Abstract
Aims
Glucose-6-phosphate dehydrogenase (G6PDH) has been reported to be involved in resistance to various environmental stresses. However, the role of G6PDH in aluminum (Al) toxicity remains unclear.
Methods
Physiological and biochemical methods together with histochemical analysis were used to investigate the participation of G6PDH in Al-induced inhibition of root growth.
Results
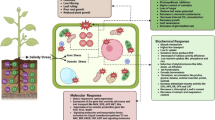
Exposure to high Al concentration caused a significant increase in the activities of total and cytosolic G6PDH in roots of soybean. Al-induced inhibition of root growth and oxidative stress were alleviated by a G6PDH inhibitor. Reactive oxygen species (ROS) accumulation in Al-treated root apexes could be abolished by a NADPH oxidase inhibitor. Furthermore, treatment with a G6PDH inhibitor reduced NADPH content and NADPH oxidase activity in Al-treated root apexes. Further investigation demonstrates that nitric oxide (NO) mediates Al-induced increase in cytosolic G6PDH activity by modulating the expression of genes encoding cytosolic G6PDH. In addition, nitrate reductase pathway is mainly responsible for Al-induced NO production in root apexes.
Conclusions
These results indicate that NADPH produced by NO-modulated cytosolic G6PDH in root apexes is responsible for ROS accumulation mediated by NADPH oxidase under Al stress, subsequently suffering from oxidative stress and thus causing the inhibition of root elongation.










Similar content being viewed by others
Abbreviations
- DAB:
-
Diaminobenzidine
- DPI:
-
Diphenylene iodonium
- G6PDH:
-
Glucose-6-phosphate dehydrogenase
- GN6P:
-
Glucosamine 6-phosphate
- L-NNA:
-
N ω-nitro-L-Arg
- MDA:
-
Malondialdehyde
- NBT:
-
Nitroblue tetrazolium
- NOS:
-
Nitric oxide synthase
- NR:
-
Nitrate reductase
- PTIO:
-
2-phenyl-4,4,5,5-tetramethyl-imidazoline-1-oxyl-3-oxide
- ROS:
-
Reactive oxygen species
- SNP:
-
Sodium nitroprusside
References
Asai S, Yoshioka M, Nomura H, Tone C, Nakajima K, Nakane E, Doke N, Yoshioka H (2011) A plastidic glucose-6-phosphate dehydrogenase is responsible for hypersensitive response cell death and reactive oxygen species production. J Gen Plant Pathol 77:152–162
Bowsher CG, Lacey AE, Hanke GT, Clarkson DT, Saker LR, Stulen I, Emes JM (2007) The effect of Glc6P uptake and its subsequent oxidation within pea root plastids on nitrite reduction and glutamate synthesis. J Exp Bot 58:1109–1118
Cardi M, Chibani K, Cafasso D, Rouhier N, Jacquot JP, Esposito S (2011) Abscisic acid effects on activity and expression of barley (Hordeum vulgare) plastidial glucose-6-phosphate dehydrogenase. J Exp Bot 62:4013–4023
Chen WW, Yang JL, Qin C, Jin CW, Mo JH, Ye T, Zheng SJ (2010) Nitric oxide acts downstream of auxin to trigger root ferric-chelate reductase activity in response to iron deficiency in Arabidopsis. Plant Physiol 154:810–819
Dal Santo S, Stampfl H, Krasensky J, Kempa S, Gibon Y, Petutschnig E, Rozhon W, Heuck A, Clausen T, Jonak C (2012) Stress-induced GSK3 regulates the redox stress response by phosphorylating glucose-6-phosphate dehydrogenase in Arabidopsis. Plant Cell 24:3380–3392
Esposito S, Guerriero G, Vona V, Di Martino RV, Carfagna S, Rigano C (2005) Glutamate synthase activities and protein changes in relation to nitrogen nutrition in barley: the dependence on different plastidial glucose-6P dehydrogenase isoforms. J Exp Bot 56:55–64
Gong H, Chen G, Li F, Wang X, Hu Y, Bi Y (2013) Involvement of G6PDH in heat stress tolerance in the calli from Przewalskiatangutica and Nicotianatabacum. Biol Plant 56:422–430
Hao F, Wang X, Chen J (2006) Involvement of plasma-membrane NADPH oxidase in nickel-induced oxidative stress in roots of wheat seedlings. Plant Sci 170:151–158
Hauschild R, von Schaewen A (2003) Differential regulation of glucose-6-phosphate dehydrogenase isoenzyme activities in potato. Plant Physiol 133:47–62
Hu Y, You J, Liang X (2015) Nitrate reductase-mediated nitric oxide production is involved in copper tolerance in shoots of hulless barley. Plant Cell Rep 34:367–379
Kochian LV (1995) Cellular mechanisms of aluminum toxicity and resistance in plants. Annu Rev Plant Physiol Plant Mol Biol 46:237–260
Lamattina L, Garcia-Mata C, Graziano M, Pagnussat G (2003) Nitric oxide: the versatility of an extensive signal molecule. Annu Rev Plant Biol 54:109–136
Lin A, Wang Y, Tang J, Xue P, Li C, Liu L, Hu B, Yang F, Loake GJ, Chu C (2012) Nitric oxide and protein S-nitrosylation are integral to hydrogen peroxide-induced leaf cell death in rice. Plant Physiol 158:451–464
Liu Y, Wu R, Wan Q, Xie G, Bi Y (2007) Glucose-6-phosphate dehydrogenase plays a pivotal role in nitric oxide-involved defense against oxidative stress under salt stress in red kidney bean roots. Plant Cell Physiol 48:511–522
Ma JF (2005) Plant root responses to three abundant soil minerals: silicon, aluminum and iron. Crit Rev Plant Sci 24:267–281
Mackintosh C, Douglas P, Lillo C (1995) Identification of a protein that inhibits the phosphorylated form of nitrate reductase from spinach (Spinacia oleracea) leaves. Plant Physiol 107:451–457
Maffei ME, Mithofer A, Arimura G, Uchtenhagen H, Bossi S, Bertea CM, Cucuzza LS, Novero M, Volpe V, Quadro S, Boland W (2006) Effects of feeding Spodoptera Littoralis on lima bean leaves. III. Membrane depolarization and involvement of hydrogen peroxide. Plant Physiol 140:1022–1035
Matsumura H, Miyachi S (1980) Cycling assay for nicotinamide adenine dinucleotides. Methods Enzymol 69:465–470
Neill SJ, Desikan R, Hancock JT (2003) Nitric oxide signaling in plants. New Phytol 159:11–35
Nemoto Y, Sasakuma T (2000) Specific expression of glucose-6-phosphate dehydrogenase (G6PDH) gene by salt stress in wheat (Triticum aestivum L.). Plant Sci 158:53–60
Scharte J, Schön H, Tjaden Z, Weis E, von Schaewen A (2009) Isoenzyme replacement of glucose-6-phosphate dehydrogenase in the cytosol improves stress tolerance in plants. Proc Natl Acad Sci U S A 106:8061–8066
Ślaski JJ, Zhang G, Basu U, Stephens JL, Taylor GJ (1996) Aluminum resistance in wheat (Tritucum aestivum) is associated with rapid, Al-induced changes in activities of glucose-6-phosphate dehydrogenase and 6-phosphogluconate dehydrogenase in root apices. Physiol Plant 98:477–484
Sun C, Lu L, Liu L, Liu W, Yu Y, Liu X, Hu Y, Jin C, Lin X (2014) Nitrate reductase-mediated early nitric oxide burst alleviates oxidative damage induced by aluminum through enhancement of antioxidant defenses in roots of wheat (Triticum aestivum). New Phytol 201:1240–1250
Sun C, Liu L, Yu Y, Liu W, Lu L, Jin C, Lin X (2015) Nitric oxide alleviates aluminum-induced oxidative damage through regulating the ascorbate-glutathione cycle in roots of wheat. J Integr Plant Biol 57:550–561
Torres MA, Dangl JL, Jones JDG (2002) Arabidopsis gp91phox homologues AtrbohD and AtrbohF are required for accumulation of reactive oxygen intermediates in the plant defense response. Proc Natl Acad Sci U S A 99:517–522
Van Gestelen P, Asard H, Caubergs RJ (1997) Solubilization and separation of a plant plasma membrane NADPH-O2 − synthase from other NAD(P)H oxidoreductase. Plant Physiol 115:543–550
Wakao S, Benning C (2005) Genome-wide analysis of glucose-6-phosphate dehydrogenases in Arabidopsis. Plant J 41:243–256
Wakao S, Andre C, Benning C (2008) Functional analyses of cytosolic glucose-6-phosphate dehydrogenases and their contribution to seed oil accumulation in Arabidopsis. Plant Physiol 146:277–288
Wang YS, Yang ZM (2005) Nitric oxide reduces aluminum toxicity by preventing oxidative stress in the roots of Cassia tora L. Plant Cell Physiol 46:1915–1923
Wang HH, Liang XL, Wan Q, Wang XM, Bi YR (2009) Ethylene and nitric oxide are involved in maintaining ion homeostasis in Arabidopsis Callus under salt stress. Planta 230:293–307
Wang HH, Huang JJ, Bi YR (2010) Nitrate reductase-dependent nitric oxide production is involved in aluminum tolerance in red kidney bean roots. Plant Sci 179:281–288
Wang H, Huang J, Liang X, Bi Y (2012) Involvement of hydrogen peroxide, calcium, and ethylene in the induction of the alternative pathway in chilling-stressed Arabidopsis Callus. Planta 235:53–67
Wang H, Li Y, Hou JJ, Huang JJ, Liang WH (2016a) Nitrate reductase-mediated nitric oxide production alleviates Al-induced inhibition of root elongation by regulating the ascorbate-glutathione cycle in soybean roots. Plant Soil. doi:10.1007/s11104-016-3045-4
Wang H, Yang L, Li Y, Hou J, Huang J, Liang W (2016b) Involvement of ABA- and H2O2-dependent cytosolic glucose-6-phosphate dehydrogenase in maintaining redox homeostasis in soybean roots under drought stress. Plant Physiol Biochem 107:126–136
Wenderoth I, Scheibe R, von Schaewen A (1997) Identification of the cysteine residues involved in redox modification of plant plastidic glucose-6-phosphate dehydrogenase. J Biol Chem 272:26985–26990
Wendt UK, Hauschild R, Lange C, Pietersma M, Wenderoth I, von Schaewen A (1999) Evidence for functional convergence of redox regulation in G6PDH isoforms of cyanobacteria and higher plants. Plant Mol Biol 40:487–494
Xu J, Yin H, Li Y, Liu X (2010) Nitric oxide is associated with long-term zinc tolerance in Solanum nigrum. Plant Physiol 154:1319–1334
Yamamoto Y, Kobayashi Y, Matsumoto H (2001) Lipid peroxidation is an early symptom triggered by aluminum, but not the primary cause of elongation inhibition in pea roots. Plant Physiol 125:199–208
Yamamoto Y, Kobayashi Y, Devi SR, Rikishi S, Matsumoto H (2002) Aluminum toxicity is associated with mitochondrial dysfunction and the production of reactive oxygen species in plant cells. Plant Physiol 128:63–72
Yamamoto Y, Kobayashi Y, Devi SR, Rikiishi S, Matsumoto H (2003) Oxidative stress triggered by aluminum in plant roots. Plant Soil 255:239–243
Yang YL, Zhang F, He WL, Wang XM, Zhang LX (2003) Iron-mediated inhibition of H+-ATPase in plasma membrane vesicles isolated from wheat roots. Cell Mol Life Sci 60:1249–1257
Zhang F, Wang Y, Yang Y, Wu H, Wang D, Liu J (2007) Involvement of hydrogen peroxide and nitric oxide in salt resistance in the calluses from Populus euphratica. Plant Cell Environ 30:775–785
Zhao MG, Tian QY, Zhang WH (2007) Nitric oxide synthase-dependent nitric oxide production is associated with salt tolerance in Arabidopsis. Plant Physiol 144:206–217
Zhao MG, Chen L, Zhang LL, Zhang WH (2009) Nitric reductase-dependent nitric oxide production is involved in cold acclimation and freezing tolerance in Arabidopsis. Plant Physiol 151:755–767
Zhou Y, Xu XY, Chen LQ, Yang JL, Zheng SJ (2012) Nitric oxide exacerbates Al-induced inhibition of root elongation in rice bean by affecting cell wall and plasma membrane properties. Phytochemistry 76:46–51
Acknowledgments
This work was financially supported by the National Natural Science Foundation of China (No. 31301252), Science Foundation of the Henan Normal University for Outstanding Young Scholars (No. 14YQ003), and Program for Innovative Research Team (in Science and Technology) in University of Henan Province (No. 15IRSTHN020).
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible Editor: Jian Feng Ma.
Electronic supplementary material
Fig. S1
Root elongation in response to GN6P and AlCl3 separately. Seedlings were pretreated with GN6P for 12 h and then exposed to AlCl3 for 24 h. Data represent means ± SD of at least three biological replicates. Bars with different letters indicate significant differences at P < 0.05. CK, control. (JPEG 53 kb)
Fig. S2
Effect of NADPH oxidase inhibitor on the lipid peroxidation under Al stress. Treatments for seedlings were performed as described in Fig. 5. Data represent means ± SD of at least three biological replicates. Bars with different letters indicate significant differences at P < 0.05. CK, control. (JPEG 49 kb)
Fig. S3
Effect of different Al concentrations on NO production in soybean roots. Seedlings were exposed to the indicated Al concentrations for 24 h. Representative images showing endogenous NO production are given. CK, control. (JPEG 34 kb)
Rights and permissions
About this article
Cite this article
Wang, H., Hou, J., Li, Y. et al. Nitric oxide-mediated cytosolic glucose-6-phosphate dehydrogenase is involved in aluminum toxicity of soybean under high aluminum concentration. Plant Soil 416, 39–52 (2017). https://doi.org/10.1007/s11104-017-3197-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11104-017-3197-x