Abstract
Purpose
The aim of this study was to investigate the depth-dependent intradermal immunogenicity of inactivated polio vaccine (IPV) delivered by depth-controlled microinjections via hollow microneedles (HMN) and to investigate antibody response enhancing effects of IPV immunization adjuvanted with CpG oligodeoxynucleotide 1826 (CpG) or cholera toxin (CT).
Methods
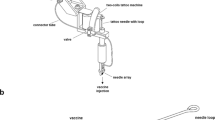
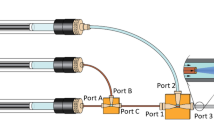
A novel applicator for HMN was designed to permit depth- and volume-controlled microinjections. The applicator was used to immunize rats intradermally with monovalent IPV serotype 1 (IPV1) at injection depths ranging from 50 to 550 μm, or at 400 μm for CpG and CT adjuvanted immunization, which were compared to intramuscular immunization.
Results
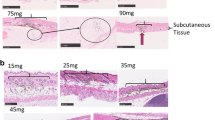
The applicator allowed accurate microinjections into rat skin at predetermined injection depths (50–900 μm), -volumes (1–100 μL) and -rates (up to 60 μL/min) with minimal volume loss (±1–2%). HMN-mediated intradermal immunization resulted in similar IgG and virus-neutralizing antibody titers as conventional intramuscular immunization. No differences in IgG titers were observed as function of injection depth, however IgG titers were significantly increased in the CpG and CT adjuvanted groups (7-fold).
Conclusion
Intradermal immunogenicity of IPV1 was not affected by injection depth. CpG and CT were potent adjuvants for both intradermal and intramuscular immunization, allowing effective vaccination upon a minimally-invasive single intradermal microinjection by HMN.




Similar content being viewed by others
Abbreviations
- CpG:
-
CpG oligodeoxynucleotide 1826
- CT:
-
Cholera toxin
- DC:
-
Dendritic cell
- DDC:
-
Dermal dendritic cell
- H&E:
-
Hematoxylin and eosin
- HMN:
-
Hollow microneedle
- IPV:
-
Inactivated polio vaccine
- IPV1:
-
Monovalent inactivated polio vaccine serotype 1
- LC:
-
Langerhans cell
- OPV:
-
Oral polio vaccine
- PBS:
-
Phosphate buffered saline
- TMB:
-
3,3′5,5′-tetramethylbenzidine
- VN:
-
Virus neutralizing
References
Kew OM, Sutter RW, de Gourville EM, Dowdle WR, Pallansch MA. Vaccine-derived polioviruses and the endgame strategy for global polio eradication. Annu Rev Microbiol. 2005;59:587–635.
World Health Organization W. Polio eradication and endgame strategic plan 2013–2018. Geneva: World Health Organization (WHO); 2013.
Okayasu H, Sutter RW, Jafari HS, Takane M, Aylward RB. Affordable inactivated poliovirus vaccine: strategies and progress. J Infect Dis. 2014;210 Suppl 1:S459–64.
Cadorna-Carlos J, Vidor E, Bonnet MC. Randomized controlled study of fractional doses of inactivated poliovirus vaccine administered intradermally with a needle in the Philippines. Int J Infect Dis. 2012;16(2):e110–6.
Mohammed AJ, AlAwaidy S, Bawikar S, Kurup PJ, Elamir E, Shaban MM, et al. Fractional doses of inactivated poliovirus vaccine in Oman. N Engl J Med. 2010;362(25):2351–9.
Soonawala D, Verdijk P, Wijmenga-Monsuur AJ, Boog CJ, Koedam P, Visser LG, et al. Intradermal fractional booster dose of inactivated poliomyelitis vaccine with a jet injector in healthy adults. Vaccine. 2013;31(36):3688–94.
van der Maaden K, Jiskoot W, Bouwstra J. Microneedle technologies for (trans)dermal drug and vaccine delivery. J Control Release. 2012;161(2):645–55.
van der Maaden K, Trietsch SJ, Kraan H, Varypataki EM, Romeijn S, Zwier R, et al. Novel hollow microneedle technology for depth-controlled microinjection-mediated dermal vaccination: a study with polio vaccine in rats. Pharm Res. 2014;31(7):1846–54.
van der Maaden K, Sekerdag E, Schipper P, Kersten G, Jiskoot W, Bouwstra JA. Layer-by-layer assembly of inactivated poliovirus and N-trimethyl chitosan on pH-sensitive microneedles for dermal vaccination. Langmuir. 2015.
Kraan H, Ploemen I, van de Wijdeven G, Que I, Lowik C, Kersten G, et al. Alternative delivery of a thermostable inactivated polio vaccine. Vaccine. 2015;33(17):2030–7.
Kouiavskaia D, Mirochnitchenko O, Dragunsky E, Kochba E, Levin Y, Troy S, et al. Intradermal inactivated poliovirus vaccine: a preclinical dose-finding study. J Infect Dis. 2015;211(9):1447–50.
Edens C, Dybdahl-Sissoko NC, Weldon WC, Oberste MS, Prausnitz MR. Inactivated polio vaccination using a microneedle patch is immunogenic in the rhesus macaque. Vaccine. 2015.
Troy SB, Kouiavskaia D, Siik J, Kochba E, Beydoun H, Mirochnitchenko O, et al. Comparison of the immunogenicity of various booster doses of inactivated polio vaccine delivered intradermally versus intramuscularly to HIV-infected adults. J Infect Dis. 2015;211(12):1969–76.
Combadiere B, Liard C. Transcutaneous and intradermal vaccination. Hum Vaccin. 2011;7(8):811–27.
Hawken J, Troy SB. Adjuvants and inactivated polio vaccine: a systematic review. Vaccine. 2012;30(49):6971–9.
Auerswald W, Juergenssen O. On the problem of antibody formation against poliomyelitis in small children. Studies on the effect of an adjuvant vaccine with antigen components against poliomyelitis, diphtheria, pertussis and tetanus. Wien Med Wochenschr. 1960;110:646–8.
Li RC, Li FX, Li YP, Hou QM, Li CG, Li YN, et al. Immunogenicity and safety of a pentavalent acellular pertussis combined vaccine including diphtheria, tetanus, inactivated poliovirus and conjugated Haemophilus Influenzae type b polysaccharide for primary vaccination at 2, 3, 4 or 3, 4, 5 months of age in infants in China. Vaccine. 2011;29(10):1913–20.
Yeh SH, Ward JI, Partridge S, Marcy SM, Lee H, Jing J, et al. Safety and immunogenicity of a pentavalent diphtheria, tetanus, pertussis, hepatitis B and polio combination vaccine in infants. Pediatr Infect Dis J. 2001;20(10):973–80.
Guerra FA, Blatter MM, Greenberg DP, Pichichero M, Noriega FR, Study G. Pentacel. Safety and immunogenicity of a pentavalent vaccine compared with separate administration of licensed equivalent vaccines in US infants and toddlers and persistence of antibodies before a preschool booster dose: a randomized, clinical trial. Pediatrics. 2009;123(1):301–12.
Gupta RK. Aluminum compounds as vaccine adjuvants. Adv Drug Deliv Rev. 1998;32(3):155–72.
Albrecht P, van Steenis G, van Wezel AL, Salk J. Standardization of poliovirus neutralizing antibody tests. Rev Infect Dis. 1984;6 Suppl 2:S540–4.
Westdijk J, Koedam P, Barro M, Steil BP, Collin N, Vedvick TS, et al. Antigen sparing with adjuvanted inactivated polio vaccine based on Sabin strains. Vaccine. 2013;31(9):1298–304.
Martanto W, Moore JS, Kashlan O, Kamath R, Wang PM, O’Neal JM, et al. Microinfusion using hollow microneedles. Pharm Res. 2006;23(1):104–13.
Wang PM, Cornwell M, Hill J, Prausnitz MR. Precise microinjection into skin using hollow microneedles. J Invest Dermatol. 2006;126(5):1080–7.
Roxhed N, Samel B, Nordquist L, Griss P, Stemme G. Painless drug delivery through microneedle-based transdermal patches featuring active infusion. IEEE Trans Biomed Eng. 2008;55(3):1063–71.
Hafeli UO, Mokhtari A, Liepmann D, Stoeber B. In vivo evaluation of a microneedle-based miniature syringe for intradermal drug delivery. Biomed Microdevices. 2009;11(5):943–50.
Burton SA, Ng CY, Simmers R, Moeckly C, Brandwein D, Gilbert T, et al. Rapid intradermal delivery of liquid formulations using a hollow microstructured array. Pharm Res. 2011;28(1):31–40.
Kim YC, Park JH, Prausnitz MR. Microneedles for drug and vaccine delivery. Adv Drug Deliv Rev. 2012;64(14):1547–68.
Roxhed N, Griss P, Stemme G. Membrane-sealed hollow microneedles and related administration schemes for transdermal drug delivery. Biomed Microdevices. 2008;10(2):271–9.
Hirschberg HJ, van Riet E, Oosterhoff D, Bouwstra JA, Kersten GF. Animal models for cutaneous vaccine delivery. Eur J Pharm Sci. 2015;71:112–22.
Resik S, Tejeda A, Lago PM, Diaz M, Carmenates A, Sarmiento L, et al. Randomized controlled clinical trial of fractional doses of inactivated poliovirus vaccine administered intradermally by needle-free device in Cuba. J Infect Dis. 2010;201(9):1344–52.
Resik S, Tejeda A, Sutter RW, Diaz M, Sarmiento L, Alemani N, et al. Priming after a fractional dose of inactivated poliovirus vaccine. N Engl J Med. 2013;368(5):416–24.
Anand A, Zaman K, Estivariz CF, Yunus M, Gary HE, Weldon WC, et al. Early priming with inactivated poliovirus vaccine (IPV) and intradermal fractional dose IPV administered by a microneedle device: a randomized controlled trial. Vaccine. 2015;33(48):6816–22.
Estivariz CF, Jafari H, Sutter RW, John TJ, Jain V, Agarwal A, et al. Immunogenicity of supplemental doses of poliovirus vaccine for children aged 6–9 months in Moradabad, India: a community-based, randomised controlled trial. Lancet Infect Dis. 2012;12(2):128–35.
Resik S, Tejeda A, Mach O, Fonseca M, Diaz M, Alemany N, et al. Immune responses after fractional doses of inactivated poliovirus vaccine using newly developed intradermal jet injectors: a randomized controlled trial in Cuba. Vaccine. 2015;33(2):307–13.
Scheiermann J, Klinman DM. Clinical evaluation of CpG oligonucleotides as adjuvants for vaccines targeting infectious diseases and cancer. Vaccine. 2014;32(48):6377–89.
Eypper EH, Johnson PV, Purro EI, Hohmann EL. Transcutaneous immunization of healthy volunteers with an attenuated Listeria monocytogenes vaccine strain and cholera toxin adjuvant. Vaccine. 2013;31(32):3257–61.
Yang C, Shi H, Zhou J, Liang Y, Xu H. CpG oligodeoxynucleotides are a potent adjuvant for an inactivated polio vaccine produced from Sabin strains of poliovirus. Vaccine. 2009;27(47):6558–63.
Norton EB, Bauer DL, Weldon WC, Oberste MS, Lawson LB, Clements JD. The novel adjuvant dmLT promotes dose sparing, mucosal immunity and longevity of antibody responses to the inactivated polio vaccine in a murine model. Vaccine. 2015;33(16):1909–15.
Dietrich J, Andreasen LV, Andersen P, Agger EM. Inducing dose sparing with inactivated polio virus formulated in adjuvant CAF01. PLoS One. 2014;9(6), e100879.
ACKNOWLEDGMENTS AND DISCLOSURES
The authors thank Heleen Kraan for contributions to analysis of IPV-specific IgG and VN responses, Raphael Zwier for contributions to the applicator design and Anne Römgens for contributions to discussions related to this study.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
This study was funded by the Dutch Technology Foundation STW, which is part of the Netherlands Organization for Scientific Research (NWO), and which is partly funded by the Ministry of Economic Affairs (project no. 11259).
Conflict of Interest
The authors declare that they have no conflict of interest.
Ethical Approval
All applicable international, national, and/or institutional guidelines for the care and use of animals were followed. This article does not contain any studies with human participants performed by any of the authors.
Rights and permissions
About this article
Cite this article
Schipper, P., van der Maaden, K., Romeijn, S. et al. Determination of Depth-Dependent Intradermal Immunogenicity of Adjuvanted Inactivated Polio Vaccine Delivered by Microinjections via Hollow Microneedles. Pharm Res 33, 2269–2279 (2016). https://doi.org/10.1007/s11095-016-1965-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11095-016-1965-6