Abstract
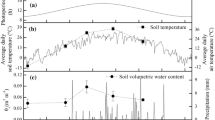
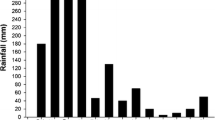
We studied the survival adaptation strategy of Sophora alopecuroides L. to habitat conditions in an arid desert riparian ecosystem. We examined the responses of heliotropic leaf movement to light conditions and their effects on plant photochemical performance. S. alopecuroides leaves did not show any observable nyctinastic movement but they presented sensitive diaheliotropic and paraheliotropic leaf movement in the forenoon and at midday. Solar radiation was a major factor inducing leaf movement, in addition, air temperature and vapour pressure deficit could also influence the heliotropic leaf movement in the afternoon. Both diaheliotropic leaf movement in the forenoon and paraheliotropic leaf movement at midday could help maintain higher photochemical efficiency and capability of light utilisation than fixed leaves. Paraheliotropic leaf movement at midday helped plants maintain a potentially higher photosynthetic capability and relieve a risk of photoinhibition. Our findings indicated the effective adaptation strategy of S. alopecuroides to high light, high temperature, and dry conditions in arid regions. This strategy can optimise the leaf energy balance and photochemical performance and ensure photosystem II function.
Similar content being viewed by others
Abbreviations
- D:
-
fraction of energy lost by thermal dissipation
- ETR:
-
electron transport rate
- F0 :
-
minimum fluorescence yield of the dark-adapted state
- F0′:
-
minimum fluorescence yield of the light-adapted state
- Fm :
-
maximum fluorescence yield of the dark-adapted state
- Fm′:
-
maximum fluorescence yield of the light-adapted state
- Fs :
-
steady-state fluorescence
- NPQ:
-
nonphotochemical quenching
- P:
-
fraction of energy allocated to PSII photochemistry
- qP :
-
photochemical quenching
- SEM:
-
scanning electron microscopy
- VPD:
-
vapor pressure deficit
- X:
-
excess excitation energy
- ΦPSII :
-
effective quantum yield of PSII in light-adapted leaves
References
Arena C., Vitale L., De Santo A.V.: Paraheliotropism in Robinia pseudocacia L.: an efficient strategy to optimise photosynthetic performance under natural environmental conditions. — Plant Biol. 10: 194–201, 2008.
Atta-ur-Rahman A., Choudhary M.I., Parvez K. et al.: Quinolizidine alkaloids from Sophora alopecuroides. — J. Nat. Prod. 63: 190–192, 2000.
Babani F., Lichtenthaler H.K.: Light-induced and age-dependent development of chloroplasts in etiolated barley leaves as visualized by determination of photosynthetic pigments, CO2 assimilation rates and different kinds of chlorophyll fluorescence ratios. — J. Plant Physiol. 148: 555–566, 1996.
Barker D.H., Adams III W.W.: The xanthophyll cycle and energy dissipation in differently oriented faces of the cactus Opuntia macrorhiza. — Oecologia 109: 353–361, 1997.
Berg V.S., Heuchelin S.: Leaf orientation of soybean seedlings. I. Effect of water potential and photosynthetic photon flux density on paraheliotropism. — Crop Sci. 30: 631–638, 1990.
Berg V.S., Hsiao T.C.: Solar tracking light avoidance induced by water stress in leaves of kidney bean seedlings in the field. — Crop Sci. 26: 980–986, 1986.
Bielenberg D.G., Miller J.D., Berg V.S.: Paraheliotropism in two Phaseolus species: combined effects of photon flux density and pulvinus temperature, and consequences for leaf gas exchange. — Environ. Exp. Bot. 49: 95–105, 2003.
Bilger W., Björkman O.: Role of the xanthophyll cycle in photoprotection elucidated by measurements of light-induced absorbance changes, fluorescence and photosynthesis in leaves of Hedera canariensis. — Photosynth. Res. 25: 173–185, 1990.
Caldas L.S., Lüttge U., Franco A.C., Haridasan M.: Leaf heliotropism in Pterodon pubescens, a woody legume from the brasilian cerrado. — Rev. Bras. Fisiol. Veg. 9: 1–7, 1997.
Demmig-Adams B., Adams, III W.W., Barker D.H. et al.: Using chlorophyll fluorescence to assess the fraction of absorbed light allocated to thermal dissipation of excess excitation. — Physiol. Plantarum 98: 253–264, 1996.
Ehleringer J.R., Forseth I.N.: Solar tracking by plants. — Science 210: 1094–1098, 1980.
Fu Q.A., Ehleringer J.R.: Heliotropic leaf movements in common beans controlled by air temperature. — Plant Physiol. 91: 1162–1167, 1989.
Fu Q.A., Ehleringer J.R.: Crop physiology and metabolism: Paraheliotropic leaf movements in common bean under different soil nutrient levels. — Crop Sci. 32: 1192–1196, 1992.
Genty B., Briantais J.M., Baker N.R.: The relationship between the quantum yield of photosynthetic electron transport and quenching of chlorophyll fluorescence. — Biochim. Biophys. Acta 990: 87–92, 1989.
Greer D.H., Laing W.A., Woolley D.H.: The effect of chloramphenicol on photoinhibition of photosynthesis and its recovery in intact kiwifruit (Actinidia deliciosa) leaves. — Aust. J. Plant Physiol. 20: 33–43, 1993.
Guo Q.L., Feng Q., Li J.L.: Environmental changes after ecological water conveyance in the lower reaches of Heihe River, northwest China. — Environ. Geol. 58: 1387–1396, 2009.
Habermann G., Ellsworth P.F.V., Cazoto J.L. et al.: Leaf paraheliotropism in Styrax camporum confers increased light use efficiency and advantageous photosynthetic responses rather than photoprotection. — Environ. Exp. Bot. 71: 10–17, 2011.
Habermann G., Machado S.R., Guimarães V.F., Rodrigues J.D.: Leaf heliotropism in Styrax camporum Pohl from the Brazilian cerrado — distinct gas exchange and leaf structure, but similar leaf temperature and water relations. — Braz. J. Plant Physiol. 20: 71–83, 2008.
Herbert T.J.: A latitudinal cline in leaf inclination of Dryas octopetala and implications for maximization of whole plant photosynthesis. — Photosynthetica 41: 631–633, 2003.
Ji X.B., Kang E.S., Chen R.S. et al.: The impact of the development of water resources on environment in arid inland river basins of Hexi region, Northwestern China. — Environ. Geol. 50: 793–801, 2006.
Jiang C.D., Gao H.Y., Zou Q. et al.: Leaf orientation, photorespiration and xanthophyll cycle protect young soybean leaves against high irradiance in field. — Environ. Exp. Bot. 55: 87–96, 2006.
Jurik T.W., Akey W.C.: Solar-tracking leaf movements in velvetleaf (Abutilon theophrasti). — Plant Ecol. 112: 93–99, 1994.
Kao W.Y., Forseth I.N.: Diurnal leaf movement, chlorophyll fluorescence and carbon assimilation in soybean grown under different nitrogen and water availabilities. — Plant Cell Environ. 15: 7037–7043, 1992.
Kato E., Nagano H., Yamamura S., Ueda M.: Synthetic inhibitor of leaf-closure that reveals the biological importance of leaf movement for the survival of leguminous plants. — Tetrahedron 59: 5909–5917, 2003.
Koller D.: The control of leaf orientation by light. — Photochem. Photobiol. 44: 819–826, 1986.
Koller D.: Light-driven leaf movements. — Plant Cell Environ. 13: 615–632, 1990.
Liu C.C., Welham C.V.J., Zhang X.Q., Wang R.Q.: Leaflet movement of Robinia pseudoacacia in response to a changing light environment. — J. Integr. Plant Biol. 49: 419–424, 2007.
Liu L.X., Xu S.M., Woo K.C.: Influence of leaf angle on photosynthesis and the xanthophyll cycle in the tropical tree species Acacia crassicarpa. — Tree Physiol. 23: 1255–1261, 2003.
Long S.P., Humphries S., Falkowski P.G.: Photoinhibition of photosynthesis in nature. — Annu. Rev. Plant Physiol. Plant Mol. Biol. 45: 633–662, 1994.
Minoru U., Takanori S., Yoshiyuki S., Shosuke Y.: The biological significance of leaf-movement, an approach using a synthetic inhibitor of leaf-closure. — Tetrahedron Lett. 43: 7545–7548, 2002.
Murata N., Takahashi S., Nishiyama Y., Allakhverdiev S.I.: Photoinhibition of photosystem II under environmental stress. — Biochim. Biophys. Acta 1767: 414–421, 2007.
Müller P., Li X.P., Niyogi K.K.: Non-photochemical quenching: A response to excess light energy. — Plant Physiol. 125 1558–1566, 2001.
Oxborough K., Baker N.R.: Resolving chlorophyll fluorescence images of photosynthetic efficiency into photochemical and non-photochemical components — calculation of qP and Fv/Fm without measuring Fo′. — Photosynth. Res. 54: 135–142, 1997.
Pastenes C., Pimentel P., Lillo J.: Leaf movements and photoinhibition in relation to water stress in field-grown beans. — J. Exp. Bot. 56: 425–433, 2005.
Powles S.B., Berry J.A., Björkman O.: Interaction between light and chilling temperature on the inhibition of photosynthesis in chilling sensitive plants. — Plant Cell Environ. 6: 117–23, 1983.
Proietti P., Palliotti A.: Contribution of adaxial and abaxial surfaces of olive leaves to photosynthesis. — Photosynthetica 33: 63–69, 1997.
Pu Q.L., Li Y., Yang J.: [Study on mass spectra of alkaloids from Sophora alopecuroides L.] — Acta Pharm. Sin. 22: 438–44, 1987. [In Chinese]
Richards R.A., Rawson H.M., Johnson D.A.: Glaucousness in wheat: its development and effect on water-use efficiency, gas exchange and photosynthetic tissue temperatures. — Aust. J. Plant Physiol. 13: 465–473, 1986.
Roháček K.: Chlorophyll fluorescence parameters: the definitions, photosynthetic meaning, and mutual relationships. — Photosynthetica 40: 13–29, 2002.
Rosa L.M., Forseth I.N.: Diurnal patterns of soybean leaf inclination angles and azimuthal orientation under different levels of ultraviolet B radiation. — Agr. Forest Meteorol. 78: 107–119, 1995.
Schreiber U.: Pulse-amplitude-modulation (PAM) fluorometry and saturation pulse method: an overview. — In: Papageorgiou G.C., Govindjee (ed.): Chlorophyll a Fluorescence: A Signature of Photosynthesis. Pp. 279–319. Springer, Netherlands 2004.
Schreiber U., Schliwa U., Bilger W.: Continuous recording of photochemical and non-photochemical fluorescence quenching with a new type of modulation fluorometer. — Photosynth. Res. 10: 51–62, 1986.
Siam A.M.J., Radoglou K.M., Noitsakis B.: Physiological and growth responses of three Mediterranean oak species to different water availability regimes. — J. Arid Environ. 72: 583–592, 2008.
Smith H.: Plants that track the sun. — Nature 308: 774–774, 1984.
Takahashi S., Murata N.: Glycerate-3-phosphate, produced by CO2 fixation in the Calvin cycle, is critical for the synthesis of the D1 protein of photosystem II. — Biochim. Biophys. Acta 1757:198–205, 2006.
Takahashi S., Murata N.: How do environmental stresses accelerate photoinhibition? — Trends Plant Sci. 13: 178–182, 2008.
Ueda M., Nakamura Y.: Chemical basis of plant leaf movement. — Plant Cell Physiol. 48: 900–907, 2007.
Wang G.X., Cheng G.D.: Water resource development and its influence on the environment in arid areas of China — the case of the Hei River basin. — J. Arid Environ. 43: 121–131, 1999.
Wang H.Y.; Li Y.X.; Dun L.L. et al.: Antinociceptive effects of matrine on neuropathic pain induced by chronic constriction injury. — Pharm. Biol. 51: 844–850, 2013.
Yu F., Berg V.S.: Control of paraheliotropism in two Phaseolus species. — Plant Physiol. 106: 1567–1573, 1994.
Yu T.F., Wang J., Bian L., Zhu H.Z.: [Effect of total alkaloids of kudouzi (Sophora alopecuroides L.) on tolerance to anoxia in mice.] — China J. Chin. Mat. Med. 18: 500–502, 511, 1993. [In Chinese]
Zhang S.R., Gao R.F.: Light induces leaf orientation and chloroplast movements of hybrid poplar clones. — Acta Ecol. Sin. 21: 68–74, 2001.
Zhang S.R., Ma K.P., Chen L.Z.: Photosynthetic gas exchange and leaflet movement of Robinia pseudoacacia in relation to changing light environments. — Acta Bot. Sin. 44: 858–863, 2002.
Zhang Y.C., Yu J.G., Wang P., Fu G.B.: Vegetation responses to integrated water management in the Ejina basin, northwest China. — Hydrol. Process. 25: 3448–3461, 2011.
Zhang Y.J., Feng Y.L., Feng Z.L., Cao K.F.: Morphological and physiological acclimation to growth light intensities in Pometica tomentosa. — J. Plant Physiol. Mol. Biol. 29: 206–214, 2003.
Zhang Y.L., Zhang G.Y., Feng J.S., Zhang W.F.: Leaf diaheliotropic movement can improve carbon gain and water use efficiency and not intensify photoinhibition in upland cotton (Gossypium hirsutum L.). — Photosynthetica 47: 609–615, 2009.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Acknowledgements: This work was supported by the National Natural Science Foundation of China (No. 91025025, No. 41101533).
Rights and permissions
About this article
Cite this article
Zhu, C.G., Chen, Y.N., Li, W.H. et al. Heliotropic leaf movement of Sophora alopecuroides L.: An efficient strategy to optimise photochemical performance. Photosynthetica 53, 231–240 (2015). https://doi.org/10.1007/s11099-015-0089-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11099-015-0089-2