Abstract
Purpose
This study was to conduct the psychometric validation of the patient and parent versions of the Hunter Syndrome-Functional Outcomes for Clinical Understanding Scale (HS-FOCUS).
Methods
Data collected in a 53-week placebo-controlled multinational trial were used to evaluate item performance and reliability, validity, and ability to detect change of the six HS-FOCUS function domains.
Results
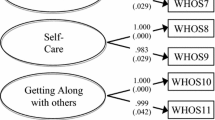
HS-FOCUS was completed by 49 patients above 12 years old and 84 parents. Floor effects and high average inter-item correlations suggested that some items were less informative or redundant. For both patients and parents, the internal consistency and test–retest reliability met the >0.70 criteria for all domains except for the breathing, sleeping, and schooling/work in patients. Construct validity showed moderate to high correlations with CHAQ, CHQ, and HUI3 in activity-related concepts. Significant differences in domain scores were found in most domains among severity in disability measured by CHAQ DIS. Significant differences in HS-FOCUS change scores were found in patients whose CHAQ DIS score also changed.
Conclusions
Psychometric validation of the HS-FOCUS demonstrates it is a reliable, valid, and responsive instrument that can be applied in clinical trials or disease registries. Findings on the individual item performance suggest some items could be removed without compromising its validity.
Similar content being viewed by others
Abbreviations
- 6MWT:
-
Six-minute walk test
- ANCOVA:
-
Analysis of covariance
- ANOVA:
-
Analysis of variance
- BP:
-
Bodily Pain
- CH:
-
Change in Health
- CHAQ:
-
Childhood Health Assessment Questionnaire
- CHQ:
-
Childhood Health Questionnaire
- CHQ-CF:
-
Child Health Questionnaire-Child Form
- CHQ-PF:
-
Child Health Questionnaire-Parent Form
- DIS:
-
Disability Index Score
- FA:
-
Family Activities
- FC:
-
Family Cohesion
- FEV:
-
Forced expiratory volume
- FIM:
-
Functional Independence Measure
- FVC:
-
Forced vital capacity
- GAG:
-
Glycosaminoglycans
- GBE:
-
Global Behavior
- GH:
-
General Health Perceptions
- HRQL:
-
Health-related quality of life
- HS:
-
Hunter syndrome
- HS-FOCUS:
-
Hunter Syndrome-Functional Outcomes for Clinical Understanding Scale
- HUI:
-
Health Utilities Index
- I2S:
-
Iduronate-2-sulfatase
- ICC:
-
Intraclass correlation coefficient
- MH:
-
Mental Health
- MPS II:
-
Mucopolysaccharidosis type II
- PE:
-
Parental Impact–Emotional
- PF:
-
Physical Functioning
- PODCI:
-
Pediatric Outcomes Data Collection Instrument
- PRO:
-
Patient-reported outcome
- PT:
-
Parental Impact–Time
- RB:
-
Role/Social–Behavioral
- RE:
-
Role/Social–Emotional
- REB:
-
Role/Social–Emotional and the Role/Social–Behavioral
- RP:
-
Role/Social–Physical
- SD:
-
Standard deviation
- SE:
-
Self Esteem
- WHO-ICIDH:
-
World Health Organization International Classification of Impairment, Disability and Handicap
References
Alcalde-Martin, C., Muro-Tudelilla, J. M., Cancho-Candela, R., Gutierrez-Solana, L. G., Pintos-Morell, G., Marti-Herrero, M., et al. (2010). First experience of enzyme replacement therapy with idursulfase in Spanish patients with Hunter syndrome under 5 years of age: Case observations from the Hunter Outcome Survey (HOS). European Journal of Medical Genetics, 53(6), 371–377.
Martin, R., Beck, M., Eng, C., Giugliani, R., Harmatz, P., Munoz, V., et al. (2008). Recognition and diagnosis of mucopolysaccharidosis II (Hunter syndrome). Pediatrics, 121(2), e377–e386.
Muenzer, J., Beck, M., Eng, C. M., Escolar, M. L., Giugliani, R., Guffon, N. H., et al. (2009). Multidisciplinary management of Hunter syndrome. Pediatrics, 124(6), e1228–e1239.
Young, I. D., & Harper, P. S. (1982). Mild form of Hunter’s syndrome: Clinical delineation based on 31 cases. Archives of Disease in Childhood, 57(11), 828–836.
Young, I. D., & Harper, P. S. (1983). The natural history of the severe form of Hunter’s syndrome: A study based on 52 cases. Developmental Medicine and Child Neurology, 25(4), 481–489.
Keilmann, A., Nakarat, T., Bruce, I. A., Molter, D., Malm, G., & on behalf of the HOS Investigators. Hearing loss in patients with mucopolysaccharidosis II: Data from HOS—the hunter outcome survey.
Pascolini, D., & Smith, A. (2009). Hearing impairment in 2008: A compilation of available epidemiological studies. International Journal of Audiology, 48(7), 473–485.
World Health Organization (WHO). (1999). WHO ear and hearing disorders survey protocol for a population-based survey of prevalence and causes of deafness and hearing impairment and other ear disorders. Retrieved April 14, 2011, from http://whqlibdoc.who.int/hq/1999/WHO_PBD_PDH_99.8(1).pdf.
Singh, G., Athreya, B. H., Fries, J. F., & Goldsmith, D. P. (1994). Measurement of health status in children with juvenile rheumatoid arthritis. Arthritis and Rheumatism, 37(12), 1761–1769.
Tran, K. T., Gold, K. F., Stephens, J. M., Kimura, A., & Muenzer, J. The development and validation of the hunter syndrome—functional outcomes for clinical understanding scale.
Abt. Associates. (2004). Clinical trials. Confirmatory validation study of the CHAQ. Video documentation of functional tasks, and development of a new supplement Questionnaire to assess functional status in hunter syndrome. Final Report.
Giannini, E. H., Ruperto, N., Ravelli, A., Lovell, D. J., Felson, D. T., & Martini, A. (1997). Preliminary definition of improvement in juvenile arthritis. Arthritis and Rheumatism, 40(7), 1202–1209.
Landgraf, J. M., Abetz, L. N., & Ware, J. E. (1999). The CHQ user’s manual. Boston: The Health Institute, New England Medical Center.
Valayannopoulos, V., de Blic, J., Mahlaoui, N., Stos, B., Jaubert, F., Bonnet, D., et al. (2010). Laronidase for cardiopulmonary disease in Hurler syndrome 12 years after bone marrow transplantation. Pediatrics, 126(5), e1242–e1247.
McGovern, M. M., Wasserstein, M. P., Giugliani, R., Bembi, B., Vanier, M. T., Mengel, E., et al. (2008). A prospective, cross-sectional survey study of the natural history of Niemann-Pick disease type B. Pediatrics, 122(2), e341–e349.
Horsman, J., Furlong, W., Feeny, D., & Torrance, G. (2003). The health utilities index (HUI): Concepts, measurement properties and applications. Health and Quality of Life Outcomes, 1, 54.
StataCorp. (2009). Stata statistical software: Release 11. College Station, TX: StataCorp LP.
Cronbach, L. (1951). Coefficient alpha and the internal structure of tests. Psychometrika, 16, 297–334.
Hays, R. D., & Revicki, D. A. (2005). Assessing reliability and validity of measurement in clinical trials. In P. Fayers & R. D. Hays (Eds.), Quality of life assessment in clinical trials: Methods and practice (2nd ed.). New York: Oxford University Press.
Nunnally, J. C., & Bernstein, I. H. (1994). Psychometric theory (3rd ed.). New York: McGraw-Hill.
Dempster, H., Porepa, M., Young, N., & Feldman, B. M. (2001). The clinical meaning of functional outcome scores in children with juvenile arthritis. Arthritis and Rheumatism, 44(8), 1768–1774.
Leidy, N. K., Revicki, D. A., & Geneste, B. (1999). Recommendations for evaluating the validity of quality of life claims for labeling and promotion. Value Health, 2(2), 113–127.
Rutten-van Molken, M., Roos, B., & Van Noord, J. A. (1999). An empirical comparison of the St George’s Respiratory Questionnaire (SGRQ) and the Chronic Respiratory Disease Questionnaire (CRQ) in a clinical trial setting. Thorax, 54(11), 995–1003.
Daltroy, L. H., Liang, M. H., Fossel, A. H., & Goldberg, M. J. (1998). The POSNA pediatric musculoskeletal functional health questionnaire: Report on reliability, validity, and sensitivity to change. Pediatric Outcomes Instrument Development Group. Pediatric Orthopaedic Society of North America. Journal of Pediatric Orthopedics, 18(5), 561–571.
Keith, R. A., Granger, C. V., Hamilton, B. B., & Sherwin, F. S. (1987). The functional independence measure: A new tool for rehabilitation. Advances in Clinical Rehabilitation, 1, 6–18.
Author information
Authors and Affiliations
Corresponding author
Additional information
Donald E. Stull was a United BioSource Corporation employee when this work was conducted.
Yvonne Jangelind was a Shire employee when this work was conducted.
Rights and permissions
About this article
Cite this article
Wiklund, I., Raluy-Callado, M., Stull, D.E. et al. The Hunter Syndrome-Functional Outcomes for Clinical Understanding Scale (HS-FOCUS) Questionnaire: evaluation of measurement properties. Qual Life Res 22, 875–884 (2013). https://doi.org/10.1007/s11136-012-0196-5
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11136-012-0196-5