Abstract
Purpose
Recently, efficacy studies in mice have shown that amine-terminated cationic (CNP) nanoparticulate carriers of DTX offer an improved formulation of the drug for intravesical delivery. It is hypothesized that this improved efficacy may arise from a carrier mediated bladder exfoliation process that removes the urothelial barrier allowing for increased drug uptake into bladder tissue. The objective of this study was to investigate exfoliation processes in fresh pig’s bladders (ex vivo) exposed to three cationic polyglycerols with increasing degrees of amination (denoted 350, 580 and 780). The study also compared the tissue depth profile of DTX uptake into these tissues using these different carriers.
Materials and Methods
Aminated polyglycerols were synthesized and characterized in the laboratory with low (CNP-360), medium (CNP-580) and high (CNP-780) levels of amine content. CNP-based DTX solutions and commercial DTX solutions in polysorbate 80 (Taxotere®) were doped with 3H-radiolabeled DTX and prepared by solvent evaporation from acetonitrile, followed by drying and reconstitution in pH 6.4 buffer. Sections of fresh pig’s bladder tissue were clamped into Franz diffusion cells and the urothelial side was exposed to the DTX solutions for 2 h. Tissue sections were then frozen for sectioning by cryotome sectioning and subsequently processed for drug analysis by liquid scintillation counting. Alternatively tissue sections were fixed in 2% glutaraldehyde and 2% paraformaldehyde in 0.1 M sodium cacodylate buffer for the purposes of scanning electron microscopy (SEM).
Results
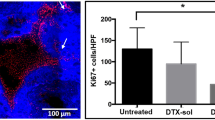
Exposure of the urothelial surface to the amine-terminated polyglycerol solutions resulted in the exfoliation of bladder tissues in a time- and concentration-dependent manner. Exfoliation was significantly more pronounced when using CNPs with a medium or high levels of amination whereas only minor levels of exfoliation were seen with low levels. Following incubation of tissues in Tween-based commercial formulations (Taxotere) of DTX (0.5 mg/mL) the drug was detectable at low levels (10–40 μg/g tissue) in all depths of tissue. Similar drug uptake was observed using the CNP-360 formulation. However drug uptake levels were increased to 60–100 μg/g tissue when samples were incubated with either the CNP-580 or CNP-780 formulations.
Conclusion
The use of cationic polyglycerols with higher levels of amine termination allows for an enhanced uptake of DTX into bladder tissues as compared to commercial (Taxotere) formulations. These increased drug levels probably arise from exfoliation processes resulting in a temporary elimination of the urothelial permeability barrier and increased drug penetration into the tissue.












Similar content being viewed by others
Abbreviations
- CNP:
-
Cationic polyglycerol
- CNP-C8/10-MePEG:
-
Hydrophobically modified CNPs with C8/C10 alkyl chains and Methoxy poly (ethylene glycol)
- CNP-C8/10-MePEG-NH2 :
-
CNP-C8/10-MePEG with surface amine groups
- DTX:
-
Docetaxel
- NMIBC:
-
Non-muscle invasive bladder cancer
- O/DEG:
-
Octyl/decyl glycidyl ether
- PTX:
-
Paclitaxel
- SEM:
-
Scanning electron microscopy
References
Statistics CCSsACoC. Advisory Committee on Cancer Statistics (2013) Bladder cancer statistics. In: Society. CC, editor. Canadian Cancer Statistics 2013. Toronto, ON; 2013.
Falke J, Witjes JA. Contemporary management of low-risk bladder cancer. Nat Rev Urol. 2011;8(1):42–9.
Gkritsios P, Hatzimouratidis K, Kazantzidis S, Dimitriadis G, Ioannidis E, Katsikas V. Hexaminolevulinate-guided transurethral resection of non-muscle-invasive bladder cancer does not reduce the recurrence rates after a 2-year follow-up: a prospective randomized trial. Int Urol Nephrol. 2013.
Prasad SM, Decastro GJ, Steinberg GD. Urothelial carcinoma of the bladder: definition, treatment and future efforts. Nat Rev Urol. 2011;8(11):631–42.
Richards KA, Smith ND, Steinberg GD. The importance of transurethral resection of bladder tumor in the management of non-muscle invasive bladder cancer: A systematic review of novel technologies. J Urol. 2014.
Gierth M, Burger M. Bladder cancer: progress in defining progression in NMIBC. Nat Rev Urol. 2013;10(12):684–5.
Hicks RM. The mammalian urinary bladder: an accommodating organ. Biol Rev Camb Philos Soc. 1975;50(2):215–46.
Jost SP, Gosling JA, Dixon JS. The morphology of normal human bladder urothelium. J Anat. 1989;167:103–15.
Khandelwal P, Abraham SN, Apodaca G. Cell biology and physiology of the uroepithelium. Am J Physiol Ren Physiol. 2009;297(6):F1477–501.
Romih R, Jezernik K, Masera A. Uroplakins and cytokeratins in the regenerating rat urothelium after sodium saccharin treatment. Histochem Cell Biol. 1998;109(3):263–9.
Veranic P, Romih R, Jezernik K. What determines differentiation of urothelial umbrella cells? Eur J Cell Biol. 2004;83(1):27–34.
Schulzke JD, Fromm M. Tight junctions: molecular structure meets function. Ann N Y Acad Sci. 2009;1165:1–6.
Peppas NA, Huang Y. Nanoscale technology of mucoadhesive interactions. Adv Drug Deliv Rev. 2004;56(11):1675–87.
Lewis SA, Diamond JM. Na+ transport by rabbit urinary bladder, a tight epithelium. J Membr Biol. 1976;28(1):1–40.
Hadaschik BA, ter Borg MG, Jackson J, Sowery RD, So AI, Burt HM, et al. Paclitaxel and cisplatin as intravesical agents against non-muscle-invasive bladder cancer. BJU Int. 2008;101(11):1347–55.
Logan C, Brown M, Hayne D. Intravesical therapies for bladder cancer—indications and limitations. BJU Int. 2012;110 Suppl 4:12–21.
Jost SP. Renewal of normal urothelium in adult mice. Virchows Arch B Cell Pathol Incl Mol Pathol. 1986;51(1):65–70.
Grabnar I, Bogataj M, Mrhar A. Influence of chitosan and polycarbophil on permeation of a model hydrophilic drug into the urinary bladder wall. Int J Pharm. 2003;256(1–2):167–73.
Erman A, Kerec Kos M, Zakelj S, Resnik N, Romih R, Veranic P. Correlative study of functional and structural regeneration of urothelium after chitosan-induced injury. Histochem Cell Biol. 2013;140(5):521–31.
Lavelle J, Meyers S, Ramage R, Bastacky S, Doty D, Apodaca G, et al. Bladder permeability barrier: recovery from selective injury of surface epithelial cells. Am J Physiol Ren Physiol. 2002;283(2):F242–53.
Mugabe C, Matsui Y, So AI, Gleave ME, Heller M, Zeisser-Labouebe M, et al. In vitro and in vivo evaluation of intravesical docetaxel loaded hydrophobically derivatized hyperbranched polyglycerols in an orthotopic model of bladder cancer. Biomacromolecules. 2011;12(4):949–60.
Nagarwal RC, Singh PN, Kant S, Maiti P, Pandit JK. Chitosan coated PLA nanoparticles for ophthalmic delivery: characterization, in-vitro and in-vivo study in rabbit eye. J Biomed Nanotechnol. 2010;6(6):648–57.
Jain SK, Chourasia MK, Jain AK, Jain RK, Shrivastava AK. Development and characterization of mucoadhesive microspheres bearing salbutamol for nasal delivery. Drug Deliv. 2004;11(2):113–22.
Kumar K, Dhawan N, Sharma H, Vaidya S, Vaidya B. Bioadhesive polymers: novel tool for drug delivery. Artif Cells Nanomedicine Biotechnol. 2014;42(4):274–83.
Veranic P, Erman A, Kerec-Kos M, Bogataj M, Mrhar A, Jezernik K. Rapid differentiation of superficial urothelial cells after chitosan-induced desquamation. Histochem Cell Biol. 2009;131(1):129–39.
Galsky MD. The role of taxanes in the management of bladder cancer. Oncologist. 2005;10(10):792–8.
JM McKiernan, PM Pierorazio. Beyond BCG: taxanes. In: Lee CT, Wood, David P., editors. Bladder Cancer, Current Clinical Urology Humana Press. 2010. p. 127–31.
McKiernan JM, Masson P, Murphy AM, Goetzl M, Olsson CA, Petrylak DP, et al. Phase I trial of intravesical docetaxel in the management of superficial bladder cancer refractory to standard intravesical therapy. J Clin Oncol Off J Am Soc Clin Oncol. 2006;24(19):3075–80.
Laudano MA, Barlow LJ, Murphy AM, Petrylak DP, Desai M, Benson MC, et al. Long-term clinical outcomes of a phase I trial of intravesical docetaxel in the management of non-muscle-invasive bladder cancer refractory to standard intravesical therapy. Urology. 2010;75(1):134–7.
Mugabe C, Raven PA, Fazli L, Baker JH, Jackson JK, Liggins RT, et al. Tissue uptake of docetaxel loaded hydrophobically derivatized hyperbranched polyglycerols and their effects on the morphology of the bladder urothelium. Biomaterials. 2012;33(2):692–703.
Mugabe C, Liggins RT, Guan D, Manisali I, Chafeeva I, Brooks DE, et al. Development and in vitro characterization of paclitaxel and docetaxel loaded into hydrophobically derivatized hyperbranched polyglycerols. Int J Pharm. 2011;404(1–2):238–49.
Mugabe C, Matsui Y, So AI, Gleave ME, Baker JH, Minchinton AI, et al. In vivo evaluation of mucoadhesive nanoparticulate docetaxel for intravesical treatment of non-muscle-invasive bladder cancer. Clin Cancer Res Off J Am Assoc Cancer Res. 2011;17(9):2788–98.
Kainthan RK, Mugabe C, Burt HM, Brooks DE. Unimolecular micelles based on hydrophobically derivatized hyperbranched polyglycerols: ligand binding properties. Biomacromolecules. 2008;9(3):886–95.
Kainthan RK, Brooks DE. Unimolecular micelles based on hydrophobically derivatized hyperbranched polyglycerols: biodistribution studies. Bioconjug Chem. 2008;19(11):2231–8.
Mugabe C, Hadaschik BA, Kainthan RK, Brooks DE, So AI, Gleave ME, et al. Paclitaxel incorporated in hydrophobically derivatized hyperbranched polyglycerols for intravesical bladder cancer therapy. BJU Int. 2009;103(7):978–86.
Tsallas A, Jackson J, Burt H. The uptake of paclitaxel and docetaxel into ex vivo porcine bladder tissue from polymeric micelle formulations. Cancer Chemother Pharmacol. 2011;68(2):431–44.
Schneider CA, Rasband WS, Eliceiri KW. NIH Image to ImageJ: 25 years of image analysis. Nat Methods. 2012;9(7):671–5.
Jaal J, Dorr W. Radiation-induced damage to mouse urothelial barrier. Radiother Oncol. 2006;80(2):250–6.
Mulvey MA, Lopez-Boado YS, Wilson CL, Roth R, Parks WC, Heuser J, et al. Induction and evasion of host defenses by type 1-piliated uropathogenic Escherichia coli. Science. 1998;282(5393):1494–7.
Jevprasesphant R, Penny J, Jalal R, Attwood D, McKeown NB, D’Emanuele A. The influence of surface modification on the cytotoxicity of PAMAM dendrimers. Int J Pharm. 2003;252(1–2):263–6.
Song D, Wientjes MG, Au JL. Bladder tissue pharmacokinetics of intravesical taxol. Cancer Chemother Pharmacol. 1997;40(4):285–92.
Acknowledgements
The research work was funded by the Canadian Institute of Health and Research (CIHR). We thank the Centre for Drug and Research Development (CDRD) for providing the polymer and expertise that greatly assisted the research.
Author information
Authors and Affiliations
Corresponding author
Additional information
In this research article amine group substitution on CNP’s is represented by the number of micromoles (x) of amine per gram of CNP’s and is denoted by CNP- (x) where x is 360 (low), 580 (medium) and 780 (high) μmoles of amine per gram of CNP.
Rights and permissions
About this article
Cite this article
Pandey, R., Jackson, J.K., Mugabe, C. et al. Tissue Permeability Effects Associated with the Use of Mucoadhesive Cationic Nanoformulations of Docetaxel in the Bladder. Pharm Res 33, 1850–1861 (2016). https://doi.org/10.1007/s11095-016-1920-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11095-016-1920-6