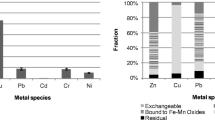
Abstract
In the present study a chemical washing process was applied to dredged sea and river sediments. Five chelating agents (ethylenediaminetetraacetic, nitrilotriacetic, citric, [S,S]-ethylenediaminedisuccinic and oxalic acids), three surfactants (sodium dodecyl sulphate, cetylpyridinium chloride and saponin Q), as well as monobasic potassium phosphate were used as the extracting agents. The effect of sonication was also tested as a conditioning step to couple with chelant-based extraction. The extraction experiments were performed in both single and multiple washing stages. The results indicated that, for given experimental conditions, the remediation efficiency was strongly dependent on the specific contaminant under concern, with contaminant speciation and distribution in the solid matrix as well as affinity for the extracting agent playing a major role in the decontamination process. In this respect, the study also showed that in the presence of multiple contaminants the use of a single extraction agent is not sufficient to attain adequate quality levels for the treated sediment.








Similar content being viewed by others
References
Alam, M. G. M., Tokunaga, S., & Maekawa, T. (2001). Extraction of arsenic in a synthetic arsenic-contaminated soil using phosphate. Chemosphere, 43(8), 1035–1041. doi:10.1016/S0045-6535(00)00205-8.
Allison, J. D., Brown, D. S., & Novo-Gradac, K. J. (1991). MINTEQA2/PRODEFA2. A geochemical assessment model for environmental systems. Version 3.0 User’s Manual. Athens, GA: U.S. Environmental Protection Agency.
Andrade, M. D., Prasher, S. O., & Hendershot, W. H. (2007). Optimizing the molarity of a EDTA washing solution for saturated-soil remediation of trace metal contaminated soils. Environmental Pollution, 147(3), 781–790. doi:10.1016/j.envpol.2006.07.010.
Chang, F.-C., Loa, S.-L., & Ko, C.-H. (2007). Recovery of copper and chelating agents from sludge extracting solutions. Separation and Purification Technology, 53(1), 49–56. doi:10.1016/j.seppur.2006.06.011.
Chang, S.-H., Wang, K.-S., Kuo, C.-Y., Chang, C.-Y., & Chou, C.-T. (2005). Remediation of metal-contaminated soil by an integrated soil washing-electrolysis process. Soil and Sediment Contamination, 14(6), 559–569. doi:10.1080/15320380500263758.
Ceremigna, D., Polettini, A., Pomi, R., Rolle, E., De Propris, L., Gabellini, M., et al.(2005). Comparing sediment washing yields using traditional and innovative biodegradable chelating agents, Proc. Third International Conference on Remediation of Contaminated Sediments, New Orleans, LA, USA (January 24–27, 2005).
Dezileau, L., Pizarro, C., & Rubio, M. A. (2007). Sequential extraction of iron in marine sediments from the Chilean continental margin. Marine Geology, 241(1–4), 111–116. doi:10.1016/j.margeo.2007.03.006.
Doong, R.-A., Wu, Y.-W., & Lei, W.-G. (1998). Surfactant enhanced remediation of cadmium contaminated soils. Water Science and Technology, 37(8), 65–71. doi:10.1016/S0273-1223(98)00235-2.
Elliott, H. A., Linn, J. H., & Shields, G. A. (1989). Role of Fe in extractive decontamination of Pb-polluted soils. Hazardous Waste & Hazardous Materials, 6(3), 223–229.
Ergican, E., Gecol, H., & Fuchs, A. (2005). The effect of co-occurring inorganic solutes on the removal of arsenic (V) from water using cationic surfactant micelles and an ultrafiltration membrane. Desalination, 181(1–3), 9–26. doi:10.1016/j.desal.2005.02.011.
Gecol, H., Ergican, E., & Fuchs, A. (2004). Molecular level separation of arsenic(V) from water using cationic surfactant micelles and ultrafiltration membrane. Journal of Membrane Science, 241(1), 105–119. doi:10.1016/j.memsci.2004.04.026.
Giannis, A., Gidarakos, E., & Skouta, A. (2007). Application of sodium dodecyl sulfate and humic acid as surfactants on electrokinetic remediation of cadmium-contaminated soil. Desalination, 211, 249–260. doi:10.1016/j.desal.2006.02.097.
Hong, K. J., Tokunaga, S., Ishigami, Y., & Kajiuchi, T. (2000). Extraction of heavy metals from MSW incinerator fly ash using saponins. Chemosphere, 41(3), 345–352. doi:10.1016/S0045-6535(99)00489-0.
Iqbal, J., Kim, H.-J., Yang, J.-S., Baek, K., & Yang, J.-W. (2007). Removal of arsenic from groundwater by micellar-enhanced ultrafiltration (MEUF). Chemosphere, 66(5), 970–976. doi:10.1016/j.chemosphere.2006.06.005.
IRSA.Istituto di Ricerca sulle Acque (Water Research Institute)–CNR, Consiglio Nazionale delle Ricerche (National Research Council) (1985). Metodiche analitiche per i fanghi: Parametri chimico-fisici, biochimici e biologici (Methods for the analysis of sludge: Physico-chemical, biochemical and biological parameters), Quaderni n. 64 (in Italian).
Jean, L., Bordas, F., & Bollinger, J.-C. (2007). Chromium and nickel mobilization from a contaminated soil using chelants. Environmental Pollution, 147(3), 729–736. doi:10.1016/j.envpol.2006.09.003.
Jean, L., Bordas, F., Gautier-Moussard, C., Vernay, P., Hitmi, A., & Bollinger, J. C. (2008). Effect of citric acid and EDTA on chromium and nickel uptake and translocation by Datura innoxia. Environmental Pollution, 153(3), 555–563. doi:10.1016/j.envpol.2007.09.013.
Kos, R., & Leštan, D. (2003). Induced phytoextraction/soil washing of lead using biodegradable chelate and permeable barriers. Environmental Science & Technology, 37(3), 624–629. doi:10.1021/es0200793.
Lo, I. M. C., & Zhang, W. (2005). Study on optimal conditions for recovery of EDTA from soil washing effluents. Journal of Environmental Engineering, 131(11), 1507–1513. doi:10.1061/(ASCE)0733-9372(2005)131:11(1507).
May, P. M., & Murray, K. (1991). JESS, a joint expert speciation system: II. The thermodynamic database. Talanta, 38(12), 1419–1426. doi:10.1016/0039-9140(91)80290-G.
Ministero delle Politiche Agricole e Forestali (Ministry for Agriculture and Forests) (1999). Metodi ufficiali di analisi chimica del suolo (Standard methods for chemical analysis of soil). D.M. 13/9/1999, Suppl. Ord. n. 185 G.U. n. 248 (in Italian).
Mulligan, C., Yong, R., Gibbs, B., James, S., & Bennett, H. P. J. (1999). Metal removal from contaminated soil and sediments by the biosurfactant surfactin. Environmental Science & Technology, 33(21), 3812–3820. doi:10.1021/es9813055.
Neaman, A., Waller, B., Mouélé, F., Trolard, F., & Bourrié, G. (2004). Improved methods for selective dissolution of manganese oxides from soils and rocks. European Journal of Soil Science, 55(1), 47–54. doi:10.1046/j.1351-0754.2003.0545.x.
Nord, L. I., Kenne, L., & Jacobsson, S. P. (2001). Multivariate analysis of 1H NMR spectra for saponins from Quillaja saponaria Molina. Analytica Chimica Acta, 446(1–2), 199–209. doi:10.1016/S0003-2670(01)00934-5.
Paria, S. (2008). Surfactant-enhanced remediation of organic contaminated soil and water. Advances in Colloid and Interface Science, 138(1), 24–58. doi:10.1016/j.cis.2007.11.001.
Peters, R. W. (1999). Chelant extraction of heavy metals from contaminated soils. Journal of Hazardous Materials, 66(1), 151–210. doi:10.1016/S0304-3894(99)00010-2.
Polettini, A., Pomi, R., & Rolle, E. (2007). The effect of operating variables on chelant-assisted remediation of contaminated dredged sediment. Chemosphere, 66(5), 866–877. doi:10.1016/j.chemosphere.2006.06.023.
Polettini, A., Pomi, R., Rolle, E., Ceremigna, D., De Propris, L., Gabellini, M., et al. (2006). A kinetic study of chelant-assisted remediation of contaminated dredged sediment. Journal of Hazardous Materials, B137(3), 1458–1465. doi:10.1016/j.jhazmat.2006.04.022.
Rämö, J. (2003). Hydrogen peroxide–metals–chelating agents; interactions and analytical techniques. Academic Dissertation. Acta Universitatis Ouluensis Technica, C 183.
Shin, M., & Barrington, S. (2005). Effectiveness of the iodide ligand along with two surfactants on desorbing heavy metals from soils. Water, Air, and Soil Pollution, 161(1–4), 193–208.
Tandy, S., Bossart, K., Mueller, R., Ritschel, J., Hauser, L., Schulin, R., et al. (2004). Extraction of heavy metals from soils using biodegradable chelating agents. Environmental Science & Technology, 38(3), 937–944. doi:10.1021/es0348750.
Tessier, A., Campbell, P. G. C., & Bisson, M. (1979). Sequential extraction procedure for the speciation of particulate trace metals. Analytical Chemistry, 51(7), 844–850. doi:10.1021/ac50043a017.
Tokunaga, S., & Hakuta, T. (2002). Acid washing and stabilization of an artificial arsenic-contaminated soil. Chemosphere, 46(1), 31–38. doi:10.1016/S0045-6535(01)00094-7.
Vandevivere, P., Hammes, F., Verstraete, W., Feijtel, T., & Schowanek, D. (2001b). Metal decontamination of soil, sediment, and sewage sludge by means of transition metal chelant [S,S]-EDDS. Journal of Environmental Engineering, 127(9), 802–811. doi:10.1061/(ASCE)0733-9372(2001)127:9(802).
Vandevivere, P. C., Saveyn, H., Verstraete, W., Feijtel, T. C. J., & Schowanek, D. R. (2001a). Biodegradation of metal-[S,S]-EDDS complexes. Environmental Science & Technology, 35(9), 1765–1770. doi:10.1021/es0001153.
Yuan, C., & Chiang, T.-S. (2008). Enhancement of electrokinetic remediation of arsenic spiked soil by chemical reagents. Journal of Hazardous Materials, 152(1), 309–315. doi:10.1016/j.jhazmat.2007.06.099.
Zhang, W., Tsang, D. C. W., & Lo, I. M. C. (2008). Removal of Pb by EDTA-washing in the presence of hydrophobic organic contaminants or anionic surfactant. Journal of Hazardous Materials, 155(3), 433-439. doi:10.1016/j.jhazmat.2007.11.084.
Author information
Authors and Affiliations
Corresponding author
Electronic Supplementary Materials
Below is the link to the electronic supplementary materials.
Fig. SM1
Residual metal concentrations resulting from single-stage washing (river sediments)—average values from two replicates; error bars are drawn at one standard deviation (PDF 62.6 KB)
Fig. SM2
Residual metal concentrations resulting from two-stage washing (river sediments) (PDF 21.2 KB)
Fig. SM3
Residual metal concentrations resulting from four-stage washing (river sediments) (PDF 31.2 KB)
Rights and permissions
About this article
Cite this article
Polettini, A., Pomi, R. & Calcagnoli, G. Assisted Washing for Heavy Metal and Metalloid Removal from Contaminated Dredged Materials. Water Air Soil Pollut 196, 183–198 (2009). https://doi.org/10.1007/s11270-008-9767-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11270-008-9767-z