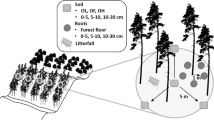
Abstract
Background and aims
The carbon (C) sequestration potential of land-use practices is increasingly important. Trees sequester atmospheric C into biomass and above and belowground litter but may also prime the decomposition of soil organic matter (SOM). We compared the influence of Acer pseudoplatanus (Sycamore) and Larix x. europlepsis (Hybrid Larch) on soil C decomposition.
Methods
We used natural abundance 13C to partition soil-surface CO2 efflux into root and SOM sources. CO2 was sampled from incubated root-free soil and from live tree roots using in-situ chambers. Combined surface efflux δ13CO2 was measured using dynamic chambers and cavity-ringdown spectroscopy.
Results
Under Sycamore, CO2 emissions were dominated (80–90 %) by root respiration. SOM contributed 10–20 % with a mean residence time of centuries. Under Larch, 24–33 % of total CO2 efflux was root respiration, the remainder originating from an SOM pool with a turnover time of decades. Total soil C stocks were similar between the two plot types. Root-respired δ13CO2 was consistently different by c. 2 ‰ between the species.
Conclusions
The decomposition rate of soil C and its mean residence time are markedly different under the two tree species. Species differences in root-respired δ13CO2 may reflect plant C allocation or respiratory fractionation.





Similar content being viewed by others
References
Ayres E, Steltzer H, Berg S, Wallenstein MD, Simmons BL, Wall DH (2009) Tree species traits influence soil physical, chemical, and biological properties in high elevation forests. PLoS One 4(6). doi:10.1371/journal.pone.0005964
Beckert MR, Smith P, Lilly A, Chapman S (2015) Soil and tree biomass carbon sequestration potential of silvopastoral and woodland-pasture systems in North East Scotland. Agrofor Syst. doi:10.1007/s10457-015-9860-4
Bond-Lamberty B, Thomson A (2010) Temperature-associated increases in the global soil respiration record. Nature 464:579–582
Bowling DR, Pataki DE, Randerson JT (2008) Carbon isotopes in terrestrial ecosystem pools and CO2 fluxes. New Phytol 178:24–40
Brüggemann N, Gessler A, Kayler Z, Keel SG, Badeck F, Barthel M, Boeckx P, Buchmann N, Brugnoli E, Esperschütz J, Gavrichkova O, Ghashghaie J, Gomez-Casanovas N, Keitel C, Knohl A, Kuptz D, Palacio S, Salmon Y, Uchida Y, Bahn M (2011) Carbon allocation and carbon isotope fluxes in the plant-soil-atmosphere continuum: a review. Biogeosciences 8:3457–3489
Brzostek ER, Finzi AC (2011) Substrate supply, fine roots, and temperature control proteolytic enzyme activity in temperate forest soils. Ecology 92:892–902
Brzostek ER, Greco A, Drake JE, Finzi AC (2013) Root carbon inputs to the rhizosphere stimulate extracellular enzyme activity and increase nitrogen availability in temperate forest soils. Biogeochemistry 115:65–76
Chen D, Zhang Y, Lin Y, Chen H, Fu S (2009) Stand level estimation of root respiration for two subtropical plantations based on in situ measurement of specific root respiration. For Ecol Manag 257:2088–2097
Chen G, Yang Y, Robinson D (2014) Allometric constraints on, and trade-offs in, belowground carbon allocation and their control of soil respiration across global forest ecosystems. Glob Chang Biol 20:1674–1684
Cheng L, Booker FL, Tu C, Burkey KO, Zhou L, Shew HD, Rufty TW, Hu S (2012) Arbuscular mycorrhizal fungi increase organic carbon decomposition under elevated CO2. Science 337:1084–1087
Churchland C, Grayston SJ (2014) Specificity of plant-microbe interactions in the tree mycorrhizosphere biome and consequences for soil C cycling. Front Microbiol 5:261
Cornelissen J, Aerts R, Cerabolini B, Werger M, Van der Heijden M (2001) Carbon cycling traits of plant species are linked with mycorrhizal strategy. Oecologia 129:611–619
Crow SE, Sulzman EW, Rugh WD, Bowden RD, Lajtha K (2006) Isotopic analysis of respired CO2 during decomposition of separated soil organic matter pools. Soil Biol Biochem 38:3279–3291
Dijkstra F, Cheng W (2007) Interactions between soil and tree roots accelerate long-term soil carbon decomposition. Ecol Lett 10:1046–1053
Drake JE, Oishi AC, Giasson M, Oren R, Johnsen KH, Finzi AC (2012) Trenching reduces soil heterotrophic activity in a loblolly pine (Pinus taeda) forest exposed to elevated atmospheric [CO2] and N fertilization. Agric For Meteorol 165:43–52
Dungait JAJ, Hopkins DW, Gregory AS, Whitmore AP (2012) Soil organic matter turnover is governed by accessibility not recalcitrance. Glob Chang Biol 18:1781–1796
Graham SL, Millard P, Hunt JE, Rogers GN, Whitehead D (2012) Roots affect the response of heterotrophic soil respiration to temperature in tussock grass microcosms. Ann Bot 110:253–258
Grayston SJ (2000) Rhizodeposition and its impact on microbial community structure and function in trees. Phyton-Annales Rei Botanicae 40:27–36
Harley JL, Harley EL (1987) A check list of mycorrhiza in the British flora. New Phytol 105:1–102
Heinemeyer A, Tortorella D, Petrovičová B, Gelsomino A (2012) Partitioning of soil CO2 flux components in a temperate grassland ecosystem. Eur J Soil Sci 63:249–260
Hogberg P, Plamboeck AH, Taylor AFS, Fransson PMA (1999) Natural 13C abundance reveals trophic status of fungi and host-origin of carbon in mycorrhizal fungi in mixed forests. Proc Natl Acad Sci U S A 96:8534–8539
Johansson EM, Fransson PMA, Finlay RD, van Hees PAW (2009) Quantitative analysis of soluble exudates produced by ectomycorrhizal roots as a response to ambient and elevated CO2. Soil Biol Biochem 41:1111–1116
Keith AM, Rowe RL, Parmar K, Perks MP, Mackie E, Dondini M, McNamara NP (2014) Implications of land-use change to short rotation forestry in Great Britain for soil and biomass carbon. GCB Bioenergy. doi:10.1111/gcbb.12168
Kuzyakov Y (2002) Review: factors affecting rhizosphere priming effects. J Plant Nutr Soil Sci 165:382–396
Kuzyakov Y (2006) Sources of CO2 efflux from soil and review of partitioning methods. Soil Biol Biochem 38:425–448
Kuzyakov Y, Friedel JK, Stahr K (2000) Review of mechanisms and quantification of priming effects. Soil Biol Biochem 32:1485–1498
Lal R (2010) Beyond Copenhagen: mitigating climate change and achieving food security through soil carbon sequestration. Food Sec 2:169–177
Lambers H, Chapin FS, Pons TL (2008) Plant physiological ecology, Second edn. Springer Science + Business Media, New York, pp. 56–57
Marsden C, Nouvellon Y, Epron D (2008) Relating coarse root respiration to root diameter in clonal Eucalyptus stands in the Republic of the Congo. Tree Physiol 28:1245–1254
Michalet S, Rohr J, Warshan D, Bardon C, Roggy J-C, Domenach A-M, Czarnes S, Pommier T, Combourieu B, Guillaumaud N, Bellvert F, Comte G, Poly F (2013) Phytochemical analysis of mature tree root exudates in situ and their role in shaping soil microbial communities in relation to tree N-acquisition strategy. Plant Physiol Biochem 72:169–177
Midwood AJ, Millard P (2011) Challenges in measuring the δ13C of the soil surface CO2 efflux. Rapid Commun Mass Spectrom 25:232–242
Midwood AJ, Thornton B, Millard P (2008) Measuring the 13C content of soil-respired CO2 using a novel open chamber system. Rapid Commun Mass Spectrom 22:2073–2081
Millard P, Midwood AJ, Hunt JE, Barbour MM, Whitehead D (2010) Quantifying the contribution of soil organic matter turnover to forest soil respiration, using natural abundance δ13C. Soil Biol Biochem 42:935–943
Ouimette A, Guo D, Hobbie E, Gu J (2013) Insights into root growth, function, and mycorrhizal abundance from chemical and isotopic data across root orders. Plant Soil 367:313–326
Pérez-Harguindeguy N, Díaz S, Cornelissen JHC, Vendramini F, Cabido M, Castellanos A (2000) Chemistry and toughness predict leaf litter decomposition rates over a wide spectrum of functional types and taxa in central Argentina. Plant Soil 218:21–30
Phillips RP, Fahey TJ (2006) Tree species and mycorrhizal associations influence the magnitude of rhizosphere effects. Ecology 87:1302–1313
Phillips DL, Gregg JW (2001) Uncertainty in source partitioning using stable isotopes. Oecologia 127:171–179
Phillips RP, Brzostek E, Midgley MG (2013) The mycorrhizal-associated nutrient economy: a new framework for predicting carbon-nutrient couplings in temperate forests. New Phytol 199:41–51
Powlson DS, Gregory PJ, Whalley WR, Quinton JN, Hopkins DW, Whitmore AP, Hirsch PR, Goulding KWT (2011) Soil management in relation to sustainable agriculture and ecosystem services. Food Policy 36:S72–S87
Prescott CE, Grayston SJ (2013) Tree species influence on microbial communities in litter and soil: current knowledge and research needs. For Ecol Manag 309:19–27
Raich JW, Schlesinger WH (1992) The global carbon dioxide flux in soil respiration and its relationship to vegetation and climate. Tellus 44B:81–99
Reich PB, Oleksyn J, Modrzynski J, Mrozinski P, Hobbie SE, Eissenstat DM, Chorover J, Chadwick OA, Hale CM, Tjoelker MG (2005) Linking litter calcium, earthworms and soil properties: a common garden test with 14 tree species. Ecol Lett 8:811–818
Schmidt MWI, Torn MS, Abiven S, Dittmar T, Guggenberger G, Janssens IA, Kleber M, Kögel-Knabner I, Lehmann J, Manning DAC, Nannipieri P, Rasse DP, Weiner S, Trumbore SE (2011) Persistence of soil organic matter as an ecosystem property. Nature 478:49–56
Sibbald AR, Eason WR, Mcadam JH, Hislop AM (2001) The establishment phase of a silvopastoral national network experiment in the UK. Agrofor Syst 53:39–53
Six J, Conant RT, Paul EA, Paustian K (2002) Stabilization mechanisms of soil organic matter: implications for C-saturation of soils. Plant Soil 241:155–176
Snell HSK, Robinson D, Midwood AJ (2014) Minimising methodological biases to improve the accuracy of partitioning soil respiration using natural abundance 13C. Rapid Commun Mass Spectrom 28:2341–2351
Snell HSK, Robinson D, Midwood AJ (2015) Sampling root-respired CO2 in-situ for 13C measurement. Plant Soil 393:259–271
Söderström B, Read DJ (1987) Respiratory activity of intact and excised ectomycorrhizal mycelial systems growing in unsterilized soil. Soil Biol Biochem 19:231–236
Staddon PL, Robinson D, Graves JD, Fitter AH (1999) The delta C-13 signature of the external phase of a Glomus mycorrhizal fungus: determination and implications. Soil Biol Biochem 31:1067–1070
Sulman BN, Phillips RP, Oishi AC, Shevliakova E, Pacala SW (2014) Microbe-driven turnover offsets mineral-mediated storage of soil carbon under elevated CO2. Nat Clim Chang 4:1099–1102
Unger S, Máguas C, Pereira JS, Aires LM, David TS, Werner C (2010) Disentangling drought-induced variation in ecosystem and soil respiration using stable carbon isotopes. Oecologia 163:1043–1057
Werth M, Kuzyakov Y (2010) 13C fractionation at the root-microorganisms-soil interface: a review and outlook for partitioning studies. Soil Biol Biochem 42:1372–1384
West TO, Post WM (2002) Soil organic carbon sequestration rates by tillage and crop rotation: a global data analysis. Soil Sci Soc Am 66:1930–1946
Zakharova A, Midwood AJ, Hunt JE, Graham SL, Artz RRE, Turnbull MH, Whitehead D, Millard P (2014) Loss of labile carbon following soil disturbance determined by measurement of respired ∂13CO2. Soil Biol Biochem 68:125–132
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible Editor: Klaus Butterbach-Bahl.
Rights and permissions
About this article
Cite this article
Snell, H.S.K., Robinson, D. & Midwood, A.J. Tree species’ influences on soil carbon dynamics revealed with natural abundance 13C techniques. Plant Soil 400, 285–296 (2016). https://doi.org/10.1007/s11104-015-2731-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11104-015-2731-y