Abstract
Purpose
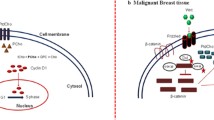
The calcium-sensing receptor (CaSR) is overexpressed in many pathological states including breast cancer. Since choline kinase may be activated in breast cancer cells by CaSR resulting in increased phosphocholine production, we sought to correlate the total choline peak in breast lesions as measured by in vivo proton magnetic resonance spectroscopy (1H-MRS) with the CaSR expression levels in surgical specimens.
Procedures
Thirty-six patients with breast lesions were MR scanned at 3T scanner. Tumour morphology and DCE-MR kinetics were evaluated. 1H-MRS was applied for Cho detection and compared with the CaSR immunohistochemistry analysis (score 0–5) on surgical breast specimens.
Results
Thirty-four lesions demonstrated a DCE malignant kinetics curve (types 2 and 3), while two lesions showed a benign (type 1). Twenty of the 23 breast cancer lesions (87 %) with a consistent Cho peak expressed a CaSR score of 3–5, and ten of the 11 breast lesions negative for Cho (91 %) had a CaSR score of 1–2. The two benign lesions showed a non-uniform/weak intense expression of the CaSR (score 3) with a consistent Cho peak.
Conclusions
The presence or absence of choline peak evaluated by 1H-MRS, well correlated with the expression of CaSR in patients with breast lesions (p < 0.01), supports the hypothesis that CaSR may play an important role in the production of choline in breast cancer.






Similar content being viewed by others
References
Ward DT, Riccardi D (2012) New concepts in calcium-sensing receptor pharmacology and signalling. Br J Pharmacol 165:35–48
Diez-Fraile A, Lammens T, Benoit Y et al (2013) The calcium-sensing receptor as a regulator of cellular fate in normal and pathological conditions. Curr Mol Med 13:282–295
Ward DT (2004) Calcium receptor-mediated intracellular signalling. Cell Calcium 35:217–228
Mihai R, Stevens J, McKinney C et al (2006) Expression of the calcium receptor in human breast cancer-a potential new marker predicting the risk of bone metastases. Eur J Surg Oncol 32:511–515
Warner E, Plewes DB, Shumak RS et al (2001) Comparison of breast magnetic resonance imaging, mammography, and ultrasound for surveillance of women at high risk for hereditary breast cancer. J Clin Oncol 19:3524–3531
Warner E, Messersmith H, Causer P et al (2008) Systematic review: using magnetic resonance imaging to screen women at high risk for breast cancer. Intern Med 148:671–679
Houssami N, Hayes DF (2009) Review of preoperative magnetic resonance imaging (MRI) in breast cancer: should MRI be performed on all women with newly diagnosed, early stage breast cancer? Cancer J Clin 59:290–302
Su MY, Yu HJ, Carpenter PM et al (2005) Pharmacokinetic parameters analyzed from MR contrast enhancement kinetics of multiple malignant and benign breast lesions detected in the same patients. Technol Cancer Res Treat 4:255–263
Aboagye EO, Bhujwall ZM (1999) Malignant transformation alters membrane choline phospholipid metabolism of human mammary epithelial cells. Cancer Res 59:80–84
Lean C, Doran S, Somorjai RL et al (2004) Determination of grade and receptor status from the primary breast lesion by magnetic resonance spectroscopy. Technol Cancer Res Treat 3:551–556
Huang C, Hydo LM, Liu S et al (2009) Activation of choline kinase by extracellular Ca2+ is Ca(2+)-sensing receptor, Galpha12 and Rho-dependent in breast cancer cells. Cell Signal 21:1894–1900
Brown EM, MacLeod RJ (2001) Extracellular calcium sensing and extracellular calcium signaling. Physiol Rev 81:239–297
O’Flynn EA, DeSouza NM (2011) Functional magnetic resonance: Biomarkers of response in breast cancer. Breast Cancer Res 13:405
Baek HM, Lee YJ (2014) Feasibility of MR spectroscopy for characterizing malignant breast lesions using a clinical 3-T scanner. Breast Cancer Jan 4. [Epub ahead of print]
Cheng I, Klingensmith ME, Chattopadhyay N et al (1998) Identification and localization of the extracellular calcium-sensing receptor in human breast. J Clin Endocrinol Metab 83:703–707
Huang W, Fisher PR, Dulaimy K et al (2004) Detection of breast malignancy: Diagnostic MR protocol for improved specificity. Radiology 232:585–591
Grinde MT, Skrbo N, Moestue SA et al (2014) Interplay of choline metabolites and genes in patient-derived breast cancer xenografts. Breast Cancer Res 16:R5
Brennan SC, Thiem U, Roth S et al (2013) Calcium sensing receptor signalling in physiology and cancer. Biochim Biophysa Acta 1833:1732–1744
Meisamy S, Bolan PJ, Baker EH et al (2004) Neoadjuvant chemotherapy of locally advanced breast cancer: predicting response with in vivo 1H MR spectroscopy a pilot study at 4T. Radiology 233:424–431
Kumar M, Jagannathan NR, Seenu V et al (2006) Monitoring the therapeutic response of locally advanced breast cancer patients: Sequential in vivo proton MR spectroscopy study. J Magn Reson Imaging 24:325–332
Promkan M, Liu G, Patmasiriwat P et al (2011) BRCA1 suppresses the expression of survivin and promotes sensitivity to paclitaxel through the calcium sensing receptor (CaSR) in human breast cancer cells. Cell Calcium 49:79–88
Acknowledgments
The authors thank Mrs Teresa Morris, Coordinator of Aberdeen Biomedical Imaging, University of Aberdeen, UK, for his helpful revision of the manuscript, in particular for English editing.
Conflict of Interests
The authors declare that they have no conflict of interests.
Ethical Approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Baio, G., Rescinito, G., Rosa, F. et al. Correlation between Choline Peak at MR Spectroscopy and Calcium-Sensing Receptor Expression Level in Breast Cancer: A Preliminary Clinical Study. Mol Imaging Biol 17, 548–556 (2015). https://doi.org/10.1007/s11307-015-0823-y
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11307-015-0823-y