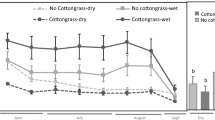
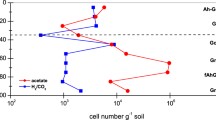
Abstract
Aims
Assess the role of Phragmites australis in the temporal variability of physico-chemical and microbiological soil properties related to biogeochemical processes in eutrophic wetlands.
Methods
A mesocosms experiment was performed with alternating flooding-drying conditions with eutrophic water at two nutrient levels, and soil Eh, pH, temperature, CO2 emissions, dissolved organic carbon, carbon from microbial biomass, and Phragmites physiological activity were measured during 44 weeks.
Results
In surface, Eh decreased with flooding and increased with drying regardless plant presence and nutrients content. In depth, Phragmites maintained oxic conditions. During warmer months, O2 diffusion promoted by Phragmites hindered the drop of pH. Soil microbial respiration was stimulated in warmer months (soil temperature ~ 20–30 °C), as shown by larger CO2 production, and higher aromaticity and phenolic compounds content in pore water. The latter occurred regardless the plant presence and nutrients content, although the combination of both contributed to a higher microbial population (shown by higher concentrations of carbon from microbial biomass).
Conclusions
The presence of Phragmites and the nutrient concentration in the flooding water had a different role in the temporal evolution of the physico-chemical and microbiological soil properties in eutrophic wetlands, and this role was strongly influenced by soil depth and temperature.










Similar content being viewed by others
References
Álvarez-Rogel J, Ortiz R, Vela de Oro N, Alcaraz F (2001) The application of the FAO and US Soil Taxonomy systems to saline soils in relation to halophytic vegetation in SE Spain. Catena 45:73–84
Álvarez-Rogel J, Jiménez-Cárceles FJ, Egea C (2006) Phosphorus and nitrogen content in the water of a coastal wetland in the Mar Menor lagoon (SE Spain): relationships with effluents from urban and agricultural areas. Water Air Soil Pollut 173:21–38
Álvarez-Rogel J, Jiménez-Cárceles FJ, Roca MJ, Ortiz R (2007) Changes in soils and vegetation in a Mediterranean coastal salt marsh impacted by human activities. Estuar Coast Shelf Sci 73:510–526
Armstrong W (1979) Aeration in higher plants. Adv Bot Res 7:225–232
Armstrong W, Cousins D, Armstrong J, Turner DW, Beckett PM (2000) Oxygen distribution in wetland plant roots and permeability barriers to gas-exchange with the rhizosphere: a microelectrode study with Phragmites australis. Ann Bot 86:687–703
Caffery JM, Kemp WM (1990) Nitrogen cycling in sediments with estuarine populations of Potamogeton perfoliatus and Zostera marina. Mar Ecol Prog Ser 66:147–160
Colmer TD (2003) Long-distance transport of gases in plants: a perspective on internal aeration and radial oxygen loss from roots. Plant Cell Environ 26:17–36
Conant RT, Drijberw RA, Haddix ML, Parton WJ, Paul EA, Plantez AF, Six J, Steinweg M (2008) Sensitivity of organic matter decomposition to warming varies with its quality. Glob Chang Biol 14:868–877
De Nobili M, Contin M, Mondini C, Brookes PC (2001) Soil microbial biomass is triggered into activity by trace amounts of substrate. Soil Biol Biochem 33:3701–3709
DeBusk WF, Reddy KR (1998) Turnover of detrital organic carbon in a nutrient-impacted Everglades marsh. Soil Sci Soc Am J 62:1460–1468
Dickopp J, Kazda M, Cízková H (2011) Differences in rhizome aeration of Phragmites australis in a constructed wetland. Ecol Eng 37:1647–1653
Fiedler S, Vepraskas MJ, Richardson JJ (2007) Soil redox potential: importance, field measurements, and observations. Adv Agron 94:1–54
Findlay S, Groffman P, Dye S (2003) Effects of Phragmites australis removal on marsh nutrient cycling. Wet Ecol Manag 11:157–165
González-Alcaraz MN, Egea C, Jiménez-Cárceles FJ, Párraga I, María-Cervantes A, Delgado MJ, Álvarez-Rogel J (2012) Storage of organic carbon, nitrogen and phosphorus in the soil–plant system of Phragmites australis stands from a eutrophicated Mediterranean salt marsh. Geoderma 185–186:61–72
Goulding K (2000) Nitrate leaching from arable and horticultural land. Soil Use Manag 16:145–151
Greenway H, Armstrong W, Colmer TD (2006) Conditions leading to high CO2 (>5 kPa) in waterlogged-flooded soils and possible effects on root growth and metabolism. Ann Bot (Lond) 98:9–32
Hinsinger P, Bengough AG, Vetterlein D, Young IM (2009) Rhizosphere: biophysics, biogeochemistry and ecological relevance. Plant Soil 321:117–152
Hulseman J (1996) An inventory of marine carbonate materials. J Sediment Petrol ASCE 36:622–625
Jackson MB, Armstrong W (1999) Formation of aerenchyma and the processes of plant ventilation in relation to soil flooding and submergence. Plant Biol 1:274–287
Jenkinson DS, Brookes PC, Powlson DS (2004) Citation classics. Measuring soil microbial biomass. Soil Biol Biochem 36:5–7
Lee B-H, Scholz M (2007) What is the role of Phragmites australis in experimental constructed wetland filters treating urban runoff? Ecol Eng 29:87–95
Lindén A, Heinonsalo J, Buchmann N, Oinonen M, Sonninen E, Hilasvuori E, Pumpanen J (2014) Contrasting effects of increased carbon input on boreal SOM decomposition with and without presence of living root system of Pinus sylvestris L. Plant Soil 77:145–158
Maxwell K, Johnson GN (2000) Chlorophyll fluorescence--a practical guide. J Exp Bot 51:659–668
McKnight DM, Boyer EW, Westerhoff PK, Doran PT, Kulbe T, Anderson DT (2001) Spectrofluorometric characterization of dissolved organic matter for indication of precursor organic material and aromaticity. Limnol Oceanogr 1:38–48
McLatchey GP, Reddy KR (1998) Regulation of organic matter decomposition and nutrient release in a wetland soil. J Environ Qual 27:1268–1274
Mentzer JL, Goodman RM, Balser TC (2006) Microbial response over time to hydrologic and fertilization treatments in a simulated wet prairie. Plant Soil 284:85–100
Mitsch WJ, Gosselink JG (2007) Wetlands. Wiley, New York
Mitsch W, Day JJW, William WJ, Groffman PN, Hey DH, Randall GW, Wang N (2001) Reducing nitrogen loading to the Gulf of Mexico from the Mississippi River Basin: strategies to counter a persistent ecological pro- blem. Bioscience 51:373–388
Muller G, Gastner M (1971) Chemical analysis. Neues Jahrbuch Mineral Monatsh 10:466–469
Nikolausza M, Kappelmeyer U, Székely A, Rusznyák A, Márialigeti K, Kästnera M (2008) Diurnal redox fluctuation and microbial activity in the rhizosphere of wetland plants. Eur J Soil Biol 44:324–333
Otero XL, Macías F (2003) Spatial variation in pyritization of trace metals in saltmarsh soils. Biogeochemistry 62:59–86
Pérez-Tortosa V, López-Orenes A, Pérez-Martínez A, Ferrer MA, Calderón AA (2012) Antioxidant activity and rosmarinic acid changes in salicylic acid-treated Thymus membranaceus shoots. Food Chem 130:362–369
Philippot L, Hallin S, Börjensson G, Baggs EM (2009) Biogeochemical cycling in the rhizosphere having an impact on global change. Plant Soil 321:61–81
Ponnamperuma FN, Martinez E, Loy T (1966) Influence of redox potential and partial pressure of carbon dioxide on pH values and suspension effect of flooded soils. Soil Sci 101:421–431
Reddy KR, DeLaune RD (2008) Biogeochemistry of wetlands: science and applications. CRC Press, Boca Raton
Rostaminia M, Mahmoodi S, Gol Sefidi HT, Pazira E, Kafaee SB (2011) Study of reduction-oxidation potential and chemical characteristics of a paddy field during rice growing season. J Appl Sci 11:1004–1011. doi:10.3923/jas.2011.1004.1011
Ruiz M, Velasco J (2010) Nutrient bioaccumulation in Phragmites australis: management tool for reduction of pollution in the Mar Menor. Water Air Soil Pollut 205:173–185
Saeed T, Sun G (2012) A review on nitrogen and organics removal mechanisms in subsurface flow constructed wetlands: dependency on environmental parameters, operating conditions and supporting media. J Environ Manag 112:429–448
Sánchez-Carrillo S, Álvarez-Cobelas M (2001) Nutrient dynamics and eutrophication patterns in a semi-arid wetland: the effects of fluctuating hydrology. Water Air Soil Pollut 131:97–118
Schöttelndreier M, Falkengren-Grerup U (1999) Plant induced alteration in the rhizosphere and the utilisation of soil heterogeneity. Plant Soil 209:297–309
Simões MP, Caladoa ML, Madeirab M, Gazarinia LC (2011) Decomposition and nutrient release in halophytes of a Mediterranean salt marsh. Aquat Bot 94:119–126
Smith VH, Tilman GD, Nekola JC (1999) Eutrophication: impacts of excess nutrient inputs on freshwater, marine, and terrestrial ecosystems. Environ Pollut 100:179–196
Somenahally AC, Hollister EB, Loeppert RH, Yan W, Gentry TJ (2011) Microbial communities in rice rhizosphere altered by intermittent and continuous flooding in fields with long-term arsenic application. Soil Biol Biochem 43:1220–1228
SPSS (2008) SPSS 17.0 for Windows Software. SPSS Inc., USA
SPSS Inc (2006) Manual del usuario del SPSS Base 15.0. SPSS Inc., Chicago, USA
Stumm W, Sulzberger B (1992) The cycling of iron in natural environments: considerations based on laboratory studies of heterogeneous redox processes. Geochim Cosmochim Acta 56:3233–3257
Thullen JS, Sartoris JJ, Walton WE (2002) Effects of vegetation management in constructed wetland treatment cells on water quality and mosquito production. Ecol Eng 18:441–457
Vance ED, Brookes PC, Jenkinson DS (1987) An extraction method for measuring microbial biomass C. Soil Biol Biochem 19:703–707
Vepraskas MJ, Faulkner SP (2001) Redox chemistry of hydric soils. In: Richardson JL, Vepraskas MJ (eds) Wetland Soils. Genesis, Hydrology, Landscape and Classification. Lewis Publishers, Florida, pp 85–106
Visser EJW, Colmer TD, Blom CWPM, Voesenek LACJ (2000) Changes in growth, porosity, and radial oxygen loss from adventitious roots of selected mono- and dicotyledonous wetland species with contrasting types of aerenchyma. Plant Cell Environ 23:1237–1245
Vymazal J (2007) Removal of nutrients in various types of constructed wetlands. Sci Total Environ 380:48–65
Wang Y, Zhang D, Shen Z, Feng C, Chen J (2013) Revealing sources and distribution changes of dissolved organic matter (DOM) in pore water of sediment from the Yangtze estuary. PLoS ONE 8(10), e76633. doi:10.1371/journal.pone.0076633
Weishaar JL, Aiken GR, Bergamaschi BA, Fram MS, Fujii R, Mopper K (2003) Evaluation of specific ultraviolet absorbance as an indicator of the chemical composition and reactivity of dissolved organic carbon. Environ Sci Technol 37:4702–4708
Zhang H, Cui B, Hong J, Zhang K (2011) Synergism of natural and constructed wetlands in Beijing, China. Ecol Eng 37:128–138
Zhu B, Cheng W (2011) Rhizosphere priming effect increases the temperature sensitivity of soil organic matter decomposition. Glob Chang Biol 17:2172–2183
Acknowledgments
Support for this research was provided by the Ministerio de Ciencia e Innovación of Spain (CGL2010-20214). Dr. M. Nazaret Gonzaléz-Alcaraz thanks the Fundación Ramón Areces for funding her post-doctoral grant in the Department of Ecological Science, Faculty of Earth and Life Sciences, VU University. Dr. Héctor M. Conesa thanks the Spanish Ministerio de Economía y Competitividad and UPCT for funding through the “Ramon y Cajal” programme (Ref. RYC-2010-05665). Antonio López-Orenes received a pre-doctoral grant (AP2012-2559) financed by the Ministerio de Educación, Cultura y Deporte of Spain.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible Editor: Elizabeth M Baggs.
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(DOCX 63 kb)
Rights and permissions
About this article
Cite this article
Tercero, M.C., Álvarez-Rogel, J., Conesa, H.M. et al. Response of biogeochemical processes of the water-soil-plant system to experimental flooding-drying conditions in a eutrophic wetland: the role of Phragmites australis . Plant Soil 396, 109–125 (2015). https://doi.org/10.1007/s11104-015-2589-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11104-015-2589-z