Abstract
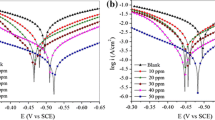
New Mannich base 1-((pyridin-2-ylamino)(pyridin-4-yl)methyl)pyrrolidine-2,5-dione (SAP) was synthesized, characterized, and investigated as a corrosion inhibitor on mild steel (MS) in 1.0 M HCl using weight loss, electrochemical experiments, and theoretical calculations. Impedance spectra validated the decrease in the double-layer capacitance (C dl) and increase in the charge-transfer resistance (R CT) of the corrosion process due to the addition of the inhibitor. Tafel polarization studies proved that the inhibitor has a mixed type inhibitive nature with predominant control of cathodic reaction. A maximum inhibition efficiency of 88.7% from weight loss measurements and above 94.5% from electrochemical analysis were observed in the presence of 200 ppm of SAP, and the adsorption of SAP on MS surface in 1.0 M HCl was found to follow Langmuir’s adsorption isotherm. The calculated values of thermodynamic parameters predict the adsorption of SAP molecules on the MS surface was physical adsorption. FTIR, SEM–EDX, and AFM were used to characterize the surface morphology of the mild steel samples in the presence and absence of inhibitor. Density functional theory based quantum chemical calculations also fortifies the experimental inferences revealing SAP as good inhibitor.
Graphical Abstract













Similar content being viewed by others
References
A. Döner, G. Kardaş, Corros. Sci. 53, 4223 (2011)
D. Gopi, K.M. Govindaraju, V.C.A. Prakash, V. Manivannan, L. Kavitha, J. Appl. Electrochem. 39, 269 (2009)
M.K. Pavithra, T.V. Venkatesha, M.K.P. Kumar, K. Manjunatha, Res. Chem. Intermed. 41, 7163 (2015)
M. Yadav, D. Behera, S. Kumar, R.R. Sinha, Ind. Eng. Chem. Res. 52, 6318 (2013)
H.B. Ouici, O. Benali, Y. Harek, L. Larabi, B. Hammouti, A. Guendouzi, Res. Chem. Intermed. 39, 3089 (2013)
J. Aljourani, K. Raeissi, M.A. Golozar, Corros. Sci. 51, 1836 (2009)
N. Raghavendra, J.I. Bhat, Res. Chem. Intermed. 42, 6351 (2016)
V.S. Sastri, Green Corrosion Inhibitors: Theory and Practice (Wiley, Hoboken, 2011), p. 144
H. Ashassi-Sorkhabi, M. Es’haghi, J. Solid State Electrochem. 13, 1297 (2009)
A. Dutta, S.K. Saha, P. Banerjee, D. Sukul, Corros. Sci. 98, 541 (2015)
R. Solmaz, E.A. Şahin, A. Döner, G. Kardaş, Corros. Sci. 53, 3231 (2011)
M. Yadav, L. Gope, T.K. Sarkar, Res. Chem. Intermed. 42, 2641 (2016)
M.G. Hosseini, H. Khalilpur, S. Ershad, L. Saghatforoush, J. Appl. Electrochem. 40, 215 (2010)
A.I. Adawy, M.A. Abbas, K. Zakaria, Res. Chem. Intermed. 42, 3385 (2016)
S.K. Saha, A. Dutta, P. Ghosh, D. Sukul, P. Banerjee, Phys. Chem. Chem. Phys. 18, 17898 (2016)
M. Muralisankar, R. Sreedharan, S. Sujith, N.S.P. Bhuvanesh, A. Sreekanth, J. Alloys Compd. 695, 171 (2017)
Y.I. Kuznetsov, Organic Inhibitors of Corrosion of Metals (Springer, Plenum Press, New York, 1996), p. 253
H.L.Y. Sin, M. Umeda, S. Shironita, A.A. Rahim, B. Saad, Res. Chem. Intermed. 43, 1919 (2017)
D. Turcio-Ortega, T. Pandiyan, J. Cruz, E. Garcia-Ochoa, J. Phys. Chem. C 111, 9853 (2007)
S.N. Pandeya, D. Sriram, G. Nath, E. DeClercq, Eur. J. Pharm. Sci. 9, 25 (1999)
M.A. Ali, M. Shaharyar, Bioorg. Med. Chem. Lett. 17, 3314 (2007)
M. Tramontini, L. Angiolini, Mannich Bases: Chemistry and Uses (CRC Press, London, 1994), pp. 250–259
C.A. Miller, L.M. Long, J. Am. Chem. Soc. 73, 4895 (1951)
G. Rankin, K. Cressey-Veneziano, R. Wang, P. Brown, J. Appl. Toxicol. 6, 349 (1986)
A.E. Tamer, C. Detellier, J. Colloid Interface Sci. 323, 338 (2008)
O. Araki, A. Andoh, Y. Fujiyama, K. Hata, J. Makino, T. Okuno, F. Nakanura, T. Bamba, J. Chromatogr. B 753, 209 (2001)
R. Ewis, Hawley’s Condensed Chemical Dictionary, 14th edn. (Wiley, New York, 2001), p. 58
C.P. Huttrer, C. Djerassi, W.L. Beears, R.L. Mayer, C.R. Scholz, J. Am. Chem. Soc. 10, 1999 (1946)
C.P. Huttrer, C. Djerassi, C.R. Scholz, US Patent 2 406, (1946) p. 594
R.J. Horclois, US Patent 2 502, (1950) p. 151
P. Thiraviyam, K. Kannan, J. Iran. Chem. Soc. 9, 911 (2012)
I. Ahamad, R. Prasad, M.A. Quraishi, Corros. Sci. 52, 1472 (2010)
D. Karthik, D. Tamilvendan, G.V. Prabhu, J. Saudi Chem. Soc. 18, 835 (2014)
M. Jeeva, G.V. Prabhu, M.S. Boobalan, C.M. Rajesh, J. Phys. Chem. C 119, 22025 (2015)
M. Yadav, U. Sharma, P.N. Yadav, Egypt. J. Pet. 22, 335 (2013)
H.B. Ouici, O. Benali, A. Guendouzi, Res. Chem. Intermed. 42, 7085 (2016)
I. Belfilali, A. Chetouani, B. Hammouti, S. Louhibi, A. Aouniti, S.S. Al-Deyab, Res. Chem. Intermed. 40, 1069 (2014)
L. Li, X. Zhang, S. Gong, H. Zhao, Y. Bai, Q. Li, L. Ji, Corros. Sci. 99, 76 (2015)
B. Gómez, N.V. Likhanova, M.A. Domínguez-Aguilar, R. Martínez-Palou, A. Vela, J.L. Gázquez, J. Phys. Chem. B 110, 8928 (2006)
P. Mourya, P. Singh, R.B. Rastogi, M.M. Singh, Appl. Surf. Sci. 380, 141 (2016)
I.B. Obot, Z.M. Gasem, Corros. Sci. 83, 359 (2014)
X. Li, S. Deng, H. Fu, T. Li, Electrochim. Acta 54, 4089 (2009)
A. Döner, R. Solmaz, M. Özcan, G. Kardaş, Corros. Sci. 53, 2902 (2011)
A.D. Raj, M. Jeeva, M. Shankar, R. Purusothaman, G.V. Prabhu, I.V. Potheher, Phys. B 501, 45 (2016)
D. Tamilvendan, S. Rajeswari, S. Ilavenil, K. Chakkaravarthy, G.V. Prabhu, Med. Chem. Res. 21, 4129 (2012)
M. Jeeva, G.V. Prabhu, C.M. Rajesh, J. Mater. Sci. (2017). doi:10.1007/s10853-017-1401-2
S.L.A. Kumar, M. Gopiraman, M.S. Kumar, A. Sreekanth, Ind. Eng. Chem. Res. 50, 7824 (2011)
S. Belkaid, K. Tebbji, A. Mansri, A. Chetouani, B. Hammouti, Res. Chem. Intermed. 38, 2309 (2012)
T. Zhang, S. Cao, H. Quan, Z. Huang, S. Xu, Res. Chem. Intermed. 41, 2709 (2015)
M.J.T. Frisch, G.W. Frisch, H.B. Schlegel, G.E. Scuseria, M.A. Robb, J.R. Cheeseman, G. Scalmani, V. Barone, B. Mennucci, G.A. Petersson, H. Nakatsuji, M. Caricato, X. Li, H.P. Hratchian, A.F. Izmaylov, J. Bloino, G. Zheng, J.L. Sonnenberg, M. Hada, M. Ehara, K. Toyota, R. Fukuda, J. Hasegawa, M. Ishida, T. Nakajima, Y. Honda, O. Kitao, H. Nakai, T. Vreven, J.A. Montgomery Jr., J.E. Peralta, F. Ogliaro, M. Bearpark, J.J. Heyd, E. Brothers, K.N. Kudin, V.N. Staroverov, R. Kobayashi, J. Normand, K. Raghavachari, A. Rendell, J.C. Burant, S.S. Iyengar, J. Tomasi, M. Cossi, N. Rega, N.J. Millam, M. Klene, J.E. Knox, J.B. Cross, V. Bakken, C. Adamo, J. Jaramillo, R. Gomperts, R.E. Stratmann, O. Yazyev, A.J. Austin, R. Cammi, C. Pomelli, J.W. Ochterski, R.L. Martin, K. Morokuma, V.G. Zakrzewski, G.A. Voth, P. Salvador, J.J. Dannenberg, S. Dapprich, A.D. Daniels, O. Farkas, J.B. Foresman, J.V. Ortiz, J. Cioslowski, D.J. Fox, Gaussian 09, Revision B.01 (Gaussian Inc., Wallingford, 2009)
J. Tomasi, B. Mennucci, R. Cammi, Chem. Rev. 105, 2999 (2005)
A.S. Fouda, T.F. Alsawy, E.S. Ahmed, B.S. Abou-elmagd, Res. Chem. Intermed. 39, 2641 (2013)
E.G. Lewars, Computational Chemistry: Introduction to the Theory and Applications of Molecular and Quantum Mechanics (Springer, New York, 2003), p. 425
L. Guo, X. Ren, Y. Zhou, S. Xu, Y. Gong, S. Zhang, Arab. J. Chem. (2015). doi:10.1016/j.arabjc.2015.01.005
M.S. Boobalan, D. Tamilvendan, M. Amaladasan, S. Ramalingam, G.V. Prabhu, M. Bououdina, J. Mol. Struct. 1081, 159 (2015)
R.G. Parr, L.V. Szentpály, S. Liu, J. Am. Chem. Soc. 121, 1922 (1999)
J. Padmanabhan, R. Parthasarathi, U. Sarkar, V. Subramanian, P.K. Chattaraj, Chem. Phys. Lett. 383, 122 (2004)
A.D. Becke, K.E. Edgecombe, J. Chem. Phys. 92, 5397 (1990)
T. Ghazouani, D.B. Hmamou, E. Meddeb, R. Salghi, O. Benali, H. Bouya, B. Hammouti, S. Fattouch, Res. Chem. Intermed. 41, 7463 (2015)
S.M. Tawfik, A.A. Abd-Elaal, I. Aiad, Res. Chem. Intermed. 42, 1101 (2016)
C.B.P. Kumar, K.N. Mohana, H.B. Muralidhara, Ionics (2014). doi:10.1007/s11581-014-1178-0
D.K. Verma, F. Khan, Res. Chem. Intermed. 42, 3489 (2016)
P.V. Raphael, K.J. Thomas, K.S. Shaju, A. Paul, Res. Chem. Intermed. 40, 2689 (2014)
M. Hoseinpoor, A. Davoodi, Res. Chem. Intermed. 41, 4255 (2015)
B.D. Mert, A.O. Yüce, G. Kardaş, B. Yazıcı, Corros. Sci. 85, 287 (2014)
V. Rajeswari, D. Kesavan, M. Gopiraman, P. Viswanathamurthi, Carbohydr. Polym. 95, 288 (2013)
D.G. Lewis, V.C. Farmer, Clay Miner. 21, 93 (1986)
A. Kokalj, S. Peljhan, Langmuir 26, 14582 (2010)
L.O. Olasunkanmi, I.B. Obot, M.M. Kabanda, E.E. Ebenso, J. Phys. Chem. C 119, 16004 (2015)
V. Pilepić, S. Uršić, J. Mol. Struct. (Theochem) 538, 41 (2001)
Acknowledgements
The author M. Jeeva wishes to acknowledge MHRD, Government of India, for a research fellowship. We thank DST-FIST for characterization facilities, Department of Chemistry, National Institute of Technology, Tiruchirappalli, India. Authors are highly thankful to Department of Chemistry, Gandhigram Rural Institute, Dindigul, Tamilnadu, India for SEM−EDX analysis, and STIC, Cochin University, Kerala, India for Elemental analysis.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Jeeva, M., Boobalan, M.s. & Prabhu, G.V. Adsorption and anticorrosion behavior of 1-((pyridin-2-ylamino)(pyridin-4-yl)methyl)pyrrolidine-2,5-dione on mild steel surface in hydrochloric acid solution. Res Chem Intermed 44, 425–454 (2018). https://doi.org/10.1007/s11164-017-3112-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11164-017-3112-3