UV-C induced photochemical degradation of pentachlorophenol in low and high salinity water. J Agric Food Chem 64:4214–4219. doi:10.1021/acs.jafc.6b01449
Ren XM, Guo LH, Gao Y, Zhang BT, Wan B (2013) Hydroxylated polybrominated diphenyl ethers exhibit different activities on thyroid hormone receptors depending on their degree of bromination. Toxicol Appl Pharmacol 268:256–263. doi:10.1016/j.taap.2013.01.026
Stapleton HM, Kelly SM, Pei R, Letcher RJ, Gunsch C (2009) Metabolism of polybrominated diphenyl ethers (PBDEs) by human hepatocytes in vitro. Environ Health Perspect 117:197–202. doi:10.1289/ehp.11807
Steen PO, Grandbois M, McNeill K, Arnold WA (2009) Photochemical formation of halogenated dioxins from hydroxylated polybrominated diphenyl ethers (OH-PBDEs) and chlorinated derivatives (OH-PBCDEs). Environmental science & technology 43:4405–4411
Sun J, Liu J, Liu Q, Qu G, Ruan T, Jiang G (2012) Sample preparation method for the speciation of polybrominated diphenyl ethers and their methoxylated and hydroxylated analogues in diverse environmental matrices. Talanta 88:669–676. doi:10.1016/j.talanta.2011.11.059
Wang S, Zhang S, Huang H, Niu Z, Han W (2014) Characterization of polybrominated diphenyl ethers (PBDEs) and hydroxylated and methoxylated PBDEs in soils and plants from an e-waste area, China. Environ Pollut 184:405–413. doi:10.1016/j.envpol.2013.09.021
Wu MH et al (2016) Occurrence and profiles of polybrominated diphenyl ethers (PBDEs) in riverine sediments of Shanghai: a combinative study with human serum from the locals. Environ Geochem Health. doi:10.1007/s10653-016-9843-z
Xie Q, Chen J, Zhao H, Qiao X, Cai X, Li X (2013) Different photolysis kinetics and photooxidation reactivities of neutral and anionic hydroxylated polybrominated diphenyl ethers. Chemosphere 90:188–194. doi:10.1016/j.chemosphere.2012.06.033
Xie Q, Chen J, Zhao H, Wang X, Xie HB (2015) Distinct photoproducts of hydroxylated polybromodiphenyl ethers from different photodegradation pathways: a case study of 2′-HO-BDE-68. Environmental science Processes & impacts 17:351–357. doi:10.1039/c4em00395k
Xu G, Yao JZ, Tang L, Yang XY, Zheng M, Wang H, Wu MH (2015) Electron beam induced degradation of atrazine in aqueous solution. Chem Eng J 275:374–380. doi:10.1016/j.cej.2015.04.063
Zhang YN, Xie Q, Chen J, Li Y, Fu Z (2015) Insights into the photochemical transformation pathways of triclosan and 2'-HO-BDE-28. J Hazard Mater 300:354–358. doi:10.1016/j.jhazmat.2015.07.036
Zhang YN et al (2016) Effects of dissolved organic matter on phototransformation rates and dioxin products of triclosan and 2'-HO-BDE-28 in estuarine water. Environmental science Processes & impacts 18:1177–1184. doi:10.1039/c6em00122j
Acknowledgements
This work was financially supported by the National Natural Science Foundation of China (Nos. 41430644, 41473090, 11675098, and 41373098).
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible editor: Vítor Pais Vilar
Highlights
• Direct UV irradiation could lead to complete 2′-HO-BDE-68 removal in 30 min.
• Superior conditions were discussed in the UV photolysis.
• Photoproducts were identified and degradation pathways were proposed.
Rights and permissions
About this article
Cite this article
Xu, B., Wu, M., Pan, C. et al. Aquatic photolysis of hydroxylated polybromodiphenyl ethers under direct UV irradiation: a case study of 2′-HO-BDE-68. Environ Sci Pollut Res 24, 14409–14416 (2017). https://doi.org/10.1007/s11356-017-8726-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-017-8726-x