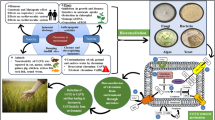
Abstract
Dissimilatory iron-reducing bacteria play a fundamental role in catalysing the redox transformations that ultimately control the mobility of As in anoxic environments, a process also controlled by the presence of competing anions. In this study, we investigated the decoupling of As from loaded Al and Fe (hydr)oxides by competing anions in the presence of iron-reducing bacteria. Hematite, goethite, ferrihydrite, gibbsite and three aluminium-substituted goethites (AlGts) were synthesised and loaded with arsenate, followed by anaerobic incubation with different phosphate or carbonate-containing media in the presence of catalytic iron-reducing bacteria. Soluble Al, As, Fe and P contents were measured in aliquots by inductively coupled plasma optical emission spectrometry following periodical sampling. Shewanella putrefaciens cells were able to utilise both non-crystalline and crystalline Fe (hydr)oxides as electron acceptors, releasing Fe and As into solution. Phosphate and carbonate affected the Fe bioreduction, probably due to the precipitation of metastable mineral phases and also to phosphate-induced stabilisation on the hydroxide surfaces. Phosphate precipitation acted as a sink for As, thus limiting its mobilisation. The highest fraction of desorbed As by phosphate was observed for gibbsite, followed by AlGts. Similarly, gibbsite showed significant amounts of arsenate displaced by carbonate. In spite of its low crystallinity, ferrihydrite was the most efficient compound in retaining arsenate, possibly due to As co-precipitation. This study provides new insight into the management of As-contaminated soils and sediments containing Al-goethites and gibbsite, where the Fe activity may be too low to co-precipitate As-bearing vivianite. Thus, the dynamics of As(V) in flooded soils are significant in agriculture and environmental management.



Similar content being viewed by others
References
Appelo, C. A. J., Van Der Weiden, M. J. J., Tournassat, C., & Charlet, L. (2002). Surface complexation of ferrous iron and carbonate on ferrihydrite and the mobilization of arsenic. Environmental Science & Technology, 36, 3096–3103.
Arai, Y., & Sparks, D. L. (2002). Residence time effects on arsenate surface speciation at the aluminium oxide–water interface. Soil Science, 167, 303–314.
Bonneville, S., Behrends, T., Cappellen, P. V., Hyacinthe, C., & Roling, W. F. M. (2006). Reduction of Fe(III) colloids by Shewanella putrefaciens: a kinetic model. Geochimica et Cosmochimica Acta, 70, 5842–5854.
Borch, T., Masue, Y., Kukkadapu, R. K., & Fendorf, S. (2007). Phosphate imposed limitations on biological reduction and alteration of ferrihydrite. Environmental Science & Technology, 41, 166–172.
Bousserrhine, N., Gasser, U. G., Jeanroy, E., & Berthelin, J. (1999). Bacterial and chemical reductive dissolution of Mn-, Co-, Cr-, and Al-substituted goethites. Geomicrobiology Journal, 16, 245–258.
Burnol, A., Garrido, F., Baranger, P., Joulian, C., Dictor, M. C., Bodenan, F., Morin, G., & Charlet, L. (2007). Decoupling of arsenic and iron release from ferrihydrite suspension under reducing conditions: a biogeochemical model. Geochemical Transactions, 8, 12.
Chacon, N., Flores, S., & Ganzalez, A. (2006). Implication of iron solubilization on soil phosphorus release in seasonally flooded forest of the lower Orinoco River, Venezuela. Soil Biology and Biochemistry, 38, 1494–1499.
Cummings, D. E., Caccavo, J. F., Fendorf, S., & Rosenzweig, F. (1999). Arsenic mobilization by the dissimilatory Fe(III)-reducing bacterium Shewanella alga BrY. Environmental Science & Technology, 33, 723–729.
Evangelou, V. P. (2001). Pyrite microencapsulation technologies: principles and potential field application. Ecological Engeneering, 17, 165–178.
Fendorf, S., Eick, M. J., Grossl, P., & Sparks, D. L. (1997). Arsenate and chromate retention mechanisms on goethite. 1. Surface structure. Environmental Science & Technology, 31, 315–320.
Goldberg, S., & Johnston, C. T. (2001). Mechanisms of arsenic adsorption on amorphous oxides evaluated using macroscopic measurements, vibrational spectroscopy, and surface complexation modelling. Journal of Colloid and Interface Science, 234, 204–216.
Gustafsson, J. P. (2000). Visual Minteq 2.30: a free equilibrium speciation model. Available at http://www2.lwr.kth.se/English/OurSoftware/Vminteq/download.html. Accessed 20 January 2008.
Haldar, M., & Mandal, L. N. (1981). Effect of Phosphorus and zinc on the growth and phosphorus, zinc, copper, iron and manganese nutrition of rice. Plant and Soil, 59, 415–425.
Hongshao, Z., & Stanforth, R. (2001). Competitive adsorption of phosphate and arsenate on goethite. Environmental Science & Technology, 35, 4753–4757.
Jeanroy, E., Rajot, J. L., Pillon, P., & Herbillon, A. J. (1991). Differential dissolution of hematite and goethite in dithionite and its implication on soil yellowing. Geoderma, 50, 79–94.
Kim, M. J., Nriagu, J., & Haack, S. (2002). Arsenic species and chemistry in groundwater of southeast Michigan. Environmental Pollution, 120, 379–390.
Kyle, J. H., Posner, A. M., & Quirk, J. P. (1975). Kinetics of isotopic exchange of phosphate adsorbed on gibbsite. Journal of Soil Science, 75, 34–43.
Ladeira, A. C. Q., & Ciminelli, V. S. T. (2004). Adsorption and desorption of arsenic on an oxisol and its constituents. Water Research, 38, 2087–2094.
Ladeira, A. C. Q., Ciminelli, V. S. T., Duarte, H. A., Alves, M. C. M., & Ramos, A. Y. (2001). Mechanism of anion retention from EXAFS and density functional calculations: arsenic (V) adsorbed on gibbsite. Geochimica et Cosmochimica Acta, 65(8), 1211–1217.
Leist, M., Casey, R. J., & Caridi, D. (2000). The management of arsenic wastes: problems and prospects. Journal of Hazardous Materials, 76(1), 125–138.
Makris, K. C., Sarkar, D., Parsons, J. G., Datta, R., & Gardea-Torresdey, J. L. (2007). Surface arsenic speciation of a drinking-water treatment residual using X-ray absorption spectroscopy. Journal of Colloid and Interface Science, 311, 544–550.
Manning, B. A., & Goldberg, S. (1996). Modelling competitive adsorption of arsenate with phosphate and molybdate on oxide minerals. Soil Science Society of American Journal, 60, 121–131.
Masue, Y., Loeppert, R. H., & Kramer, T. A. (2007). Arsenate and arsenite adsorption and desorption behaviour on coprecipitated aluminium:iron hydroxides. Environmental Science & Technology, 41, 837–842.
Matschullat, J. (2000). Arsenic in the geosphere—a review. The Science of the Total Environment, 249, 297–312.
Mello, J. W. V., Barrón, V., & Torrent, J. (1998). Phosphorus and iron mobilization in flooded soils from Brazil. Soil Science, 163, 122–132.
NHMRC, NRMMC (2004). Australian Drinking Water Guidelines. In: National Water Quality Management Strategy Paper No 6. National Health and Medical Research Council and Natural Resource Management Ministerial Council. Canberra: Australian Government Publishing Service.
O'Day, P. A. (2006). Chemistry and mineralogy of arsenic. Elements, 2(2), 77–83.
Radu, T., Subacz, J. L., Phillippi, J. M., & Barnett, M. O. (2005). Effects of dissolved carbonate on arsenic adsorption and mobility. Environmental Science & Technology, 39, 7875–7882.
Sahai, N., Lee, Y. J., Xu, H., Ciardelli, M., & Gaillard, J. F. (2007). Role of Fe(II) and phosphate in arsenic uptake by coprecipitation. Geochimica et Cosmochimica Acta, 71(13), 3193–3210.
Schwertmann, U. (1984). The Influence of aluminum on iron-oxides. 9. Dissolution of Al-goethites in 6 M HCl. Clay Mineralogy, 19, 9–19.
Schwertmann, U., & Cornell, R. M. (2000). Iron oxides in the laboratory: preparation and characterization (2nd ed.). Weinheim: Wiley-VCH. Completely revised and extended.
Sherman, D. M., & Randall, S. R. (2003). Surface complexation of arsenic(V) to iron(III) (hydr)oxides: structural mechanism from ab initio molecular geometries and EXAFS spectroscopy. Geochimica et Cosmochimica Acta, 67(22), 4223–4230.
Silva, J., Mello, J. W. V., Gasparon, M., Abrahão, W. A. P., Ciminelli, V. S. T., & Jong, T. (2010). The role of As-goethites on arsenate mobility. Water Research, 44, 5684–5692.
Smedley, P. L., & Kinniburgh, D. G. (2002). A review of the source, behaviour and distribution of arsenic in natural waters. Applied Geochemistry, 17, 517–568.
Thinnappan, V., Merrifield, C. M., Islam, F. S., Polya, D. A., Wincott, P., & Wogelius, R. A. (2008). A combined experimental study of vivianite and As(V) reactivity in the pH range 2–11. Applied Geochemistry, 23, 3187–3204.
Torrent, J., Schwertmann, U., & Barron, V. (1992). Fast and slow phosphate sorption by goethite-rich natural materials. Clays and Clay Mineralogy, 40, 14–21.
Wang, H. D., Harris, W. G., & Yuan, T. L. (1991). Relation between phosphorus and iron in Florida phosphatic soils. Soil Science Society of America Journal, 55, 554–560.
Zachara, J. M., Fredrickson, J. K., Smith, S. C., & Gassman, P. L. (2001). Solubilisation of Fe(III) oxide-bound trace metals by a dissimilatory Fe(III) reducing bacterium. Geochimica et Cosmochimica Acta, 65(1), 75–93.
Zhang, H., & Selim, H. M. (2008). Competitive sorption–desorption kinetics of arsenate and phosphate in soils. Soil Science, 173, 3–12.
Zhang, J. S., Stanforth, R., & Pehkonen, S. O. (2008). Irreversible adsorption of methyl arsenic, arsenate, and phosphate onto goethite in arsenic and phosphate binary systems. Journal of Colloid and Interface Science, 31, 35–43.
Acknowledgments
The support from the Minas Gerais State Foundation (FAPEMIG) and the Brazilian Research Council (CNPq) are gratefully acknowledged. Part of this study was carried out while the first author was a visiting researcher at The University of Queensland (UQ), with funding from CNPq and a UQ research grant. Laboratory work was carried out in the School of Earth Sciences at UQ (Australia) and in the Soil Department of the Federal University of Viçosa (Brazil). The authors are also grateful to Ms. Jenny Spratley (Australian Collection of Microorganisms at UQ), who provided the S. putrefaciens cells used in this study.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Silva, J., de Mello, J.W.V., Gasparon, M. et al. Effects of Competing Anions and Iron Bioreduction on Arsenic Desorption. Water Air Soil Pollut 223, 5707–5717 (2012). https://doi.org/10.1007/s11270-012-1308-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11270-012-1308-0