Abstract
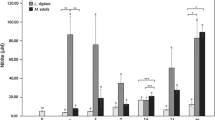
Mussels are widespread in coastal environments and experience various physical, chemical, and bacteriological conditions. Owing to the increase of coastal urbanization, mussels are now commonly exposed not only to indigenous bacteria, but also to enteric bacteria originating from pulsed and chronic sewage discharges into coastal environments. Due to its broad resilience to environmental variations, the blue mussel Mytilus edulis is commonly used as an indicator of environmental quality in bio-monitoring programs. However, since mussel immune system capabilities may be affected by the presence of exogenous fecal bacteria in coastal seawater subjected to sewage discharges, we aimed to determine the effect of in vivo bacterial challenges on mussels' immune competency by using two exogenous enteric bacterial strains, Escherichia coli and Enterococcus faecalis, and an indigenous bacterial strain Vibrio splendidus (as control). Bacterial strains were tested individually, by injection into the posterior adductor muscle at three different cell densities (102, 103, and 104 cells). Unlike classic in vitro experiments using higher bacterial concentrations, neither the enteric bacteria nor the indigenous strain induced significant increase or decrease of either cell-mediated (phagocytosis, reactive oxygen species, and NO x production) or humoral components (prophenoloxidase-like, acid phosphatase, and l-leucine-aminopeptidase production) of the immune system. This study demonstrates that, at low concentrations, E. coli and E. faecalis do not represent an additional threat that could impair M. edulis immune competency and, as a consequence, its potential of survival in coastal areas subjected to sewage discharges.
Similar content being viewed by others
References
Akaishi FM, St-Jean SD, Bishay F, Clarke J, IdS R, de Oliveira Ribeiro CA (2007) Immunological responses, histopathological finding and disease resistance of blue mussel (Mytilus edulis) exposed to treated and untreated municipal wastewater. Aquat Toxicol 82:1–14. doi:10.1016/j.aquatox.2007.01.008
Andersch MA, Szczypinski AJ (1947) Use of p-nitrophenylphosphate as the substrate in determination of serum acid phosphatase. Am J Clin Pathol 17:571–574
Anonymous (2012) Aquaculture Canada: facts and figures. http://www.dfo-mpo.gc.ca/aquaculture/ref/stats/aqua-ff-fc-2009-eng.htm. Accessed 21 March 2012
Araya MT, Markham F, Mateo DR, McKenna P, Johnson GR, Berthe FCJ, Siah A (2010) Identification and expression of immune-related genes in hemocytes of soft-shell clams, Mya arenaria, challenged with Vibrio splendidus. Fish Shellfish Immunol 29:557–564. doi:10.1016/j.fsi.2010.05.017
Asokan R, Arumugam M, Mullainadhan P (1997) Activation of prophenoloxidase in the plasma and haemocytes of the marine mussel Perna viridis Linnaeus. Dev Comp Immunol 21:1–12. doi:10.1016/s0145-305x(97)00004-9
Auffret M, Rousseau S, Boutet I, Tanguy A, Baron J, Moraga D, Duchemin M (2006) A multiparametric approach for monitoring immunotoxic responses in mussels from contaminated sites in Western Mediterranean. Ecotoxicol Environ Saf 63:393–405. doi:10.1016/j.ecoenv.2005.10.016
Bao Y, Wang Q, Liu H, Lin Z (2011) A small HSP gene of bloody clam (Tegillarca granosa) involved in the immune response against Vibrio parahaemolyticus and lipopolysaccharide. Fish Shellfish Immunol 30:729–733. doi:10.1016/j.fsi.2010.12.002
Beutler B (2004) Innate immunity: an overview. Mol Immunol 40:845–859. doi:10.1016/j.molimm.2003.10.005
Birkbeck TH, McHenery JG (1982) Degradation of bacteria by Mytilus edulis. Mar Biol 72:7–15. doi:10.1007/bf00393942
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Bussell JA, Gidman EA, Causton DR, Gwynn-Jones D, Malham SK, Jones MLM, Reynolds B, Seed R (2008) Changes in the immune response and metabolic fingerprint of the mussel, Mytilus edulis (Linnaeus) in response to lowered salinity and physical stress. J Exp Mar Biol Ecol 358:78–85. doi:10.1016/j.jembe.2008.01.018
Canesi L, Gallo G, Gavioli M, Pruzzo C (2002) Bacteria–hemocyte interactions and phagocytosis in marine bivalves. Microsc Res Tech 57:469–476. doi:10.1002/jemt.10100
Cavallo RA, Stabili L (2002) Presences of vibrios in seawater and Mytilus galloprovincialis (Lam.) from the Mar Piccolo of Taranto (Ionian Sea). Water Res 36:3719–3726. doi:10.1016/S0043-1354(02)00107-0
Cheng TC (1992) Selective induction of release of hydrolases from Crassostrea virginica hemocytes by certain bacteria. J Invertebr Pathol 59:197–200. doi:10.1016/0022-2011(92)90033-z
Chu F-LE (1988) Humoral defense factors in marine bivalves. In: Fisher WS (ed) Disease processes in marine bivalve molluscs. Fisheries Society special publication, Bethesda, pp 178–188
Ciacci C, Citterio B, Betti M, Canonico B, Roch P, Canesi L (2009) Functional differential immune responses of Mytilus galloprovincialis to bacterial challenge. Comp Biochem Physiol B Biochem Mol Biol 153:365–371. doi:10.1016/j.cbpb.2009.04.007
Coles JA, Pipe RK (1994) Phenoloxidase activity in the haemolymph and haemocytes of the marine mussel Mytilus edulis. Fish Shellfish Immunol 4:337–352. doi:10.1006/fsim.1994.1030
Costa MM, Prado-Alvarez M, Gestal C, Li H, Roch P, Novoa B, Figueras A (2009) Functional and molecular immune response of Mediterranean mussel (Mytilus galloprovincialis) haemocytes against pathogen-associated molecular patterns and bacteria. Fish Shellfish Immunol 26:515–523. doi:10.1016/j.fsi.2009.02.001
De Decker S, Saulnier D (2011) Vibriosis induced by experimental cohabitation in Crassostrea gigas: evidence of early infection and down-expression of immune-related genes. Fish Shellfish Immunol 30:691–699. doi:10.1016/j.fsi.2010.12.017
Delaporte M, Soudant P, Lambert C, Jegaden M, Moal J, Pouvreau S, Dégremont L, Boudry P, Samain J-F (2007) Characterisation of physiological and immunological differences between Pacific oysters (Crassostrea gigas) genetically selected for high or low survival to summer mortalities and fed different rations under controlled conditions. J Exp Mar Biol Ecol 353:45–57. doi:10.1016/j.jembe.2007.09.003
Donaghy L, Volety AK (2011) Functional and metabolic characterization of hemocytes of the green mussel, Perna viridis: in vitro impacts of temperature. Fish Shellfish Immunol 31:808–814. doi:10.1016/j.fsi.2011.07.018
Duchemin MB, Auffret M, Wessel N, Fortier M, Morin Y, Pellerin J, Fournier M (2008) Multiple experimental approaches of immunotoxic effects of mercury chloride in the blue mussel, Mytilus edulis, through in vivo, in tubo and in vitro exposures. Environ Pollut 153:416–423. doi:10.1016/j.envpol.2007.08.015
Feng SY (1988) Cellular defense mechanisms of oysters and mussels. In: Fisher WS (ed) Disease processes in marine bivalve molluscs. Fisheries Society Special Publication, Bethesda, pp 153–168
Frischer ME, Nierzwicki-Bauer SA, Parsons RH, Vathanodorn K, Waitkus KR (2000) Interactions between zebra mussels (Dreissena polymorpha) and microbial communities. Can J Fish Aquat Sci 57:591–599. doi:10.1139/f00-001
Gagnaire B, Gay M, Huvet A, Daniel J-Y, Saulnier D, Renault T (2007) Combination of a pesticide exposure and a bacterial challenge: in vivo effects on immune response of Pacific oyster, Crassostrea gigas (Thunberg). Aquat Toxicol 84:92–102. doi:10.1016/j.aquatox.2007.06.002
Girard M, Roussy M, Coulombe F, Cauvier C, Moisan N (2008) Optimisation des opérations de dépuration en vrac des moules bleues de la baie de Gaspé par la réduction des débits de l’eau de mer à basse température, vol 169. MAPAQ, Quebec
Goedken M, Morsey B, Sunila I, Dungan C, De Guise S (2005) The effects of temperature and salinity on apoptosis of Crassostrea virginica hemocytes and Perkinsus marinus. J Shellfish Res 24:177–183. doi:10.2983/0730-8000(2005)24[177:teotas]2.0.co;2
Jacquet S, Lennon J-F, Marie D, Vaulot D (1998) Picoplankton population dynamics in coastal waters of the Northwestern Mediterranean Sea. Limnol Oceanogr 43:1916–1931
Kuchel RP, Raftos DA, Nair S (2010) Immunosuppressive effects of environmental stressors on immunological function in Pinctada imbricata. Fish Shellfish Immunol 29:930–936. doi:10.1016/j.fsi.2010.07.033
Labreuche Y, Lambert C, Soudant P, Boulo V, Huvet A, Nicolas J-L (2006) Cellular and molecular hemocyte responses of the Pacific oyster, Crassostrea gigas, following bacterial infection with Vibrio aestuarianus strain 01/32. Microbes Infect 8:2715–2724. doi:10.1016/j.micinf.2006.07.020
Lacoste A, Malham SK, Gélébart F, Cueff A, Poulet SA (2002) Stress-induced immune changes in the oyster Crassostrea gigas. Dev Comp Immunol 26:1–9. doi:10.1016/s0145-305x(01)00067-2
Lambert C, Soudant P, Choquet G, Paillard C (2003) Measurement of Crassostrea gigas hemocyte oxidative metabolism by flow cytometry and the inhibiting capacity of pathogenic vibrios. Fish Shellfish Immunol 15:225–240. doi:10.1016/s1050-4648(02)00160-2
Lane E, Birkbeck TH (2000) Species specificity of some bacterial pathogens of bivalve molluscs is correlated with their interaction with bivalve haemocytes. J Fish Dis 23:275–279. doi:10.1046/j.1365-2761.2000.00224.x
Lebaron P, Servais P, Baudoux A-C, Bourrain M, Courties C, Parthuisot N (2002) Variations of bacterial-specific activity with cell size and nucleic acid content assessed by flow cytometry. Aquat Microb Ecol 28:131–140. doi:10.3354/ame028131
Lemaire N, Pellerin J, Fournier M, Girault L, Tamigneaux E, Cartier S, Pelletier E (2006) Seasonal variations of physiological parameters in the blue mussel Mytilus spp. from farm sites of eastern Quebec. Aquaculture 261:729–751. doi:10.1016/j.aquaculture.2006.08.017
MdM L, Signoretto C, Canepari P (2005) Gram-positive bacteria in the marine environment. In: Belkin S, Colwell RR (eds) Oceans and health: pathogens in the marine environment. Springer, USA, pp 307–330
Lucena F, Lasobras J, McIntosh D, Forcadell M, Jofre J (1994) Effect of distance from the polluting focus on relative concentrations of Bacteroides fragilis phages and coliphages in mussels. Appl Environ Microbiol 60:2272–2277
Malagoli D, Casarini L, Orraviani E (2007) Monitoring of the immune efficiency of Mytilus galloprovincialis in Adriatic sea mussel farms in 2006: regular changes in cytotoxicity during the year. Invertebr Surviv J 4:10–12
Marino A, Lombardo L, Fiorentino C, Orlandella B, Monticelli L, Nostro A, Alonzo V (2005) Uptake of Escherichia coli, Vibrio cholerae non-O1 and Enterococcus durans by, and depuration of mussels (Mytilus galloprovincialis). Int J Food Microbiol 99:281–286. doi:10.1016/j.ijfoodmicro.2004.09.003
Mateo DR, Siah A, Araya MT, Berthe FCJ, Johnson GR, Greenwood SJ (2009) Differential in vivo response of soft-shell clam hemocytes against two strains of Vibrio splendidus: changes in cell structure, numbers and adherence. J Invertebr Pathol 102:50–56. doi:10.1016/j.jip.2009.06.008
Munari M, Matozzo V, Marin MG (2011) Combined effects of temperature and salinity on functional responses of haemocytes and survival in air of the clam Ruditapes philippinarum. Fish Shellfish Immunol 30:1024–1030. doi:10.1016/j.fsi.2011.01.025
Parisi M-G, Li H, Jouvet LBP, Dyrynda EA, Parrinello N, Cammarata M, Roch P (2008) Differential involvement of mussel hemocyte sub-populations in the clearance of bacteria. Fish Shellfish Immunol 25:834–840. doi:10.1016/j.fsi.2008.09.005
Pipe RK (1992) Generation of reactive oxygen metabolites by the haemocytes of the mussel Mytilus edulis. Dev Comp Immunol 16:111–122. doi:10.1016/0145-305x(92)90012-2
Prato E, Danieli A, Maffia M, Biandolino F (2010) Lipid and fatty acid compositions of Mytilus galloprovincialis cultured in the Mar Grande of Taranto (Southern Italy): feeding strategies and trophic relationships. Zool Stud 49:211–219
Pruzzo C, Gallo G, Canesi L (2005) Persistence of vibrios in marine bivalves: the role of interactions with haemolymph components. Environ Microbiol 7:761–772. doi:10.1111/j.1462-2920.2005.00792.x
Roslev P, Iversen L, Sonderbo HL, Iversen N, Bastholm S (2009) Uptake and persistence of human associated Enterococcus in the mussel Mytilus edulis: relevance for faecal pollution source tracking. J Appl Microbiol 107:944–953. doi:10.1111/j.1365-2672.2009.04272.x
Samain JF, Dégremont L, Soletchnik P, Haure J, Bédier E, Ropert M, Moal J, Huvet A, Bacca H, Van Wormhoudt A, Delaporte M, Costil K, Pouvreau S, Lambert C, Boulo V, Soudant P, Nicolas JL, Le Roux F, Renault T, Gagnaire B, Geret F, Boutet I, Burgeot T, Boudry P (2007) Genetically based resistance to summer mortality in the Pacific oyster (Crassostrea gigas) and its relationship with physiological, immunological characteristics and infection processes. Aquaculture 268:227–243. doi:10.1016/j.aquaculture.2007.04.044
Saulnier D, De Decker S, Haffner P, Cobret L, Robert M, Garcia C (2010) A large-scale epidemiological study to identify bacteria pathogenic to Pacific oyster Crassostrea gigas and correlation between virulence and metalloprotease-like activity. Microb Ecol 59:787–798. doi:10.1007/s00248-009-9620-y
Travers M-A, Le Goïc N, Huchette S, Koken M, Paillard C (2008) Summer immune depression associated with increased susceptibility of the European abalone, Haliotis tuberculata to Vibrio harveyi infection. Fish Shellfish Immunol 25:800–808. doi:10.1016/j.fsi.2008.08.003
Weidenmaier C, Peschel A (2008) Teichoic acids and related cell-wall glycopolymers in Gram-positive physiology and host interactions. Nat Rev Microbiol 6:276–287
Winters AD, Marsh TL, Faisal M (2011) Heterogeneity of bacterial communities within the zebra mussel (Dreissena polymorpha) in the Laurentian Great Lakes Basin. J Great Lakes Res 37:318–324. doi:10.1016/j.jglr.2011.01.010
Wootton EC, Dyrynda EA, Pipe RK, Ratcliffe NA (2003) Comparisons of PAH-induced immunomodulation in three bivalve molluscs. Aquat Toxicol 65:13–25. doi:10.1016/s0166-445x(03)00098-5
Wright AC, Hill RT, Johnson JA, Roghman MC, Colwell RR, Morris JG (1996) Distribution of Vibrio vulnificus in the Chesapeake Bay. Appl Environ Microbiol 62:717–724
Yu JH, Song JH, Choi MC, Park SW (2009) Effects of water temperature change on immune function in surf clams, Mactra veneriformis (Bivalvia: Mactridae). J Invertebr Pathol 102:30–35. doi:10.1016/j.jip.2009.06.002
Acknowledgments
The authors are grateful to Ahmed Siah and Dante Mateo from the Department of Pathology and Microbiology of the Atlantic Veterinary College of PEI University for providing us the bacterial strain V. splendidus 7SHRW. Thanks are addressed to Marie-Gil Fortin and Bertrand Genard for their technical support. This study was supported by grants from the Natural Sciences and Engineering Research Council of Canada, Strategic Projects to M. Fournier, and the Société de Développement de l’Industrie Maricole du Québec. Isabelle Boily received ISMER and Réseau Aquaculture Québec supports during this work.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible editor: Robert Duran
Rights and permissions
About this article
Cite this article
Gauthier-Clerc, S., Boily, I., Fournier, M. et al. In vivo exposure of Mytilus edulis to living enteric bacteria: a threat for immune competency?. Environ Sci Pollut Res 20, 612–620 (2013). https://doi.org/10.1007/s11356-012-1200-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-012-1200-x