Abstract
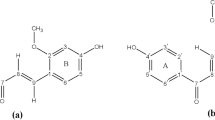
Gallic acid, one of the naturally occurring antioxidants, has been characterized in terms of its structural and chemical properties using the density functional approach. The intramolecular interactions have been identified using natural bond orbital (NBO) analysis. The gallic acid anion and dianion have also been studied for determining the first two pK a values. Further, an investigation of the molecular orbitals reveals a dependence of the frontier molecular orbitals on external factors, viz. pH and dielectric of the medium. Consequently, the electronic spectra have been simulated in a range of solvents using steered molecular dynamics. Furthermore, the IR and NMR spectra of the neutral and the deprotonated state have been simulated. Additionally, the spectra have been experimentally recorded to validate the methodology followed. The present work also examines the variation in global and local reactivities of gallic acid under the influence of external factors. Each atom has been quantitatively assessed for its susceptibility towards nucleophilic, electrophilic, and radical attack.








Similar content being viewed by others
References
Giftson JS, Jayanthi S, Nalini N (2010) Chemopreventive efficacy of gallic acid, an antioxidant and anticarcinogenic polyphenol, against 1, 2-dimethyl hydrazine induced rat colon carcinogenesis. Investig New Drug 28:251–259
Ou TT, Wang CJ, Lee YS, Wu CH, Lee HJ (2010) Gallic acid induces G2/M phase cell cycle arrest via regulating 14-3-3β release from cdc25c and chk2 activation in human bladder transitional carcinoma cells. Mol Nutr Food Res 54:1781–1790
Palafox-Carlos H, Gil-Chávez J, Sotelo-Mundo R, Namiesnik J, Gorinstein S, González-Aguilar GA (2012) Antioxidant interactions between major phenolic compounds found in ‘ataulfo’ mango pulp: chlorogenic, gallic, protocatechuic and vanillic acids. Molecules 17:12657–12664
Yoshioka K, Kataoka T, Hayashi T, Hasegawa M, Ishi Y, Hibasami H (2000) Induction of apoptosis by gallic acid in human stomach cancer KATO III and colon adenocarcinoma COLO 205 cell lines. Oncol Rep 7:1221–1223
You BR, Kim SZ, Kim SH, Park WH (2011) Gallic acid-induced lung cancer cell death is accompanied by ROS increase and glutathione depletion. Mol Cell Biochem 357:295–303
You BR, Moon HJ, Han YH, Park WH (2010) Gallic acid inhibits the growth of HeLa cervical cancer cells via apoptosis and/or necrosis. Food Chem Toxicol 48:1334–1340
Badhani B, Sharma N, Kakkar R (2015) Gallic acid: a versatile antioxidant with promising therapeutic and industrial applications. RSC Adv 5:27540–27557
Elango S, Balwas R, Padma VV (2011) Gallic acid isolated from pomegranate peel extract induces reactive oxygen species mediated apoptosis in A549 cell line. J Cancer Ther 2:638–645
González-Abuín N, Martínez-Micaelo N, Margalef M, Blay M, Arola-Arnal A, Muguerza B, Ardévol A, Pinent M (2014) A grape seed extract increases active glucagon-like peptide-1 levels after an oral glucose load in rats. Food Funct 5:2357–2364
Hsiang CY, Hseu YC, Chang YC, Kumar KJS, Ho TY, Yang HL (2013) Toona sinensis and its major bioactive compound gallic acid inhibit LPS-induced inflammation in nuclear factor-κB transgenic mice as evaluated by in vivo bioluminescence imaging. Food Chem 136:426–434
Du GJ, Wang CZ, Qi LW, Zhang ZY, Calway T, He TC, Du W, Yuan CS (2013) The synergistic apoptotic interaction of panaxadiol and epigallocatechin gallate in human colorectal cancer cells. Phytother Res 27:272–277
Gray AL, Stephens CA, Bigelow RL, Coleman DT, Cardelli JA (2014) The polyphenols (−)-epigallocatechin-3-gallate and luteolin synergistically inhibit TGF-β-induced myofibroblast phenotypes through RhoA and ERK inhibition. PLoS One 9:e109208
Lecumberri E, Dupertuis YM, Miralbell R, Pichard C (2013) Green tea polyphenol epigallocatechin-3-gallate (EGCG) as adjuvant in cancer therapy. Clin Nutr 32:894–903
Locatelli C, Rosso R, Santos-Silva MS, de Souza CA, Licinio MA, Leal P, Bazzo ML, Yunes RA, Creczynsk-Pasa TB (2008) Ester derivatives of gallic acid with potential toxicity toward L1210 leukemia cells. Bioorg Med Chem 16:3791–3799
Lu Z, Nie G, Belton PS, Tang H, Zhao B (2006) Structure–activity relationship analysis of antioxidant ability and neuroprotective effect of gallic acid derivatives. Neurochem Int 48:263–274
Saxena HO, Faridi U, Srivastava S, Kumar JK, Darokar MP, Luqman S, Chanotiya CS, Krishna V, Negi AS, Khanuja SPS (2008) Gallic acid-based indanone derivatives as anticancer agents. Bio Med Chem Lett 18:3914–3918
Zeng L, Holly JM, Perks CM (2014) Effects of physiological levels of the green tea extract epigallocatechin-3-gallate on breast cancer cells. Front Endocrinol (Lausanne) 5:61
Chen YH, Luo YM (2011) Oxidation stability of biodiesel derived from free fatty acids associated with kinetics of antioxidants. Fuel Process Technol 92:1387–1393
Jackson JK, Zhao J, Wong W, Burt HM (2010) The inhibition of collagenase induced degradation of collagen by the galloyl-containing polyphenols tannic acid, epigallocatechin gallate and epicatechin gallate. J Mater Sci Mater Med 21:1435–1443
Jaén JA, González L, Vargas A, Olave G (2003) Gallic acid, ellagic acid and pyrogallol reaction with metallic iron. Hyperfine Interact 148/149:227–235
Ramamurthy G, Krishnamoorthy G, Sastry TP, Mandal AB (2014) Rationalized method to enhance the chromium uptake in tanning process: role of gallic acid. Clean Techn Environ Policy 16:647–654
Sroka Z, Cisowski W (2003) Hydrogen peroxide scavenging, antioxidant and anti-radical activity of some phenolic compounds. Food Chem Toxicol 41:753–758
Mohammed-Ziegler I, Billes F (2002) Vibrational spectroscopic calculations on pyrogallol and gallic acid. J Mol Struc (Theochem) 618:259–265
Cappelli C, Mennucci B, Monti S (2005) Environmental effects on the spectroscopic properties of gallic acid: a combined classical and quantum mechanical study. J Phys Chem A 109:1933–1943
Pardeshi S, Dhodapkar R, Kumar A (2013) Quantum chemical density functional theory studies on the molecular structure and vibrational spectra of gallic acid imprinted polymers. Spectrochim Acta Part A: Mol Biomol Spectrosc 116:562–573
Leopoldini M, Marino T, Russo N, Toscano M (2004) Antioxidant properties of phenolic compounds: H-atom versus electron transfer mechanism. J Phys Chem A 108:4916–4922
Wright JS, Johnson ER, DiLabio GA (2001) Predicting the activity of phenolic antioxidants: theoretical method, analysis of substituent effects, and application to major families of antioxidants. J Am Chem Soc 123:1173–1183
Masoud MS, Hagagg SS, Ali AE, Nasrc NM (2012) Solvatochromic behavior of the electronic absorption spectra of gallic acid and some of its azo derivatives. Spectrochim Acta Part A: Mol Biomol Spectrosc 94:256–264
Masoud MS, Ali AE, Haggag SS, Nasr NM (2014) Spectroscopic studies on gallic acid and its azo derivatives and their iron(III) complexes. Spectrochim Acta Part A: Mol Biomol Spectrosc 120:505–511
Becke AD (1988) Density-functional exchange-energy approximation with correct asymptotic behaviour. Phys Rev A 38:3098–3100
Lee C, Yang W, Parr RG (1988) Development of the Colle-Salvetti correlation-energy formula into a functional of the electron density. Phys Rev B: Condensed Matter and Materials Physics 37:785–789
Vosko SH, Wilk L, Nusair M (1980) Accurate spin-dependent electron liquid correlation energies for local spin density calculations: a critical analysis. Can J Phys 58:1200–1211
Frisch MJ, Trucks GW, Schlegel HB, Scuseria GE, Robb MA, Cheeseman JR, Scalmani G, Barone V, Mennucci B, Petersson GA, Nakatsuji H, Caricato M, Li X, Hratchian HP, Izmaylov AF, Bloino J, Zheng G, Sonnenberg JL, Hada M, Ehara M, Toyota K, Fukuda R, Hasegawa J, Ishida M, Nakajima T, Honda Y, Kitao O, Nakai H, Vreven T, Montgomery JA Jr, Peralta JE, Ogliaro F, Bearpark M, Heyd JJ, Brothers E, Kudin KN, Staroverov VN, Kobayashi R, Normand J, Raghavachari K, Rendell A, Burant JC, Iyengar SS, Tomasi J, Cossi M, Rega N, Millam NJ, Klene M, Knox JE, Cross JB, Bakken V, Adamo C, Jaramillo J, Gomperts R, Stratmann RE, Yazyev O, Austin AJ, Cammi R, Pomelli C, Ochterski JW, Martin RL, Morokuma K, Zakrzewski VG, Voth GA, Salvador P, Dannenberg JJ, Dapprich S, Daniels AD, Farkas Ö, Foresman JB, Ortiz JV, Cioslowski J, Fox DJ (2009) Gaussian 09, Revision C.01,Gaussian Inc., Wallingford
Carpenter JE, Weinhold F (1988) Analysis of the geometry of the hydroxymethyl radical by the “different hybrids for different spins” natural bond orbital procedure. JMol Struct (Theochem) 169:41–62
Glendening ED, Reed AE, Carpenter JE, Weinhold F (2009) NBO Version 3.1
Reed AE, Weinhold F (1983) Natural bond orbital analysis of near‐Hartree–Fock water dimer. J Chem Phys 78:4066–4073
Reed AE, Curtiss LA, Weinhold F (1988) Intermolecular interactions from a natural bond orbital, donor-acceptor viewpoint. Chem Rev 88:899–926
Reed AE, Weinstock RB, Weinhold F (1985) Natural population analysis. J Chem Phys 83:735–746
Marenich AV, Cramer CJ, Truhlar DG (2009) Universal solvation model based on solute electron density and on a continuum model of the solvent defined by the bulk dielectric constant and atomic surface tensions. J Phys Chem B 113:6378–6396
Saielli G (2010) Differential solvation free energies of oxonium and ammonium ions: insights from quantum chemical calculations. J Phys Chem A 114:7261–7265
Kakkar R, Bhandari M (2013) Theoretical investigation of the alloxan–dialuric acid redox cycle. Int J Quant Chem 113:2060–2069
Muir RM, Ibáñez AM, Uratsu SL, Ingham ES, Leslie CA, McGranahan GH, Batra N, Goyal S, Joseph J, Jemmis ED, Dandekar AM (2011) Mechanism of gallic acid biosynthesis in bacteria (Escherichia coli) and walnut (Juglans regia). Plant Mol Biol 75:555–565
Hirun N, Saithong S, Pakawatchai C, Tantishaiyakul V (2011) 3,4,5-Trihydroxybenzoic acid. Acta Crystallogr E67:o787
Okabe N, Kyoyama H, Suzuki M (2001) Gallic acid monohydrate. Acta Crystallogr E57:o764–o766
Zhao J, Khan IA, Fronczek FR (2011) Gallic acid. Acta Crystallogr E67:o316–o317
Schleyer PVR, Maerker C, Dransfeld A, Jiao H, Hommes NJRVE (1996) Nucleus-independent chemical shifts: a simple and efficient aromaticity probe. J Am Chem Soc 118:6317–6318
Kelly CP, Cramer CJ, Truhlar DG (2006) Aqueous solvation free energies of ions and ion−water clusters based on an accurate value for the absolute aqueous solvation free energy of the proton. J Phys Chem B 110:16066–16081
Tissandier MD, Cowen KA, Feng WY, Gundlach E, Cohen MH, Earhart AD, Coe JV (1998) The proton’s absolute aqueous enthalpy and Gibbs free energy of solvation from cluster-ion solvation data. J Phys Chem A 102:7787–7794
Šolc R, Gerzabek MH, Lischka H, Tunega D (2014) Radical sites in humic acids: a theoretical study on protocatechuic and gallic acids. Comp Theor Chem 1032:42–49
Kakkar R, Bhandari M, Gaba R (2012) Tautomeric transformations and reactivity of alloxan. Comput Theor Chem 986:14–24
Maynard AT, Huang M, Rice WG, Covell DG (1998) Reactivity of the HIV-1 nucleocapsid protein p7 zinc finger domains from the perspective of density-functional theory. Proc Natl Acad Sci 95:11578–11583
Parr RG, von Szentpály L, Liu S (1999) Electrophilicity index. J Am Chem Soc 121:1922–1924
Domingo LR, Pérez P (2011) The nucleophilicity N index in organic chemistry. Org Biomol Chem 9:7168–7175
Domingo LR, Pérez P (2013) Global and local reactivity indices for electrophilic/nucleophilic free radicals. Org Biomol Chem 11:4350–4358
Domingo LR, Chamorro E, Pérez P (2008) Understanding the reactivity of captodative ethylenes in polar cycloaddition reactions. A theoretical study. J Org Chem 73:4615–4624
Parr RG, Yang W (1984) Density functional approach to the frontier-electron theory of chemical reactivity. J Am Chem Soc 106:4049–4050
Bauernschmitt R, Ahlrichs R (1996) Treatment of electronic excitations within the adiabatic approximation of time dependent density functional theory. Chem Phys Lett 256:454–464
Casida ME, Jamorski C, Casida KC, Salahub DR (1998) Molecular excitation energies to high-lying bound states from time-dependent density-functional response theory: characterization and correction of the time-dependent local density approximation ionization threshold. J Chem Phys 108:4439–4449
Stratmann RE, Scuseria GE, Frisch MJ (1998) An efficient implementation of time-dependent density-functional theory for the calculation of excitation energies of large molecules. J Chem Phys 109:8218–8224
Andersson MP, Uvdal P (2005) New scale factors for harmonic vibrational frequencies using the B3LYP density functional method with the triple-ζ basis set 6-311+G(d,p). J Phys Chem A 109:2937–2941
Wolinski K, Hinton JF, Pulay P (1990) Efficient implementation of the gauge-independent atomic orbital method for NMR chemical shift calculations. J Am Chem Soc 112:8251–8260
Eldahshan OA (2011) Isolation and structure elucidation of phenolic compounds of carob leaves grown in Egypt. Curr Res J Biol Sci 3:52–55
Acknowledgements
One of the authors (B.B.) thanks the Council of Scientific and Industrial Research (CSIR), New Delhi, for Senior Research Fellowship. The authors thank Delhi University’s “Scheme to Strengthen Doctoral Research by Providing Funds to Faculty.” We also thank USIC-CIF, University of Delhi, for providing facilities to acquire FT-IR and NMR data.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors report no conflict of interest.
Electronic supplementary material
ESM 1
(DOCX 1522 kb)
Rights and permissions
About this article
Cite this article
Badhani, B., Kakkar, R. DFT study of structural and electronic properties of gallic acid and its anions in gas phase and in aqueous solution. Struct Chem 28, 1789–1802 (2017). https://doi.org/10.1007/s11224-017-0958-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11224-017-0958-3