Abstract
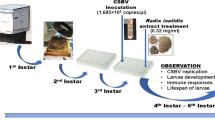
Sacbrood virus (SBV) is a serious threat to honey bees. Currently, there is no specific drug available for the treatment of SBV that does not affect the quality of the bee product. RNA interference (RNAi) is an important antiviral strategy for disease control. To effectively utilize this technology, the large-scale production and purification of double-stranded RNA (dsRNA) is necessary. Here, a dsRNA-expressing plasmid targeting the VP1 gene of Chinese sacbrood virus (CSBV) was constructed and expressed in Escherichia coli (E. coli) HT115 (DE3). After lysing and ethanol precipitation from E. coli, dsRNA VP1 was purified with an anion exchange chromatography column. Second instar larvae of Apis cerana were fed the purified dsRNA VP1. A significant decrease in larval mortality and the level of expression of the VP1 gene after CSBV infection was demonstrated after the ingestion of dsRNA VP1. This result provides a potential method for the large-scale production of dsRNA to protect A. cerana from CSBV infection.






Similar content being viewed by others
References
E.J. Bailes, J. Ollerton, J.G. Pattrick, B.J. Glover, Curr. Opin. Plant Biol. 26, 72–79 (2015)
D.P. Abrol, Pollination Biology: Biodiversity Conservation and Agricultural Production (Springer, Netherlands, 2012), pp. 603–633
M. Shen, L. Cui, N. Ostiguy, D. Cox-Foster, J. Gen. Virol. 86, 2281–2289 (2005)
O. Berenyi, T. Bakonyi, I. Derakhshifar, H. Koglberger, N. Nowotny, Appl. Environ. Microbiol. 72, 2414–2420 (2006)
Y.P. Chen, J.S. Pettis, A. Collins, M.F. Feldlaufer, Appl. Environ. Microbiol. 72, 606–611 (2006)
L. Bailey, A.J. Gibbs, R.D. Woods, Virology 23, 425–429 (1964)
R.C. Ghosh, B.V. Ball, M.M. Willcocks, M.J. Carter, J. Gen. Virol. 80, 1541–1549 (1999)
B.V. Ball, L. Bailey, in Honey Bee Pests, Predators and Diseases, ed. by R.A. Morse, K. Flottum (Northern Bee Books, West Yorkshire, 2013), pp. 11–32
M.X. Ma, J.H. Liu, Y.J. Song, L. Li, Y.F. Li, PLoS ONE 8(2), e52670 (2013)
J. Zhang, J. Feng, Y. Liang, D. Chen, Z.H. Zhou, Q. Zhang, X. Lu, Sci. China C Life Sci. 44, 443–448 (2001)
M.X. Ma, M. Li, J. Cheng, S. Yang, S.D. Wang, P.F. Li, Comp. Funct. Genomics 2011, 409386 (2011)
S.M. Hammond, A.A. Caudy, G.J. Hannon, Nat. Rev. Genet. 2, 110–119 (2001)
J.T. van Mierlo, A.W. Bronkhorst, G.J. Overheul, S.A. Sadanandan, J.O. Ekstrom, M. Heestermans, D. Hultmark, C. Antoniewski, R.P. van Rij, PLoS Pathog. 8, e1002872 (2012)
X. Liu, Y. Zhang, X. Yan, R. Han, Curr. Microbiol. 61, 422–428 (2010)
E. Maori, N. Paldi, S. Shafir, H. Kalev, E. Tsur, E. Glick, I. Sela, Insect Mol. Biol. 18, 55–60 (2009)
M. Beye, S. Hartel, A. Hagen, M. Hasselmann, S.W. Omholt, Insect Mol. Biol. 11, 527–532 (2002)
G.V. Amdam, Z.L. Simoes, K.R. Guidugli, K. Norberg, S.W. Omholt, BMC Biotechnol. 3, 1 (2003)
K. Aronstein, T. Pankiw, E. Saldivar, J. Apicult. Res 45, 20–24 (2006)
K. Aronstein, T. Pankiw, E. Saldivar, Nature 443, 931–949 (2006)
F.M. Nunes, Z.L. Simoes, Insect Biochem. Mol. Biol. 39, 157–160 (2009)
Y. Chen, Y. Zhao, J. Hammond, H.T. Hsu, J. Evans, M. Feldlaufer, J. Invertebr. Pathol. 87, 84–93 (2004)
X. Yan, J.H. Chen, R.C. Han, Sociobiology 53, 687–694 (2009)
K.M. Hong, H. Najjar, M. Hawley, R.D. Press, Clin. Chem. 50, 846–856 (2004)
Q. Yao, L. Gao, K.P. Chen, H.Z. Gang, Acta Entomol. Sin. 48, 871–875 (2005)
L. Timmons, A. Fire, Nature 395, 854 (1998)
R.S. Kamath, M. Martinez-Campos, P. Zipperlen, A.G. Fraser, J. Ahringer, Genome Biol. 2, RESEARCH0002 (2001)
H. Tian, H. Peng, Q. Yao, H. Chen, Q. Xie, B. Tang, W. Zhang, PLoS ONE 4, e6225 (2009)
A. Romanovskaya, L.P. Sarin, D.H. Bamford, M.M. Poranen, J. Chromatogr. A 1278, 54–60 (2013)
A. Patel, M.K. Fondrk, O. Kaftanoglu, C. Emore, G. Hunt, K. Frederick, G.V. Amdam, PLoS ONE 2, e509 (2007)
Y. Garbian, E. Maori, H. Kalev, S. Shafir, I. Sela, PLoS Pathog. 8, e1003035 (2012)
W. Hunter, J. Ellis, D. Vanengelsdorp, J. Hayes, D. Westervelt, E. Glick, M. Williams, I. Sela, E. Maori, J. Pettis, D. Cox-Foster, N. Paldi, PLoS Pathog. 6, e1001160 (2010)
F. Tenllado, B. Martinez-Garcia, M. Vargas, J.R. Diaz-Ruiz, BMC Biotechnol. 3, 3 (2003)
G.H. Yin, Z.N. Sun, Y.Z. Song, H.L. An, C.X. Zhu, F.J. Wen, Appl. Biochem. Biotechnol. 162, 1901–1914 (2010)
M. Sarathi, M.C. Simon, V.P. Ahmed, S.R. Kumar, A.S. Hameed, Mar. Biotechnol. 10, 198–206 (2008)
T. Thammasorn, P. Somchai, C. Laosutthipong, S. Jitrakorn, S. Wongtripop, S. Thitamadee, B. Withyachumnarnkul, V. Saksmerprome, Antiviral Res. 100, 202–206 (2013)
P.C. Lima, N.A. Botwright, J.O. Harris, M. Cook, Mar. Biotechnol. 16, 447–455 (2014)
Acknowledgments
This work was supported by the Guangdong Provincial of Science and Technology (No: 2014A020208074 and 2015B070701019), Funds for Environment Construction & Capacity Building of GDAS’ Research Platform (No: 2016GDASPT-0107) and the National Natural Science Foundation of China (No: 31301924).
Author information
Authors and Affiliations
Corresponding author
Additional information
Edited by A. Lorena Passarelli.
Rights and permissions
About this article
Cite this article
Zhang, J., Zhang, Y. & Han, R. The high-throughput production of dsRNA against sacbrood virus for use in the honey bee Apis cerana (Hymenoptera: Apidae). Virus Genes 52, 698–705 (2016). https://doi.org/10.1007/s11262-016-1346-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11262-016-1346-6