Abstract
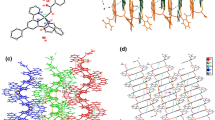
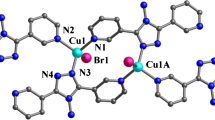
Three complexes, namely [Cu(dpt)2(H2O)4]·2H2O (1), [Cu3I2(dpt)] n (2) and [Cu6Br3(dpt)3] n (3), have been obtained from the reactions of different copper salts with 1H-3,5-bis(4-pyridyl)-1,2,4-triazole (Hdpt). Complex 1 is mononuclear such that the CuII atom exhibits an octahedral coordination geometry involving monodentate dpt ligands, being synthesized from CuII nitrate and Hdpt at ambient temperature, while the cuprous halide cluster-based coordination polymers 2 and 3 were prepared by solvothermal reactions of CuI or CuBr with Hdpt. Complex 2 consists of [Cu3I2] n chains, which are further connected by the μ 4-dpt bridges to form a 4,6-connected three-dimensional coordination polymer. Complex 3 is a 3,3,9-connected net based on [Cu6Br3] building blocks, linked by μ 3-dpt bridges. The photoluminescence properties of complex 2 in the solid state at ambient temperature have been investigated.
Graphical Abstract
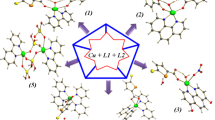
Syntheses and structures of three complexes [Cu(dpt)2(H2O)4]·2H2O (1), [Cu3I2(dpt)] n (2) and [Cu6Br3(dpt)3] n (3) (Hdpt = 1H-3,5-bis(4-pyridyl)-1,2,4-triazole) are described, in which the effect of the rich connectivity of the cuprous halide clusters, supported by the changeable coordination modes of dpt ligand, is revealed.






Similar content being viewed by others
References
Lin ZJ, Tong ML (2011) Coord Chem Rev 255:421–450
Halder GJ, Kepert CJ, Moubaraki B, Murray KS, Cashion JD (2002) Science 298:1762–1765
Moulton B, Zaworotko MJ (2001) Chem Rev 101:1629–1658
Hosseini MW (2003) Coord Chem Rev 240:157–166
Li H, Eddaoudi M, O’Keeffe M, Yaghi OM (1999) Nature 402:276–279
Hu S, Yu FY, Zhang P, Lin DR (2013) Dalton Trans 42:7731–7740
Chui SSY, Lo SMF, Charmant JPH, Orpen AG, Williams ID (1999) Science 283:1148–1150
Hu S, Chen JC, Tong ML, Wang B, Yan YX, Batten SR (2005) Angew Chem Int Ed 44:5471–5475
Hu S, Meng ZS, Tong ML (2010) Cryst Growth Des 10:1742–1748
Hu S, Liu JL, Meng ZS, Zheng YZ, Lan YH, Powell AK, Tong ML (2011) Dalton Trans 40:27–30
Ford PC, Vogler A (1993) Acc Chem Res 26:220–226
Peng R, Li M, Li D (2010) Coord Chem Rev 254:1–18
Hu S, Tong ML (2005) Dalton Trans 34:1165–1167
Hu S, Zhou AJ, Zhang YH, Tong ML (2006) Cryst Growth Des 6:2543–2550
Wang J, Zheng SL, Hu S, Zhang YH, Tong ML (2007) Inorg Chem 46:795–800
Hu S, Zhang ZM, Meng ZS, Tong ML (2010) CrystEngComm 12:4378–4385
Hu S, Yu FY, Yan Y, Tong ML (2011) Inorg Chem Commun 14:622–625
Hu S, Yu FY, Zhang P, Zhou AJ (2012) Eur J Inorg Chem 23:3669–3673
Xie XF, Chen SP, Xia ZQ, Gao SL (2009) Polyhedron 28:679–688
Huang FP, Tian JL, Gu W, Yan SP (2010) Inorg Chem Comm 13:90–94
Huang FP, Bian HD, Yu Q, Tian JL, Liang H, Yan SP, Liao DZ, Cheng P (2011) CrystEngComm 13:6538–6548
Huang FP, Tian JL, Chen GJ, Li DD, Gu W, Liu X, Yan SP, Liao DZ, Cheng P (2010) CrystEngComm 12:269–1279
Sheldrick GM (1997) SADABS 2.05. Empirical Absorption Correction Program, University of Göttingen, Germany
Sheldrick GM (2008) Acta Cryst A A64:112–122
Luo F, Long GJ, Che YX, Zheng JM (2008) Cryst Growth Des 8:3511–3513
Xu J, Yao XQ, Huang LF, Li YZ, Zheng HG (2011) CrystEngComm 13:857–865
Ma LF, Wang YY, Wang LY, Lu DH, Batten SR, Wang JG (2009) Cryst Growth Des 9:2036–2038
Fang ZL, Yang WB, He JG, Ding KN, Wu XY, Zhang QS, Yu RM, Lu CZ (2012) CrystEngComm 14:4794–4800
Zhang JP, Lin YY, Huang XC, Chen XM (2005) J Am Chem Soc 127:5495–5506
Ford PC, Cariati E, Bourassa J (1999) Chem Rev 99:3625–3648
Acknowledgments
This work was supported by the National Natural Science Foundation of China (Grant No. 21407028), the Excellent Young Teacher Development Project of Universities in Guangdong Province (Grant No. 261532106) and the Guangzhou Science and Technology Program (Grant No. 201510010156).
Author information
Authors and Affiliations
Corresponding authors
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Hu, S., Lin, D., Xie, Z. et al. Coordinative versatility of 3,5-bis(4-pyridyl)-1,2,4-triazole in the construction of monomeric copper(II) and polymeric copper(I) complexes. Transition Met Chem 40, 623–629 (2015). https://doi.org/10.1007/s11243-015-9955-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11243-015-9955-9