Abstract
Introduction
Cushing’s disease is a rare disorder characterized by overproduction of ACTH from a pituitary adenoma leading to hypercortisolemia that in turn leads to increased morbidity and mortality.
Methods
Here we review the comorbidities associated with Cushing’s disease and their impact on quality of life and mortality.
Results
Recent evidence suggests that correction of hypercortisolemia may not lead to complete resolution of comorbidities associated with this condition. In particular, increased cardiovascular risk may persist despite long-term remission of hypercortisolemia. This may be related to persistence of visceral adiposity, adverse adipokine profile, glucose intolerance, hypertension, dyslipidemia, atherosclerosis and a procoagulant phenotype. Prior prolonged exposure to glucocorticoids also may have irreversible effects on the central nervous system, leading to persistent cognitive and mood alterations. Osteoporosis and fractures, especially vertebral fractures, can further add to morbidity and a poor quality of life. Normalization of cortisol levels leads to significant improvement in comorbidities but long-term data regarding complete resolution are lacking and need further study.
Conclusion
Early diagnosis and treatment of hypercortisolemia, aggressive management of comorbidities along with long-term follow-up is crucial for the optimal recovery of these patients.
Similar content being viewed by others
Introduction
Cushing’s disease (CD) is a rare disorder caused by overproduction of adrenocorticotrophin hormone (ACTH) by a pituitary adenoma that stimulates excess cortisol secretion from the adrenal glands [1]. Its incidence is 1.2–2.4 per million and prevalence is ~40 per million population [2, 3]. CD accounts for 75–80 % of cases with ACTH-dependent Cushing’s syndrome (CS).
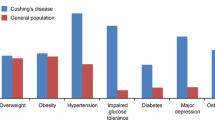
The clinical presentation of CD can be highly variable and the diagnosis can often be challenging in cases with mild or cyclic hypercortisolism, especially given the overlap in symptoms in individuals with and without the disorder [1]. This may delay the diagnosis for 2–4 years [3–5]. Hypercortisolemia is associated with increased morbidity and mortality. Resection of the adenoma via transsphenoidal surgery (TSS) remains the optimal treatment. Medical therapy (steroidogenesis inhibitors, agents that decrease ACTH levels and glucocorticoid receptor antagonists) with or without pituitary radiotherapy may be needed [1] to normalize cortisol and/or its action. In addition, management of comorbidities (Table 1) is important because of increased cardiovascular risk despite remission [3–8]. We here review the comorbidities associated with CD and their impact on quality of life and mortality.
Mortality in CD
Multiple studies show that the standardized mortality ratio (SMR) is increased in CD (1.7–4.8) [3–10], especially in patients with persistent hypercortisolism (3.7–4.2) compared to those in remission (1.8–3.17). SMR is higher in CD patients compared to those undergoing TSS for non-functioning pituitary macroadenomas (NFPA) (2.39 vs. 1.24) [6].
Cardiovascular and cerebrovascular events are the most common cause of death in CD [6, 9]. In one study, CD patients died of cardiovascular disease (n = 4), cerebrovascular disease (n = 1), malignancy (n = 1), and infectious diseases (n = 1). The average age at death (62.4 years) was significantly lower than that seen in the general Dutch population [6]. Thus, normalization of cortisol levels improves but does not normalize mortality compared to the general population.
Comorbidities in CD (Table 1)
Cardiovascular disease
Prolonged exposure to hypercortisolemia is associated with multiple cardiovascular risk factors that strongly impact morbidity and mortality. These risk factors include visceral adiposity, systemic arterial hypertension, impaired glucose tolerance, dyslipidemia and hypercoagulability [1].
Cardiovascular risk is increased in CD patients even 5 years after remission [11, 12]. Compared with controls, CD patients had significantly higher waist-to-hip ratio, diastolic blood pressure (BP), oral glucose tolerance test (OGTT)—stimulated glucose and insulin levels, total/HDL cholesterol ratio and fibrinogen levels. These patients had increased atherosclerotic carotid artery changes, with higher intima-media thickness and lower distensibility coefficient on ultrasound imaging. Atherosclerotic plaques were found in 26.7 % of CD patients and <4 % of controls [11].
Both children and adults in biochemical remission have abnormally high total body fat and visceral to subcutaneous fat ratios after a follow-up of 3–11 years [13, 14]. The adult patients also have increased inflammatory markers and decreased adiponectin levels [14]. This persistence of central adiposity and an unfavorable adipokine profile may link metabolic alterations and cardiovascular morbidity in CD.
Hypertension occurs in 55–85 % of CD patients [11, 15, 16], but resolves after remission in only 44–75 % [14, 16, 17]. Persistent hypertension despite remission may be related to microvessel remodeling and/or presence of underlying essential hypertension. Older age, longer duration of exposure to hypercortisolemia and a longer duration of untreated hypertension are associated with persistently elevated BP after biochemical cure [16–18]. This underlies the importance of early control of hypertension even while establishing the diagnosis of CD.
Cardiac structural and functional alterations, including left ventricular hypertrophy, concentric remodeling, and reduced mid-wall systolic performance with diastolic dysfunction, occurs in active CD [19–21]. They are more severe in hypertensive patients, suggesting an interaction between the deleterious effects of hypertension and cortisol excess per se [19]. With remission of hypercortisolemia, cardiac alterations significantly improve [19, 21] but may not normalize [19].
Patients with CD have an increased predisposition to thromboembolic events, especially in the perioperative period, likely related to a procoagulative phenotype due to cortisol excess [22–24]. In one systematic review, the incidence of venous thromboembolism (VTE) in CS was 2.5–3.1 per 1000 persons/year [25] compared to 1.0–2.0 per 1000 persons/year in the general population [26]. In a multicenter retrospective cohort study of 473 CS patients (360 CD), the incidence of VTE before treatment of hypercortisolemia was 14.6 per 1000 persons/year [27]. Additionally, the incidence of postoperative VTE was significantly greater compared to patients who underwent TSS for NFPAs (3.4 vs. 0 %) with the highest risk being in the first 2 months after surgery [27]. However, biochemical remission of hypercortisolemia with medical therapy (80 days) did not normalize hemostatic and fibrinolytic parameters [23]. At present, the duration and severity of risk for VTE following remission of hypercortisolism remain uncertain. There are no clear guidelines on the dose and duration of thromboprophylaxis either before or after surgery. Future long-term studies are needed to evaluate these issues.
Insulin resistance, impaired glucose tolerance and diabetes mellitus
Insulin resistance is a well-known complication of CD. Glucocorticoid excess induces the expression of several key gluconeogenesis enzymes resulting in increased glucose production. It also impairs insulin sensitivity via direct interference with the insulin receptor signaling pathway as well as indirectly, through the stimulation of lipolysis and proteolysis [28]. The prevalence of overt diabetes mellitus is 20–47 % and that of impaired glucose tolerance is 21–64 % in CD [1, 11, 15, 16]. This may be an underestimation as not all patients undergo an OGTT, which is needed to diagnose impaired glucose tolerance when the fasting glucose is normal. The homeostatic model assessment (HOMA) shows decreased insulin sensitivity index (ISI) and increased insulin resistance in CD. Normal weight, overweight and obese patients have similar values, suggesting that these alterations result from hypercortisolemia and not obesity [14, 16]. Age, genetic predisposition and lifestyle also contribute significantly to the development of glucose intolerance in CD [29].
Dyslipidemia
Lipid abnormalities (increased total and LDL cholesterol, triglycerides and total/HDL cholesterol ratio) occur in 38–71 % of CD patients [11, 16]. The multifactorial pathogenic mechanisms include direct and indirect actions of cortisol on lipolysis, free fatty acid production and turnover, very-low density lipoprotein synthesis and fatty accumulation in the liver. Insulin resistance, growth hormone deficiency and hypogonadism are also contributing factors [30]. Given the increased cardiovascular morbidity and mortality in CD, aggressive treatment of dyslipidemia is recommended [31]. Surgical remission is often associated with normalization of lipid abnormalities.
Management of dyslipidemia can be challenging in patients on medical therapy. Mitotane significantly increases cholesterol levels [32]. Drug interactions are common with ketoconazole, a potent inhibitor of cytochrome P450 3A4 (CYP3A4). Concomitant use of ketoconazole and certain statins (e.g. simvastatin and atorvastatin) that undergo metabolism via CYP3A4 can lead to significantly higher plasma levels of the statin, thus increasing the risk of complications and side-effects. In this setting, preference should be given to agents that are not metabolized by the CYP3A4 pathway (e.g. pravastatin) with close monitoring of liver function tests [31, 33].
Osteoporosis, fractures and nephrolithiasis
The prevalence of fractures and osteoporosis as assessed by dual energy X-ray absorptiometry (DXA) has been reported to be 15–50 and 38–50 % respectively [5, 34–36], and may be a presenting feature of CD. These rates are likely an underestimation as not all CD patients undergo DXA scans, and asymptomatic vertebral and rib fractures can remain undiagnosed. The prevalence of vertebral and rib fractures is significantly higher in men compared to women [5]. This results from direct and indirect effects of glucocorticoids on bone, including decreased osteoblastic and increased osteoclastic activity, hypogonadism, growth hormone deficiency, reduced intestinal calcium absorption and increased urinary calcium excretion [37].
Normalization of cortisol levels has been associated with reversal of glucocorticoid-induced osteoporosis after 6–9 years [38, 39]. However, other shorter studies with 2–3 years follow-up show incomplete recovery of bone mineral density and quality of bone after remission [36, 40].
Nephrolithiasis is reported in 50 % of active CD patients compared to 6.5 % in age- and gender-matched controls. This is accompanied by hypercalciuria, hypocitraturia, hyperuricosuria and hyperoxalaturia [41], with amelioration of this lithogenic excretion pattern after remission. During remission, the prevalence of nephrolithiasis decreases (27.3 %) but remains higher compared to controls [41].
Psychiatric dysfunction
Most (54–85 %) patients with CD have some psychiatric disturbance. Depression and irritability are most common emotional lability, mania, paranoia, acute psychosis, anxiety and panic attacks also occur [42, 43]. Many of these symptoms persist in the first few years after successful surgery [44–46]. However, overall psychopathology decreased significantly from 66.7 % during active CD to 53.6 % at 3 months, 36 % at 6 months and 24.1 % at 12 months after remission [44]. In another study, CD patients achieving long-term (mean 11 years) remission showed an increased prevalence of psychopathology and maladaptive personality traits compared to those with NFPAs and matched controls. These observations suggest irreversible effects of cortisol excess on the central nervous system rather than an effect of pituitary tumors or their treatment [46].
Cognitive impairment
Chronic hypercortisolism is associated with cognitive dysfunction including impairment of memory, visual and spatial information, reasoning, verbal learning and language performance [47–53]. The hippocampus, amygdala and the cerebral cortex, structures important for cognitive and emotional functioning, are rich in glucocorticoid receptors and presumably particularly vulnerable to hypercortisolemia. Hippocampal atrophy was seen in 27 % of 12 active CS patients and correlated with performance on cognitive tests [47]. Hippocampal formation volume increased by up to 10 % in 22 CD patients ~16 months after surgical remission of hypercortisolemia [51]. In a case control study, apparent loss of brain volume was noted in 86 % of CD patients (14 % mild, 43 % moderate, 29 % severe), 100 % of adrenal CS patients and 10 % of controls. Normalization of cortisol levels was associated with partial reversal of cerebral atrophy [50]. van der Werff et al. reported widespread reductions in fractional anisotropy (marker of tissue microstructural organization) and changes of white matter integrity in 22 CD patients in long-term remission (mean 11.9 years) compared with matched controls, with changes in the uncinate fasciculus being related to the severity of depressive symptoms [53]. Recently Tiemensma et al. [52] reported significantly lower scores on cognitive assessment tests reflecting impaired memory and executive functioning, in 74 CD patients despite long-term remission (mean 13 years) compared with age-, gender- and education-matched controls. These data suggest that the structural changes in the brain induced by hypercortisolemia are at least partially reversible with normalization of cortisol levels. However, there may be a delay of several years from eucortisolemia to the resolution of cognitive deficits, and occasionally these may be irreversible.
Health related quality of life (HRQoL)
HRQoL is significantly impaired in patients with CD [54–57]. In one study, the issues most often reported by CS patients with regards to the impact of the disease on their everyday life were fatigue and weakness (85 %), interference with family life and relationship with their partners (80 %), changes in physical appearance (63 %), emotional instability (61 %), impaired school/work performance (56 %), cognitive problems (49 %), depression (32 %) and sleeping difficulties (12 %) [51]. A prospective study of HRQol using the short-form 36 (SF-36) survey in 23 CD patients before and after TSS demonstrated that active CD was associated with low physical and mental summary scores with partial resolution after surgical remission of hypercortisolemia [55]. In a recent cross-sectional study of 102 patients with surgically treated CD (mean time since surgery 7.4 years), 92 % met the criteria for biochemical remission but only 80 % felt that they had been cured. These findings reflect the discordance between biochemical and self-assessed disease status and its impact on HRQoL in CD patients [57]. In a recent systematic review of quality of life impairments in patients with pituitary adenomas, biochemical remission of CD was associated with the smallest improvement in QoL measurements compared to those with other pituitary adenomas. Somatic factors (including hypopituitarism), psychological factors (illness perceptions), and health care environment (rural vs. urban) were identified as influencing factors [58].
Economic burden of CD
Swearingen et al. analyzed data from a US administrative claims database for the period 2004–2008 with data on hospital admissions, physician visits, emergency room visits and medication use. The total health care costs for CD patients were fourfold greater than age- and gender-matched population controls and twofold higher than age- and gender-matched patients with NFPAs. Annual outpatient costs decreased significantly after surgical remission. In contrast, there was a significant increase in postoperative health care costs in those patients not in remission [59]. A more recent analysis of health care costs in CD based on 2010 US claims data estimated the total cost of care at $35,000/year [60]. These data along with the decreased working ability of CD patients and increase in sick leave highlight the significant economic burden placed on the patients as well as on the health care resources in CD.
Conclusion
Although rare, CD is associated with a significant clinical, social, economic and quality of life burden. Uncontrolled hypercortisolemia is associated with metabolic, cardiovascular, cognitive and psychological alterations leading to increased mortality. Normalization of cortisol levels leads to significant improvement in these parameters and decreased mortality. However a growing body of evidence indicates that many risks and morbidities, in particular cardiovascular risk, persist for several years after remission. Larger studies with long-term follow-up are needed to elucidate this further. Early diagnosis and control of hypercortisolemia, aggressive management of comorbidities in a multidisciplinary setting and long-term follow-up are essential for optimal recovery in CD.
References
Newell-Price J, Bertagna X, Grossman AB, Nieman LK (2006) Cushing’s syndrome. Lancet 367:1605–1617
Lindholm J, Juul S, Jorgenson JO, Astrup J, Bjerre P, Feldt-Rasmussen U, Hagen C, Jorgensen J, Kosteljanetz M, Kristensen L, Laurberg P, Schmidt K, Weeke J (2001) Incidence and late prognosis of Cushing’s syndrome: a population based study. J Clin Endocrinol Metab 86:117–123
Etxabe J, Vazquez JA (1994) Morbidity and mortality in Cushing’s disease: an epidemiological approach. Clin Endocrinol (Oxf) 40:479–484
Bolland MJ, Holdaway IM, Berkeley JE, Lim S, Dransfield WJ, Conaglen JV, Croxson MS, Gamble GD, Hunt PJ, Toomath RJ (2011) Mortality and morbidity in Cushing’s syndrome in New Zealand. Clin Endocrinol 75:436–442
Valassi E, Santos A, Yaneva M, Toth M, Strasburger CJ, Chanson P, Wass JA, Chabre O, Pfeifer M, Feelders RA, Tsagarakis S, Trainer PJ, Franz H, Zopf K, Zacharieva S, Lamberts SW, Tabarin A, Webb SM (2011) The European registry on Cushing’s syndrome: 2-year experience. Baseline, demographic and clinical characteristics. Eur J Endocrinol 165:383–392
Dekkers OM, Biermasz NR, Pereira AM, Roelfsema F, van Aken MO, Voormolen JH, Romijn JA (2007) Mortality in patients treated for Cushing’s disease is increased compared to patients treated for nonfunctioning pituitary macroadenomas. J Clin Endocrinol Metab 92:976–981
Feelders RA, Pulgar SJ, Kempel A, Pereira AM (2012) The burden of Cushing’s disease: clinical and health-related quality of life aspects. Eur J Endocrinol 167(3):311–326
Clayton RN, Raskauskiene D, Reulen RC, Jones PW (2011) Mortality and morbidity in Cushing’s disease over 50 years in Stokeon-Trent, UK: audit and meta-analysis of literature. J Clin Endocrinol Metab 96:632–642
Hassan-Smith ZK, Sherlock M, Reulen RC, Arlt W, Ayuk J, Toogood AA, Cooper MS, Johnson AP, Stewart PM (2012) Outcome of Cushing’s disease following transsphenoidal surgery in a single center over 20 years. J Clin Endocrinol Metab 97:1194–1201
Graversen D, Vestergaard P, Stochholm K, Gravholt CH, Jørgensen JO (2012) Mortality in Cushing’s syndrome: a systematic review and meta-analysis. Eur J Intern Med 23(3):278–282
Colao A, Pivonello R, Spiezia S, Faggiano A, Ferone D, Filippella M, Marzullo P, Cerbone G, Siciliani M, Lombardi G (1999) Persistence of increased cardiovascular risk in patients with Cushing’s disease after five years of successful cure. J Clin Endocrinol Metab 84(8):664–672
Neary NM, Booker OJ, Abel BS, Matta JR, Muldoon N, Sinaii N, Pettigrew RI, Nieman LK, Gharib AM (2013) Hypercortisolism is associated with increased coronary arterial atherosclerosis: analysis of noninvasive coronary angiography using multidetector computerized tomography. J Clin Endocrinol Metab 98:2045–2052
Leong GM, Abad V, Charmandari E, Reynolds JC, Hill S, Chrousos GP, Nieman LK (2007) Effects of child- and adolescent-onset endogenous Cushing syndrome on bone mass, body composition, and growth: a 7-year prospective study into young adulthood. J Bone Miner Res 22(1):110–118
Barahona MJ, Sucunza N, Resmini E, Fernández-Real JM, Ricart W, Moreno-Navarrete JM, Puig T, Farrerons J, Webb SM (2009) Persistent body fat mass and inflammatory marker increases after long-term cure of Cushing’s syndrome. J Clin Endocrinol Metab 94:3365–3371
Faggiano A, Pivonello R, Spiezia S, De Martino MC, Filippella M, Di Somma C, Lombardi G, Colao A (2003) Cardiovascular risk factors and common carotid artery caliber and stiffness in patients with Cushing’s disease during active disease and 1 year after disease remission. J Clin Endocrinol Metab 88(6):2527–2533
Mancini T, Kola B, Mantero F, Boscaro M, Arnaldi G (2004) High cardiovascular risk in patients with Cushing’s syndrome according to 1999 WHO/ISH guidelines. Clin Endocrinol (Oxf) 61(6):768–777
Fallo F, Sonino N, Barzon L, Pistorello M, Pagotto U, Paoletta A, Boscaro M (1996) Effect of surgical treatment on hypertension in Cushing’s syndrome. Am J Hypertens 9(1):77–80
Gómez RM, Albiger NM, Díaz AG, Moncet D, Pitoia FA, Bruno OD (2007) Effect of hypercortisolism control on high blood pressure in Cushing’s syndrome. Medicina (B Aires) 67:439–444
Toja PM, Branzi G, Ciambellotti F, Radaelli P, De Martin M, Lonati LM, Scacchi M, Parati G, Cavagnini F, Pecori Giraldi F (2012) Clinical relevance of cardiac structure and function abnormalities in patients with Cushing’s syndrome before and after cure. Clin Endocrinol (Oxf) 76:332–338
Muiesan ML, Lupia M, Salvetti M, Grigoletto C, Sonino N, Boscaro M, Rosei EA, Mantero F, Fallo F (2003) Left ventricular structural and functional characteristics in Cushing’s syndrome. J Am Coll Cardiol 41:2275–2279
Pereira AM, Delgado V, Romijn JA, Smit JW, Bax JJ, Feelders RA (2010) Cardiac dysfunction is reversed upon successful treatment of Cushing’s syndrome. Eur J Endocrinol 162:331–340
Fatti LM, Bottasso B, Invitti C, Coppola R, Cavagnini F, Mannucci PM (2000) Markers of activation of coagulation and fibrinolysis in patients with Cushing’s syndrome. J Endocrinol Invest 23:145–150
van der Pas R, de Bruin C, Leebeek FW, de Maat MP, Rijken DC, Pereira AM, Romijn JA, Netea-Maier RT, Hermus AR, Zelissen PM, de Jong FH, van der Lely AJ, de Herder WW, Lamberts SW, Hofland LJ, Feelders RA (2012) The hypercoagulable state in Cushing’s disease is associated with increased levels of procoagulant factors and impaired fibrinolysis, but is not reversible after short-term biochemical remission induced by medical therapy. J Clin Endocrinol Metab 97:1303–1310
Manetti L, Bogazzi F, Giovannetti C, Raffaelli V, Genovesi M, Pellegrini G, Ruocco L, Iannelli A, Martino E (2010) Changes in coagulation indexes and occurrence of venous thromboembolism in patients with Cushing’s syndrome: results from a prospective study before and after surgery. Eur J Endocrinol 163:783–791
Van Zaane B, Nur E, Squizzato A, Dekkers OM, Twickler MT, Fliers E, Gerdes VE, Büller HR, Brandjes DP (2009) Hypercoagulable state in Cushing’s syndrome: a systematic review. J Clin Endocrinol Metab 94:2743–2750
Silverstein MD, Heit JA, Mohr DN, Petterson TM, O’Fallon WM, Melton LJ III (1998) Trends in the incidence of deep vein thrombosis and pulmonary embolism: a 25-year population-based study. Arch Intern Med 158:585–593
Stuijver DJ, van Zaane B, Feelders RA, Debeij J, Cannegieter SC, Hermus AR, van den Berg G, Pereira AM, de Herder WW, Wagenmakers MA, Kerstens MN, Zelissen PM, Fliers E, Schaper N, Drent ML, Dekkers OM, Gerdes VE (2011) Incidence of venous thromboembolism in patients with Cushing’s syndrome: a multicenter cohort study. J Clin Endocrinol Metab 96:3525–3532
Pivonello R, De Leo M, Vitale P, Cozzolino A, Simeoli C, De Martino MC, Lombardi G, Colao A (2010) Pathophysiology of diabetes mellitus in Cushing’s syndrome. Neuroendocrinology 92(Suppl 1):77–81
Giordano C, Guarnotta V, Pivonello R, Amato MC, Simeoli C, Ciresi A, Cozzolino A, Colao A (2013) Is diabetes in Cushing’s syndrome only a consequence of hypercortisolism? Eur J Endocrinol 170:311–319
Arnaldi G, Scandali VM, Trementino L, Cardinaletti M, Appolloni G, Boscaro M (2010) Pathophysiology of dyslipidemia in Cushing’s syndrome. Neuroendocrinology 92(Suppl 1):86–90
Greenman Y (2010) Management of dyslipidemia in Cushing’s syndrome. Neuroendocrinology 92(Suppl 1):91–95
Maher VM, Trainer PJ, Scoppola A, Anderson JV, Thompson GR, Besser GM (1992) Possible mechanism and treatment of o, p’DDD-induced hypercholesterolaemia. Q J Med 84:671–679
Neuvonen PJ, Niemi M, Backman JT (2006) Drug interactions with lipid-lowering drugs: mechanisms and clinical relevance. Clin Pharmacol Ther 80:565–581
Ohmori N, Nomura K, Ohmori K, Kato Y, Itoh T, Takano K (2003) Osteoporosis is more prevalent in adrenal than in pituitary Cushing’s syndrome. Endocr J 50:1–7
Trementino L, Appolloni G, Ceccoli L, Marcelli G, Concettoni C, Boscaro M, Arnaldi G (2014) Bone complications in patients with Cushing’s syndrome: looking for clinical, biochemical, and genetic determinants. Osteoporos Int 25:913–921
Di Somma C, Pivonello R, Loche S, Faggiano A, Klain M, Salvatore M, Lombardi G, Colao A (2003) Effect of 2 years of cortisol normalization on the impaired bone mass and turnover in adolescent and adult patients with Cushing’s disease: a prospective study. Clin Endocrinol (Oxf) 58:302–308
Shaker JL, Lukert BP (2005) Osteoporosis associated with excess glucocorticoids. Endocrinol Metab Clin N Am 34:341–356
Kristo C, Jemtland R, Ueland T, Godang K, Bollerslev J (2006) Restoration of the coupling process and normalization of bone mass following successful treatment of endogenous Cushing’s syndrome: a prospective, long-term study. Eur J Endocrinol 154:109–118
Manning PJ, Evans MC, Reid IR (1992) Normal bone mineral density following cure of Cushing’s syndrome. Clin Endocrinol (Oxf) 36:229–234
Hermus AR, Smals AG, Swinkels LM, Huysmans DA, Pieters GF, Sweep CF, Corstens FH, Kloppenborg PW (1995) Bone mineral density and bone turnover before and after surgical cure of Cushing’s syndrome. J Clin Endocrinol Metab 80:2859–2865
Faggiano A, Pivonello R, Melis D, Filippella M, Di Somma C, Petretta M, Lombardi G, Colao A (2003) Nephrolithiasis in Cushing’s disease: prevalence, etiopathogenesis, and modification after disease cure. J Clin Endocrinol Metab 88:2076–2080
Dorn LD, Burgess ES, Dubbert B, Simpson SE, Friedman T, Kling M, Gold PW, Chrousos GP (1995) Psychopathology in patients with endogenous Cushing’s syndrome: ‘atypical’ or melancholic features. Clin Endocrinol (Oxf) 43:433–442
Sonino N, Fava GA (2001) Psychiatric disorders associated with Cushing’s syndrome. Epidemiology, pathophysiology and treatment. CNS Drugs 15:361–373
Kelly WF, Kelly MJ, Faragher B (1996) A prospective study of psychiatric and psychological aspects of Cushing’s syndrome. Clin Endocrinol (Oxf) 45:715–720
Dorn LD, Burgess ES, Friedman TC, Dubbert B, Gold PW, Chrousos GP (1997) The longitudinal course of psychopathology in Cushing’s syndrome after correction of hypercortisolism. J Clin Endocrinol Metab 82:912–919
Tiemensma J, Biermasz NR, Middelkoop HA, van der Mast RC, Romijn JA, Pereira AM (2010) Increased prevalence of psychopathology and maladaptive personality traits after long-term cure of Cushing’s disease. J Clin Endocrinol Metab 95:E129–E141
Starkman MN, Gebarski SS, Berent S, Schteingart DE (1992) Hippocampal formation volume, memory dysfunction, and cortisol levels in patients with Cushing’s syndrome. Biol Psychiatry 32:756–765
Hook JN, Giordani B, Schteingart DE, Guire K, Giles J, Ryan K, Gebarski SS, Langenecker SA, Starkman MN (2007) Patterns of cognitive change over time and relationship to age following successful treatment of Cushing’s disease. J Int Neuropsychol Soc 13:21–29
Ragnarsson O, Berglund P, Eder DN, Johannsson G (2012) Long-term cognitive impairments and attentional deficits in patients with Cushing’s disease and cortisol-producing adrenal adenoma in remission. J Clin Endocrinol Metab 97:E1640–E1648
Bourdeau I, Bard C, Noël B, Leclerc I, Cordeau MP, Bélair M, Lesage J, Lafontaine L, Lacroix A (2002) Loss of brain volume in endogenous Cushing’s syndrome and its reversibility after correction of hypercortisolism. J Clin Endocrinol Metab 87:1949–1954
Starkman MN, Giordani B, Gebarski SS, Berent S, Schork MA, Schteingart DE (1999) Decrease in cortisol reverses human hippocampal atrophy following treatment of Cushing’s disease. Biol Psychiatry 46:1595–1602
Tiemensma J, Kokshoorn NE, Biermasz NR, Keijser BJ, Wassenaar MJ, Middelkoop HA, Pereira AM, Romijn JA (2010) Subtle cognitive impairments in patients with long-term cure of Cushing’s disease. J Clin Endocrinol Metab 95:2699–2714
van der Werff SJ, Andela CD, Nienke Pannekoek J, Meijer OC, van Buchem MA, Rombouts SA, van der Mast RC, Biermasz NR, Pereira AM, van der Wee NJ (2014) Widespread reductions of white matter integrity in patients with long-term remission of Cushing’s disease. Neuroimage Clin 4:659–667
Gotch PM (1994) Cushing’s syndrome from the patient’s perspective. Endocrinol Metab Clin N Am 23:607–617
Lindsay JR, Nansel T, Baid S, Gumowski J, Nieman LK (2006) Long-term impaired quality of life in Cushing’s syndrome despite initial improvement after surgical remission. J Clin Endocrinol Metab 91:447–453
van Aken MO, Pereira AM, Biermasz NR, van Thiel SW, Hoftijzer HC, Smit JW, Roelfsema F, Lamberts SW, Romijn JA (2005) Quality of life in patients after long-term biochemical cure of Cushing’s disease. J Clin Endocrinol Metab 90:3279–3286
Carluccio A, Sundaram NK, Chablani S, Amrock LG, Lambert JK, Post KD, Geer EB (2014) Predictors of quality of life in 102 patients with treated Cushing’s disease. Clin Endocrinol (Oxf). doi:10.1111/cen.12521
Andela CD, Scharloo M, Pereira AM, Kaptein AA, Biermasz NR (2015) Quality of life (QoL) impairments in patients with a pituitary adenoma: a systematic review of QoL studies. Pituitary. doi:10.1007/s11102-015-0636-7
Swearingen B, Wu N, Chen SY, Pulgar S, Biller BM (2011) Health care resource use and costs among patients with cushing disease. Endocr Pract 17:681–690
Broder MS, Neary MP, Chang E, Cherepanov D, Ludlam WH (2014) Burden of illness, annual healthcare utilization and costs associated with commercially insured patients with Cushing’s disease in the United States. Endocr Pract. doi:10.4158/EP14126.OR
Disclosures
LKN has received funding from Laboratoire-HRA Pharma as part of a Cooperative Research and Development Agreement to conduct research on the antiglucocorticoid agent, mifepristone. RAF has received research Grants from Novartis.
Conflict of interest
STS reports no conflict of interest in this work.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution License which permits any use, distribution, and reproduction in any medium, provided the original author(s) and the source are credited.
About this article
Cite this article
Sharma, S.T., Nieman, L.K. & Feelders, R.A. Comorbidities in Cushing’s disease. Pituitary 18, 188–194 (2015). https://doi.org/10.1007/s11102-015-0645-6
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11102-015-0645-6