Abstract
Aims
This work concentrated on understanding the allocation of Cd recently taken up between the organs of sunflower at early and middle reproductive growth stages. The roles of transpiration and allometry were investigated.
Methods
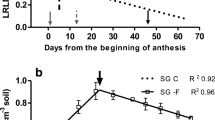
Sunflowers were grown hydroponically in greenhouse, being exposed to low concentrations of Cd (pCd2+ = 11.03). At flower bud and grain filling stages, plants were exposed for three days to 111Cd and at the same time, subjected or not to fans to increase the transpiration. The partitioning of 111Cd between plant organs measured by high resolution ICP-MS was then modelled.
Results
Although the use of fans increased the plant water uptake and transpiration by about 20%, there were no significant effects on the partitioning of recent Cd. Most of the recent Cd was recovered in roots (60%) and only 2.8% were found in seeds (0.8% for the husk and 2.0% for the almonds). The sequestration of recent Cd in a plant organ was successfully explained by its biomass and except for leaves, by the biomass of other organs acting as competitive sinks.
Conclusions
This work proposes a modelling approach for the partitioning of the labelled Cd between plant organs in sunflower.



Similar content being viewed by others
References
Alkio M, Grimm E (2003) Vascular connections between the receptacle and empty achenes in sunflower (Helianthus annuus L.). J Exp Bot 54:345–348. doi:10.1093/jxb/erg019
Arduini I, Masoni A, Mariotti M, Pampana S, Ercoli L (2014) Cadmium uptake and translocation in durum wheat varieties differing in grain-Cd accumulation. Plant Soil Environ 60:43–49
Aston MJ, Lawlor DW (1979) The relationship between transpiration, root water uptake, and leaf water potential. J Exp Bot 30:169–181. doi:10.1093/jxb/30.1.169
Cataldo DA, Garland T, Wildung RE (1983) Cadmium uptake kinetics in intact soybean plants. Plant Physiol 73:844–848
Clemens S, Palmgren MG, Krämer U (2002) A long way ahead: understanding and engineering plant metal accumulation. Trends Plant Sci 7:309–315. doi:10.1016/S1360-1385(02)02295-1
Clemens S, Aarts MGM, Thomine S, Verbruggen N (2013) Plant science: the key to preventing slow cadmium poisoning. Trends Plant Sci 18:92–99. doi:10.1016/j.tplants.2012.08.003
De Maria S, Puschenreiter M, Rivelli AR (2013) Cadmium accumulation and physiological response of sunflower plants to Cd during the vegetative growing cycle. Plant Soil Environ 59:254–261
Degryse F, Smolders E, Parker DR (2006) Metal complexes increase uptake of Zn and Cu by plants: implications for uptake and deficiency studies in chelator-buffered solutions. Plant Soil 289:171–185. doi:10.1007/s11104-006-9121-4
Enstone DE, Peterson CA, Ma FS (2002) Root endodermis and exodermis: structure, function, and responses to the environment. J Plant Growth Regul 21:335–351. doi:10.1007/s00344-003-0002-2
Farrar JF, Jones DL (2000) The control of carbon acquisition by roots. New Phytol 147:43–53. doi:10.1046/j.1469-8137.2000.00688.x
Fujimaki S, Suzui N, Ishioka S, Kawachi N, Ito S, Chino M, Nakamura S (2010) Tracing cadmium from culture to Splikelet: noninvasive imaging and quantitative characterization of absorption, transport, and accumulation of cadmium in an intact Rice Plant. Plant Physiol 152:1796–1806. doi:10.1104/pp.109.151035
Gilliham M, Dayod M, Hocking BJ, Xu B, Conn SJ, Kaiser BM, Leigh RA, Tyerman ED (2011) Calcium delivery and storage in plant leaves: exploring the link with water flow. J Exp Bot 62:2233–2250. doi:10.1093/jxb/err111
Hall JL, Williams LE (2003) Transition metal transporters in plants. J Exp Bot 54:2601–2613. doi:10.1093/jxb/erg303
Hardiman RT, Jacoby B (1984) Absorption and translocation of Cd in bush beans (Phaseolus vulgaris). Physiol Plantarum 61:670–674. doi:10.1111/j.1399-3054.1984.tb05189.x
Harris NS, Taylor GJ (2001) Remobilization of cadmium in maturing shoots of near isogenic lines of durum wheat that differ in grain cadmium accumulation. J Exp Bot 52:1473–1481. doi:10.1093/jexbot/52.360.1473
Harris NS, Taylor GJ (2004) Cadmium uptake and translocation in seedlings of near isogenic lines of durum wheat that differ in grain cadmium accumulation. BMC Plant Biol 14(4):4. doi:10.1186/1471-2229-4-4
Harris NS, Taylor GJ (2013) Cadmium uptake and partitioning in durum wheat during grain filling. BMC Plant Biol 13:103. doi:10.1186/1471-2229-13-103
Hegelund JN, Pedas P, Husted S, Schiller M, Schjoerring JK (2012) Zinc fluxes into developing barley grains: use of stable Zn isotopes to separate root uptake from remobilization in plants with contrasting Zn status. Plant Soil 361:241–250. doi:10.1007/s11104-012-1272-x
Hoagland DR, Arnon DI (1950) The water-culture method for growing plants without soil. Circular 347. California Agricultural Experiment Station. The College of Agriculture, University of California, Berkeley, California
Lai H (2015) Effects of leaf area and transpiration rate on accumulation and compartmentalization of cadmium in Impatiens walleriana. Water Air Soil Poll 226:2246. doi:10.1007/s11270-014-2246-9
Laporte M, Sterckeman T, Dauguet S, Denaix L, Nguyen C (2015) Variability in cadmium and zinc shoot concentration in 14 cultivars of sunflower (Helianthus annuus L.) as related to metal uptake and partitioning. Environ Exp Bot 109:45–53. doi:10.1016/j.envexpbot.2014.07.020
Lei H, Zhi-Shan Z, Xin-Rong L (2010) Sap flow of Artemisia ordosica and the influence of environmental factors in a revegetated desert area: Tengger Desert, China. Hydrol Process 24:1248–1253. doi:10.1002/hyp.7584
Liu J, Qian M, Cai G, Yang J, Zhu Q (2007) Uptake and translocation of Cd in different rice cultivars and the relation with Cd accumulation in rice grain. J Hazard Mater 143:443–447. doi:10.1016/j.jhazmat.2006.09.057
Liu X, Peng K, Wang A, Lian C, Shen Z (2010) Cadmium accumulation and distribution in population of Phytolacca americana L. And the role of transpiration. Chemosphere 78:1136–1141. doi:10.1016/j.chemosphere.2009.12.030
Liu K, Lv J, He W, Zhang H, Cao Y, Dai Y (2015) Major factors influencing cadmium uptake from the soil into wheat plants. Ecotoxicol Environ Saf 113:207–213. doi:10.1016/j.ecoenv.2014.12.005
Lux A, Martinka M, Vaculík M, White PJ (2011) Root responses to cadmium in the rhizosphere: a review. J Exp Bot 62:21–37. doi:10.1093/jxb/erq281
Marschner H, Marschner P (2012) Marschner’s mineral nutrition of higher plants. Academic Press, San Diego
Mori S, Uraguchi S, Ishikawa S, Arao T (2009) Xylem loading process is a critical factor in determining Cd accumulation in the shoots of Solanum melongena and Solanum torvum. Environ Exp Bot 67:127–132. doi:10.1016/j.envexpbot.2009.05.006
Olsen LI, Palmgren MG (2014) Many rivers to cross: the journey of zinc from soil to seed. Front Plant Sci 5:30. doi:10.3389/fpls.2014.00030
Patrick JW, Offler CE (2001) Compartmentation of transport and transfer events in developing seeds. J Exp Bot 52:551–564. doi:10.1093/jexbot/52.356.551
Prado K, Maurel C (2013) Regulation of leaf hydraulics: from molecular to whole plant levels. Front Plant Sci 4:255. doi:10.3389/fpls.2013.00255
Quinn CJ, Mohammad A, Macfie SM (2011) Accumulation of cadmium in near-isogenic lines of durum wheat (Triticum turgidum L. Var durum): the role of transpiration. Physiol Mol Biol Plants 17:317–325. doi:10.1007/s12298-011-0086-2
R Core Team (2015) R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. URL https://www.R-project.org/.
Rauser WE (1987) Compartmental efflux analysis and removal of extracellular cadmium from roots. Plant Physiol 85:62–65
Rivelli AR, De Maria S, Puschenreiter M, Gherbin P (2012) Accumulation of cadmium, zinc, and copper by Helianthus annuus L.: impact on plant growth and uptake of nutritional elements. Int J Phytoremediat 14:320–334
Rodríguez-Cea A, Fernández de la Campa M, García Alonso JI, Sanz-Medel A (2006) The use of enriched 111Cd as tracer to study de novo cadmium accumulation and quantitative speciation in Anguilla anguilla tissues. J Anal At Spectrom 21:270–278. doi:10.1039/B515828A
Salt DE, Prince RC, Pickering IJ, Raskin I (1995) Mechanisms of cadmium mobility and accumulation in indian mustard. Plant Physiol 109:1427–1433. doi:10.1104/pp.109.4.1427
Sanità di Toppi L, Gabbrielli R (1999) Response to cadmium in higher plants. Environ Exp Bot 41:105–130. doi:10.1016/S0098-8472(98)00058-6
Satoh-Nagasawa N, Mori M, Nakazawa N, Kawamoto T, Nagato Y, Sakurai K, Takahashi H, Watanabe A, Akagi H (2012) Mutations in rice (Oryza sativa) heavy metal ATPase 2 (OsHMA2) restrict the translocation of zinc and cadmium. Plant Cell Physiol 53:213–224. doi:10.1093/pcp/pcr166
Sattelmacher B, Horst WJ (2007) The apoplast of higher plants: compartment of storage, transport and reactions. The significance of the apoplast for the mineral nutrition of higher plants, Springer Netherlands
Schurr U (1998) Xylem sap sampling—new approaches to an old topic. Trends Plant Sci 3:293–298. doi:10.1016/S1360-1385(98)01275-8
Shah J (1998) Chapter 7. Loss of water. In: New Age International (ed) Plant Physiology, pp 92–127
Singh A, Prasad SM (2011) Reduction of heavy metal load in food chain: technology assessment. Rev Environ Sci Bio 10:199–214. doi:10.1007/s11157-011-9241-z
Spicer R (2014) Symplasmic networks in secondary vascular tissues: parenchyma distribution and activity supporting long-distance transport. J Exp Bot 65:1829–1848. doi:10.1093/jxb/ert459
Tanaka M, Wallace IS, Takano J, Roberts DM, Fujiwara T (2008) NIP6;1 is a boric acid channel for preferential transport of boron to growing shoot tissues in Arabidopsis. Plant Cell 20:2860–2875. doi:10.1105/tpc.108.058628
Tester M, Leigh RA (2001) Partitioning of nutrient transport processes in roots. J Exp Bot 52:445–457. doi:10.1093/jexbot/52.suppl_1.445
Uraguchi S, Fujiwara T (2013) Rice breaks ground for cadmium-free cereals. Curr Opin Plant Biol 16:328–334. doi:10.1016/j.pbi.2013.03.012
Uraguchi S, Mori S, Kuramata M, Kawasaki A, Arao T, Ishikawa S (2009) Root-to-shoot Cd translocation via the xylem is the major process determining shoot and grain cadmium accumulation in rice. J Exp Bot 60:2677–2688. doi:10.1093/jxb/erp119
Van Bel AJE (1990) Xylem-phloem exchange via the rays: the undervalued route of transport. J Exp Bot 41:631–644. doi:10.1093/jxb/41.6.631
Van der Schoot C, Van Bel AJE (1989) Architecture of the Internodal xylem of tomato (Solanum lycopersicum) with reference to longitudinal and lateral transfer. Am J Bot 76:487–503. doi:10.2307/2444344
Van der Vliet L, Peterson C, Hale B (2007) Cd accumulation in roots and shoots of durum wheat: the roles of transpiration rate and apoplastic bypass. J Exp Bot 58:2939–2947
Verret F, Gravot A, Auroy P, Leonhardt N, David P, Nussaume L, Vavasseur A, Richard P (2004) Overexpression of AtHMA4 enhances root-to-shoot translocation of zinc and cadmium and plant metal tolerance. FEBS Lett 576:306–312. doi:10.1016/j.febslet.2004.09.023
Waters BM, Sankaran RP (2011) Moving micronutrients from the soil to the seeds: genes and physiological processes from a biofortification perspective. Plant Sci 180:562–574. doi:10.1016/j.plantsci.2010.12.003
White PJ (2012) Chapter 3 - long-distance transport in the xylem and phloem. In: Marschner P (ed) Marschner’s mineral nutrition of higher plants, Third edn. Academic Press, San Diego, pp. 49–70
Yamaguchi N, Mori S, Baba K, Kaburagi-Yada S, Arao T, Kitajima N, Hokura A, Terada Y (2011) Cadmium distribution in the root tissues of solanaceous plants with contrasting root-to-shoot Cd translocation efficiencies. Environ Exp Bot 71:198–206. doi:10.1016/j.envexpbot.2010.12.002
Yamaguchi N, Ishikawa S, Abe T, Baba K, Arao T, Terada Y (2012) Role of the node in controlling traffic of cadmium, zinc, and manganese in rice. J Exp Bot 63:2729–2737. doi:10.1093/jxb/err455
Yamaji N, Ma JF (2014) The node, a hub for mineral nutrient distribution in graminaceous plants. Trends Plant Sci 19:556–563. doi:10.1016/j.tplants.2014.05.007
Acknowledgments
This work has been financially supported by the French National Research Agency through the SIM-TRACES (ANR-11-CESA-0008) Project.
O. Liñero is grateful to the University of the Basque Country (UPV/EHU) and the University of Bordeaux for her predoctoral fellowship, within the framework of the Cross-Border Euroregional Campus of International Excellence IdEx Bordeaux – Euskampus. O.S. Pokrovsky thanks partial support of BIO-GEO-CLIM grant No 14.B25.31.0001. The authors are grateful to L. Champolivier (Terres Inovia) for his helpful advices about the pollination and seed development of sunflower under controlled conditions.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible Editor: Henk Schat.
Electronic Supplementary Material
Online Resource 1
Observed 114Cd/111Cd ratios against theoretical ones. The slope (p < 0.001) and the intercept (p < 0.05) values of the regression line do not significantly differ from 1 and 0, respectively. (PDF 69 kb)
Online Resource 2
The best regression models selected for explaining the partitioning of labelled Cd in each plant organ, following Eq. 4 and 5 of the materials and methods (AG = aboveground organs). E i indicates the amount of labelled Cd exported by the previous plant organ, according to the general flow chart of labelled Cd described in Fig. 1. The significance of the intercept is coded as * p < 0.05, ** p < 0.01, *** p < 0.001. (PDF 118 kb)
Rights and permissions
About this article
Cite this article
Liñero, O., Cornu, JY., Candaudap, F. et al. Short-term partitioning of Cd recently taken up between sunflowers organs (Helianthus annuus) at flowering and grain filling stages: effect of plant transpiration and allometry. Plant Soil 408, 163–181 (2016). https://doi.org/10.1007/s11104-016-2915-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11104-016-2915-0