Abstract
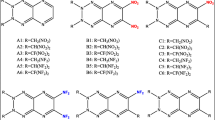
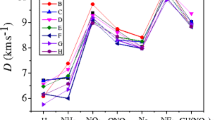
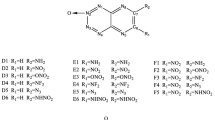
Density functional theory calculations were performed to find the relationships between the structures and performance of a series of 1,2,4,5-tetrazine-based energetic derivatives. The isodesmic reaction method was employed to estimate the heats of formation (HOFs). The result shows that the azo or azoxy group is one of the most energetic functional groups known and its substitution can drastically increase HOFs of a molecule. The detonation properties were also evaluated by the Kamlet–Jacobs equations based on the theoretical densities and HOFs. Results show that NO2 group is an effective substituent for enhancing the detonation performance. There exist better correlations between OB and detonation velocities and OB and detonation pressures. The energy gaps between the HOMO and LUMO of the studied compounds are also investigated, and from the data we estimated the relative thermal stability ordering of the title compounds.



Similar content being viewed by others
References
Lempert DB, Nechiporenko GN, Soglasnova SI (2009) Combust Explo Shock 45:160 (Engl. Transl.)
Saracoglu N (2007) Tetrahedron 63:4199
Huynh My Hang V, Hiskey Michael A, Chavez David E, Naud Darren L, Gilardi Richard D (2005) J Am Chem Soc 127:12537
Godovikova TI, Rakitin OA, Golova SP, Vozchikova SA, Khmelnitskii LI (1993) Mendeleev Commun 3:209
Sheremetev AB, Makhova NN, Friedrichsen W (2001) Advances in heterocyclic chemistry. Academic Press, New York
Britton D, Noland WE, Clark CM (2008) Acta Crystallogr C64:o187
Frisch MJ, Trucks GW, Schlegel HB, Scuseria GE, Robb MA, Cheeseman JR, Zakrzewski VG, Montgomery JA, Stratmann RE, Burant JC, Dapprich S, Millam JM, Daniels AD, Kudin KN, Strain MC, Farkas O, Tomasi J, Barone V, Cossi M, Cammi R, Mennucci B, Pomelli C, Adamo C, Clifford S, Ochterski J, Petersson GA, Ayala PY, Cui Q, Morokuma K, Malick DK, Rabuck AD, Raghavachari K, Foresman JB, Cioslowski J, Ortiz JV, Baboul AG, Stefanov BB, Liu G, Liashenko A, Piskorz P, Komaromi I, Gomperts R, Martin RL, Fox DJ, Keith T, Al-Laham MA, Peng CY, Nanayakkara A, Gonzalez C, Challacombe M, Gill PMW, Johnson B, Chen W, Wong MW, Andres JL, Gonzalez C, Head-Gordon M, Replogle ES, Pople JA (2003) Gaussian 03, Revision B.02. Gaussian Inc., Pittsburgh PA
Selmi M, Tomasi J (1995) J Phys Chem 99:5894
Chen ZX, Xiao JM, Xiao HM, Chiu YN (1999) J Phys Chem A 103:8062
Xiao HM, Chen ZX (2000) The modern theory for tetrazole chemistry, 1st edn. Science Press, Beijing
Chen PC, Chieh YC, Tzeng SC (2003) J Mol Struct 634:215
Jursic BS (2000) J Mol Struct 499:137
Dill JD, Greenberg A, Liebman JF (1979) J Am Chem Soc 101:6814
Hehre WJ, Radom L, Schleyer PVR, Pople JA (1986) Ab initio molecular orbital theory. Wiley, New York
Terrier F, Sebban M, Goumont R, Halle JC, Moutiers G, Cangelosi I, Buncel E (2000) J Org Chem 65:7391
Pasinszki T, Westwood NPC (1995) J Am Chem Soc 117:8425
Kamlet MJ, Jacobs SJ (1968) J Chem Phys 48:23
Zhang XH, Yun ZH (1989) Explosive chemistry. National Defense Industry Press, Beijing
Politzer P, Martinez J, Murray JS, Concha MC (2009) Mol Phys 107:2095
Wei T, Zhang JJ, Zhu WH, Zhang XW, Xiao HM (2010) J Mol Struct 956:55
Lide DR (2004) Handbook of chemistry and physics, 84th edn. CRC Press LLC, Boca Raton
Bai L, Hu Y, Hu R-Z, Song J-R, Ma H-X (2010) Chin J Explos Propellants 33:19
Zhang J-G, Niu X-Q, Zhang S-W, Zhang T-L, Huang H-S, Zhou Z-N (2011) Comput Theor Chem 964:291
Li X-H, Zhang R-Z, Zhang X-Z (2010) J Hazard Mater 183:622
Talawar MB, Sivabalan R, Mukundan T, Muthurajan H, Sikder AK, Gandhe BR, Rao AS (2009) J Hazard Mater 161:589
Gilardi R, Flippen-Anderson JL, Evans R (2002) Acta Crystallogr E58:o972
Shi MD (1992) Chin J Explos Propellants 1:19
Willer RL (1983) Propellants Explos Pyrotech 8:65
Fried LE, Manaa MR, Pagoria PF, Simpson RL (2001) Annu Rev Mater Res 31:291
Clare B (1994) Theor Chim Acta 87:415
Da Silva ABF (1985) M.Sc. Thesis, Universidade deSÄao Paulo, Brazil
Lewis DFV, Ioannides C, Parke DV (1994) Xenobiotica 24:401
Zhou Z, Parr RG (1990) J Am Chem Soc 112:5720
Acknowledgments
The authors gratefully thank the National Natural Science Foundation of China (Grant 10774039) and the key project of Henan Educational Committee (No. 12A140004), Henan University of Science and Technology for Young Scholars (No.2009QN0032) for their support of this study.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Li, X., Zhang, RZ. & Zhang, XZ. Theoretical studies of 1,2,4,5-tetrazine-based energetic nitrogen-rich compounds. Struct Chem 24, 393–400 (2013). https://doi.org/10.1007/s11224-012-0089-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11224-012-0089-9