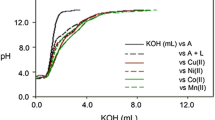
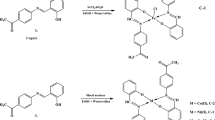
Abstract
Structures of the main resonance and tautomeric forms of three Schiff bases of 2-hydroxy-1-naphthaldehyde (1OXA), 5-bromosalicylaldehyde (2OXA) and 5-nitrosalicylaldehyde (3OXA) with 1,8-diamine-3,6-dioxaoctane, before and after protonation and complexation of monovalent metal cations, have been studied by FTIR, 1H, 13C and 15N NMR methods. The spectroscopic investigations provided clear evidence that the Schiff bases exist in acetonitrile solution as three different tautomers: 1OXA in enamine-oxo, 2OXA in imine-hydroxy and 3OXA in a low-barrier O···H···N imine-oxo forms. It was found on the basis of multinuclear NMR studies, that in solid state, the enamine-oxo and imine-hydroxy tautomers are formed exclusively, but not the untypical imine-oxo tautomer, which requires strong stabilisation by solvent molecules in solution. MOG-PM6 calculations of the different tautomers allowed visualisation of their energetically the most favourable structures. Protonation of 1OXA–3OXA Schiff bases leads to formation of common forms, i.e. protonated imine-hydroxy structure, irrespectively of the structure of tautomer before protonation. In turn, coordination of monovalent metal cations implies common formation of zwitterionic forms within all studied ligands in solution. Application of FT-IR and NMR titrations in combination with ESI MS spectrometry revealed the nature and the structure of OXA complexes, whose formation is impeded by the intra- and intermolecular interactions. MOG-PM6 calculations allowed visualisation of Li+ and Na+ metal coordination sphere geometries within structure of all investigated complexes, stabilised by intermolecular interactions with solvent molecules.









Similar content being viewed by others
References
Hadjoudis E (1995) Photochromic and thermochromic anils. Mol Eng 5:301–337 and references cited therein
Alarcón SH, Olivieri AC, Nordon A, Harris RK (1996) Solid-state electronic absorption, fluorescence and 13C CPMAS NMR spectroscopic study of thermo- and photo-chromic aromatic Schiff bases. J Chem Soc Perkin Trans 20(11):2293–2296
Alarcón SH, Olivieri AC, Sanz D, Claramunt RM, Elguero J (2004) Substituent and solvent effects on the proton transfer equilibrium in anils and azo derivatives of naphthol. Multinuclear NMR study and theoretical calculations. J Mol Struct 705:1–9
Hansen PE, Filarowski A (2004) Investigations of intramolecular hydrogen bond by nitrogen magnetic resonance methods. J Mol Struct 707:69–75
Ibañez GA, Escandar GM, Olivieri AC (2002) Proton transfer and coordination properties of aromatic α-hydroxy hydrazones. J Mol Struct 605:17–26
Hansen PE, Kamounah FS, Zhiryakova D, Manolova Y, Antonov L (2014) 1,1′,1′′-(2,4,6-Trihydroxybenzene-1,3,5-triyl)triethanone tautomerism revisited. Tetrahedron Lett 55:354–357
Albayrak C, Kaştaş G, Odabaşoğlu M, Frank R (2013) The prototropic tautomerism and substituent effect through strong electron-withdrawing group in (E)-5-(diethylamino)-2-(3-nitrophenylimino)methyl] phenol. Spectrochim Acta A 114:205–213
Houjou H, Shingai H, Yagi K, Yoshikawa I, Araki K (2013) Mutual interference between intramolecular proton transfer sites through the adjoining π-conjugated system in Schiff bases of double-headed, fused salicylaldehydes. J Org Chem 78:9021–9031
Schilf W, Szady-Chelmieniecka A, Grech E, Przybylski P, Brzezinski B (2002) Spectroscopic studies of new Schiff and Schiff–Mannich bases of ortho-derivatives of 4-bromophenol. J Mol Struct 643:115–121
Moghadam AJ, Omidyan R, Mirkhani V, Azimi G (2013) Theoretical investigation of excited state proton transfer process in the N-salicylidene-2-bromoethylamine. J Phys Chem A 117:718–725
Ortiz-Sánchez JM, Gelabert R, Moreno M, Lluch JM (2006) Theoretical study on the excited-state intramolecular proton transfer in the aromatic Schiff base salicylidene methylamine: an electronic structure and quantum dynamical approach. J Phys Chem A 110:4649–4656
Wojciechowski G, Przybylski P, Schilf W, Kamienski B, Brzezinski B (2003) Spectroscopic studies of Schiff bases of 2,2′-dihydroxybiphenyl-3-carbaldehyde and para substituted anilines. J Mol Struct 649:197–205
Mandal A, Fitzmaurice D, Waghorne E, Koll A, Filarowski A, Quinn S, Mukherjee S (2004) Proton transfer reaction of a new orthohydroxy Schiff base at room temperature and 77 K. Spectrochim Acta A 60:805–813
Golubev NS, Smirnov SN, Tolstoy PM, Sharif S, Toney MD, Denisov GS (2007) Observation by NMR of the tautomerism of an intramolecular OHOHN-charge relay chain in a model Schiff base. J Mol Struct 844–845:319–327
Schilf W, Bloxsidge JP, Jones JR, Lu S-Y (2004) Investigations of intramolecular hydrogen bonding in three types of Schiff bases by 2H and 3H NMR isotope effects. Magn Reson Chem 42:556–560
Schilf W (2004) Intramolecular hydrogen bonds in some 1,3bis(arylimino)isoindolines. J Mol Struct 689:245–249
Enchev V, Ugrinov A, Neykov GD (2000) Intramolecular proton transfer reactions in internally hydrogen-bonded Schiff bases: ab initio and semiempirical study. J Mol Struct 530:223–235
Sharif S, Powell DR, Schagen D, Steiner T, Toney MD, Fogle E, Limbach H-H (2006) X-ray crystallographic structures of enamine and amine Schiff bases of pyridoxal and its 1:1 hydrogen-bonded complexes with benzoic acid derivatives: evidence for coupled inter- and intramolecular proton transfer. Acta Crystallogr B 62:480–487
Rozwadowski Z, Ambroziak K, Szypa M, Jagodzinska E, Spychaj S, Schilf W, Kamienski B (2005) The 15N and 13C NMR study of Schiff bases of amino acids and their lithium salts in solid state and DMSO solution. J Mol Struct 734:137–142
Tezer N, Karakus N (2009) Theoretical study on the ground state intramolecular proton transfer (IPT) and solvation effect in two Schiff bases formed by 2-aminopyridine with 2-hydroxy-1-naphthaldehyde and 2-hydroxy salicylaldehyde. J Mol Model 15:223–232
Rozwadowski Z, Schilf W, Kamienski B (2005) Solid-state NMR study of Schiff base derivatives of 2- hydroxynaphthaldehyde. Deuterium isotope on 15N chemical shifts in the solid state. Magn Reson Chem 43:573–577
Ziółek M, Burdziński G, Karolczak J (2009) Influence of intermolecular hydrogen bonding on the photochromic cycle of the aromatic schiff base N,N′-bis(salicylidene)-p-phenylenediamine in solution. J Phys Chem A 113:2854–2864
Nedeltcheva D, Kamounah FS, Mirolo L, Fromm KM, Antonov L (2009) Solid state tautomerism in 2-((phenylimino)methyl)naphthalene-1-ol. Dyes Pigments 83:121–126
Wang Q, Gao F, Li H, Zhang S (2010) Photoinduced excited state intramolecular proton transfer of new schiff base derivatives with extended conjugated chromophores: a comprehensive theoretical survey. Chin J Chem 28:901–910
Albayrak C, Frank R (2010) Spectroscopic, molecular structure characterizations and quantum chemical computational studies of (E)-5-(diethylamino)-2-[(2-fluorophenylimino)methyl] phenol. J Mol Struct 984:214–220
Demircioğlu Z, Albayrak Ç, Büyükgüngör O (2014) Experimental (X-ray, FT-IR and UV–Vis spectra) and theoretical methods (DFT study) of (E)-3-methoxy-2-[(p-tolylimino)methyl]phenol. Spectrochim Acta A 128:748–758
Ali Beyramabadi S, Morsali A (2011) Intramolecular proton transfer of 2-[(2,4-dimethylphenyl)iminomethyl]-3,5-dimethoxyphenol schiff-base ligand: a density functional theory (DFT) study. Int J Phys Sci 6:1780–1788
Dhanunjayarao K, Mukundam V, Ramesh M, Venkatasubbaiah K (2014) Synthesis and optical properties of salicylaldimine-based diboron complexes. Eur J Inorg Chem 3:539–545
Sharif S, Denisov GS, Toney MD, Limbach H-H (2006) NMR studies of solvent-assisted proton transfer in a biologically relevant Schiff base: toward a distinction of geometric and equilibrium H-bond isotope effects. J Am Chem Soc 128:3375–3387
Pizzala H, Carles M, Stone WEE, Thevand A (2000) Tautomeric polymorphism in salicylideneamine derivatives: an X-ray diffraction and solid-state NMR study. J Chem Soc Perkin Trans 2:935–939
Galic N, Cimerman Z, Tomisic V (1997) Tautomeric and protonation equilibria of Schiff bases of salicylaldehyde with aminopyridines. Anal Chim Acta 343:135–143
Przybylski P, Schroeder G, Brzezinski B (2002) The Schiff base of gossypol with 2-(aminomethyl)-15-crown-5 complexes with monovalent cations studied by MS, 1H NMR, FT-IR and PM5 semiempirical methods. Phys Chem Chem Phys 4:6137–6143
Jayabharathi J, Thanikachalam V, Venkatesh Perumal M, Srinivasan N (2011) A physiochemical study of azo dyes: DFT based ESIPT process. Spectrochim Acta A 83:200–206
Przybylski P, Ratajczak-Sitarz M, Katrusiak A, Wojciechowski G, Schilf W, Brzezinski B (2003) Crystal structure of Schiff base derivative of gossypol with 3,6,9-trioxa-decylamine. J Mol Struct 655:293–300
Houjou H, Motoyama T, Banno S, Yoshikawa I, Araki K (2009) Experimental and theoretical studies on constitutional isomers of 2,6-dihydroxynaphthalene carbaldehydes: effects of resonance-assisted hydrogen bonding on the electronic absorption spectra. J Org Chem 74:520–529
Kakanejadifard A, Azarbani F, Zabardasti A, Rezayat A, Ghasemian M, Kakanejadifard S (2013) Spectroscopic and solvatochromism studies along with antioxidant and antibacterial activities investigation of 2-((2-mercaptophenyl-imino)methyl)-4-(phenyldiazenyl)phenol. Spectrochim Acta A 114:404–409
Hayvali Z, Ünver H, Svoboda I (2010) Synthesis, spectroscopic, spectrophotometric and crystallographic investigations of 4-{[(1E)-(3,4-dimethoxyphenyl)methylene]amino}-1,5-dimethyl-2-phenyl-1,2-dihydro-3H-pyrazol-3-one and 4-{[(1E)-(2-hydroxy-5-methoxyphenyl) methylene]amino}-1,5-dimethyl-2-phenyl-1,2-dihydro-3H-pyrazol-3-one. Acta Chim Slov 57:643–650
Przybylski P, Schilf W, Kamieński B, Brzezinski B, Bartl F (2008) CP/MAS spectroscopy in the determination of the tautomeric forms of gossypol, its Schiff bases and hydrazones in the solid state. Magn Reson Chem 46:534–544
Przybylski P, Bejcar G, Huczyński A, Schroeder G, Brzezinski B, Bartl F (2006) 1H- and 13C-NMR, FTIR, UV–VIS, ESI–MS, and PM5 studies as well as emission properties of a new Schiff base of gossypol with 5-methoxytryptamine and a new hydrazone of gossypol with dansylhydrazine. Biopolymers 82:521–535
Xu H, Tao X, Li Y, Shen Y, Wei Y (2012) Synthesis, characterization and metal ion-sensing properties of two Schiff base derivatives. Spectrochim Acta A 91:375–382
Tao T, Xu F, Chen X-C, Liu Q-Q, Huang W, You X-Z (2012) Comparisons between azo dyes and Schiff bases having the same benzothiazole/phenol skeleton: syntheses, crystal structures and spectroscopic properties. Dyes Pigments 92:916–922
Chan-Huot M, Sharif S, Tolstoy PM, Toney MD, Limbach H-H (2010) NMR studies of the stability, protonation states, and tautomerism of 13C- and 15N-labeled aldimines of the coenzyme pyridoxal 5′-phosphate in water. Biochemistry 49:10818–10830
Krygowski TM, Wozniak K, Anulewicz R, Pawlak D, Kołodziejski W, Grech E, Szady A (1997) Through-resonance assisted ionic hydrogen bonding in 5-nitro-N-salicylideneethylamine. J Phys Chem 101:9399–9404
Zarycz N, Aucar GA (2008) Theoretical NMR spectroscopic analysis of the intramolecular proton transfer mechanism in ortho-hydroxyaryl (un-)substitued Schiff bases. J Phys Chem A 112:8767–8774
Mukhopadhyay M, Banerjee D, Koll A, Filarowski A, Mukherjee S (2005) Proton transfer reaction of a new orthohydroxy Schiff base in some protic and aprotic solvents at room temperature and 77 K. Spectrochim Acta A 62:126–131
Hansen PE (2007) Isotope effect on chemical shifts in hydrogen-bonded systems. J Label Compd Radiopharm 50:967–981
Mohammadi A, Safarnejad M (2014) Synthesis, structural characterization and tautomeric properties of some novel bis-azo dyes derived from 5-arylidene-2,4-thiazolidinone. Spectrochim Acta A 126:105–111
Przybylski P, Pyta K, Ratajczak-Sitarz M, Katrusiak A, Brzezinski B (2008) X-ray, FT-IR, ESI MS and PM5 studies of Schiff base of gossypol with allylamine and its complexes with alkali metal cations and perchlorate anion. Struct Chem 19:983–995
Jakusová K, Donovalová J, Cigáň M, Gáplovský M, Garaj V, Gáplovský A (2014) Isatinphenylsemicarbazones as efficient colorimetric sensors for fluoride and acetate anions—anions induce tautomerism. Spectrochim Acta A 123:421–429
Przybylski P, Pyta K, Stefańska J, Ratajczak-Sitarz M, Katrusiak A, Huczyński A, Brzezinski B (2009) Synthesis, crystal structures and antibacterial activity studies of aza-derivatives of phytoalexin from cotton plant-gossypol. Eur J Med Chem 44:4393–4403
Przybylski P, Pyta K, Remlein-Starosta D, Schroeder G, Brzezinski B, Bartl F (2009) Antifungal activity of alkyl and heterocyclic aza-derivatives of gossypol as well as their complexes with NaClO4 against Fusarium oxysporum f. sp. lupini. Bioorg Med Chem Lett 19:1996–2000
Pyta K, Przybylski P, Huczyński A, Hoser A, Woźniak K, Schilf W, Kamieński B, Grech E, Brzezinski B (2010) X-ray, spectroscopic and computational studies of the tautomeric structure of a new hydrazone of 5-nitrosalicylaldehyde with indole-3-acetic hydrazide. J Mol Struct 970:147–154
Pyta K, Przybylski P, Schilf W, Kołodziej B, Szady-Chełmieniecka A, Grech E, Brzezinski B (2010) Spectroscopic and theoretical studies of the protonation of N-(5-nitrosalicylidene)-ethylamine. J Mol Struct 967:140–146
Przybylski P, Schroeder G, Brzezinski B (2004) The Schiff base of gossypol with 2-(aminomethyl)-18-crown-6 complexes and H+, Li+, Na+, K+, Rb+, Cs+ cations studied by ESI MS, 1H NMR, FT-IR and PM5 semiempirical methods. J Mol Struct 699:65–77
Przybylski P, Ilkevych N, Schroeder G, Brzezinski B, Bartl F (2004) Schiff base of gossypol with 3,6,9-trioxa-decylamine complexes, with monovalent cations studied by mass spectrometry, 1H-NMR, FTIR, and PM5 semiempirical methods. Biopolymers 73:470–483
Scigress Explorer FJ 2.5 (EU 3.1.6), CAChe 7.7.0 (2008–2014) UserGuide of PM6 method. Beaverton, OR
Stewart JJP (2007) Optimization of parameters for semiempirical methods V: modification of NDDO approximations and application to 70 elements. J Mol Model 13:1173–1213
Hoser AA, Schilf W, Szady-Chełmieniecka A, Kołodziej B, Kamieński B, Grech E, Woźniak K (2012) On the different coordination of NiII, ZnII and CdII cations in their model Schiff base complexes—single crystal X-ray and solid state NMR studies. Polyhedron 31:241–248
Claramunt RM, Lopez C, Santa-Maria MD, Sanz D, Elguero J (2006) The use of NMR spectroscopy to study tautomerism. Prog Nucl Magn Reson Spectrosc 49:169–206
Sashidhara KV, Rosaiah JN, Narender T (2007) Highly efficient and regioselective synthesis of keto-enamine Schiff bases of 7-hydroxy-4-methyl-2-oxo-2H-benzo[h]chromene-8,10-dicarbaldehyde and 1-hydroxynaphthalene-2,4-dicarbaldehyde. Tetrahedron Lett 48:1699–1702
Montalvo-Gonzales R, Ariza-Castolo A (2003) Molecular structure of di-aryl-aldimines by multinuclear magnetic resonance and X-ray diffraction. J Mol Struct 665:375–389
Przybylski P, Brzezinski B (2002) Spectroscopic studies and PM3 semiempirical calculations of Schiff bases of gossypol with l-amino acid methyl esters. Biopolymers 67:61–69
Przybylski P, Bartl F, Brzezinski B (2002) Spectroscopic studies and PM5 semiempirical calculations of new Schiff bases of gossypol with polyoxaalkylamines. Biopolymers 65:111–120
Sobczyk L, Grabowski SJ, Krygowski TM (2005) Interrelation between H-bond and Pi-electron delocalization. Chem Rev 105:3513–3560
Acknowledgments
PP, KP and KK wish to thank for the project financial support of the Polish National Science Center (NCN), Grant decision number DEC-2012/05/E/ST5/03792.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Pyta, K., Przybylski, P., Klich, K. et al. Impact of metal cation complexation and protonation on tautomeric and resonance forms of the oxaalkyl Schiff bases derived from 5-substituted salicylaldehyde and 2-hydroxy-1-naphthlaldehyde. Struct Chem 25, 1733–1746 (2014). https://doi.org/10.1007/s11224-014-0447-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11224-014-0447-x