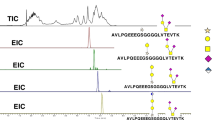
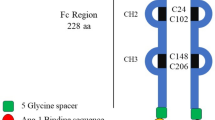
ABSTRACT
Purpose
P. pastoris has previously been genetically engineered to generate strains that are capable of producing mammalian-like glycoforms. Our objective was to investigate the correlation between sialic acid content and pharmacokinetic properties of recombinant TNFR2:Fc fusion proteins generated in glycoengineered P. pastoris strains.
Methods
TNFR2:Fc fusion proteins were generated with varying degrees of sialic acid content. The pharmacokinetic properties of these proteins were assessed by intravenous and subcutaneous routes of administration in rats. The binding of these variants to FcRn were also evaluated for possible correlations between in vitro binding and in vivo PK.
Results
The pharmacokinetic profiles of recombinant TNFR2:Fc produced in P. pastoris demonstrated a direct positive correlation between the extent of glycoprotein sialylation and in vivo pharmacokinetic properties. Furthermore, recombinant TNFR2:Fc produced in glycoengineered Pichia, with a similar sialic acid content to CHO-produced etanercept, demonstrated similar in vivo pharmacokinetic properties to the commercial material. In vitro surface plasmon resonance FcRn binding at pH6.0 showed an inverse relationship between sialic acid content and receptor binding affinity, with the higher affinity binders having poorer in vivo PK profiles.
Conclusions
Sialic acid content is a critical attribute for modulating the pharmacokinetics of recombinant TNFR2:Fc produced in glycoengineered P. pastoris.





Similar content being viewed by others
Abbreviations
- ASGPR:
-
asialoglycoprotein receptor
- AUC:
-
area under the curve
- BR3-Fc:
-
B cell-activating factor receptor 3 (BR3) Fc fusion protein
- CHO:
-
Chinese hamster ovary
- Fc:
-
crystallizable fragment of antibody
- FcRn:
-
neonatal Fc receptor
- GlcNAc:
-
n-acetylglucosamine
- IgG1:
-
immunoglobulin G subclass 1
- IMID:
-
immune-mediated inflammatory disease
- IV:
-
intravenous
- LFA3TIP:
-
lymphocyte function-associated molecule 3 Fc fusion protein
- PK:
-
pharmacokinetics
- rhEPO:
-
recombinant human erythropoietin
- SC:
-
subcutaneous
- TNF:
-
tumor necrosis factor
- TNFR2:
-
tumor necrosis factor receptor 2
- TSA:
-
total sialic acid content
REFERENCES
Keystone EC, Ware CF. Tumor necrosis factor and anti-tumor necrosis factor therapies. J Rheumatol Suppl. 2010;85:27–39.
Thalayasingam N, Isaacs JD. Anti-TNF therapy. Best Pract Res Clin Rheumatol. 2011;25(4):549–67.
Walsh G. Biopharmaceutical benchmarks 2010. Nat Biotechnol. 2010;28(9):917–24.
Mohler KM, Torrance DS, Smith CA, Goodwin RG, Stremler KE, Fung VP, et al. Soluble tumor necrosis factor (TNF) receptors are effective therapeutic agents in lethal endotoxemia and function simultaneously as both TNF carriers and TNF antagonists. J Immunol. 1993;151(3):1548–61.
Pennica D, Lam VT, Weber RF, Kohr WJ, Basa LJ, Spellman MW, et al. Biochemical characterization of the extracellular domain of the 75-kilodalton tumor necrosis factor receptor. Biochemistry. 1993;32(12):3131–8.
Tesar DB, Bjorkman PJ. An intracellular traffic jam: Fc receptor-mediated transport of immunoglobulin G. Curr Opin Struct Biol. 2010;20(2):226–33.
Punt PJ, van Biezen N, Conesa A, Albers A, Mangnus J, van den Hondel C. Filamentous fungi as cell factories for heterologous protein production. Trends Biotechnol. 2002;20(5):200–6.
Choi BK, Bobrowicz P, Davidson RC, Hamilton SR, Kung DH, Li H, et al. Use of combinatorial genetic libraries to humanize N-linked glycosylation in the yeast Pichia pastoris. Proc Natl Acad Sci U S A. 2003;100(9):5022–7.
Hamilton SR, Bobrowicz P, Bobrowicz B, Davidson RC, Li H, Mitchell T, et al. Production of complex human glycoproteins in yeast. Science. 2003;301(5637):1244–6.
Bobrowicz P, Davidson RC, Li H, Potgieter TI, Nett JH, Hamilton SR, et al. Engineering of an artificial glycosylation pathway blocked in core oligosaccharide assembly in the yeast Pichia pastoris: production of complex humanized glycoproteins with terminal galactose. Glycobiology. 2004;14(9):757–66.
Hamilton SR, Davidson RC, Sethuraman N, Nett JH, Jiang Y, Rios S, et al. Humanization of yeast to produce complex terminally sialylated glycoproteins. Science. 2006;313(5792):1441–3.
Hopkins D, Gomathinayagam S, Rittenhour AM, Du M, Hoyt E, Karaveg K, et al. Elimination of beta-mannose glycan structures in Pichia pastoris. Glycobiology. 2011;21(12):1616–26.
Lowy PH, Keighley G, Borsook H. Inactivation of erythropoietin by neuraminidase and by mild substitution reactions. Nature. 1960;185:102–3.
Lukowsky WA, Painter RH. Studies on the role of sialic acid in the physical and biological properties of erythropoietin. Can J Biochem. 1972;50(8):909–17.
Goldwasser E, Kung CK, Eliason J. On the mechanism of erythropoietin-induced differentiation. 13. The role of sialic acid in erythropoietin action. J Biol Chem. 1974;249(13):4202–6.
Stadheim TA, Li H, Kett W, Burnina IN, Gerngross TU. Use of high-performance anion exchange chromatography with pulsed amperometric detection for O-glycan determination in yeast. Nat Protoc. 2008;3(6):1026–31.
Wang W, Lu P, Fang Y, Hamuro L, Pittman T, Carr B, et al. Monoclonal antibodies with identical Fc sequences can bind to FcRn differentially with pharmacokinetic consequences. Drug Metab Dispos. 2011;39(9):1469–77.
Liu L, Stadheim A, Hamuro L, Pittman T, Wang W, Zha D, et al. Pharmacokinetics of IgG1 monoclonal antibodies produced in humanized Pichia pastoris with specific glycoforms: a comparative study with CHO produced materials. Biologicals. 2011;39(4):205–10.
Suzuki T, Ishii-Watabe A, Tada M, Kobayashi T, Kanayasu-Toyoda T, Kawanishi T, et al. Importance of neonatal FcR in regulating the serum half-life of therapeutic proteins containing the Fc domain of human IgG1: a comparative study of the affinity of monoclonal antibodies and Fc-fusion proteins to human neonatal FcR. J Immunol. 2010;184(4):1968–76.
Beck A, Reichert JM. Therapeutic Fc-fusion proteins and peptides as successful alternatives to antibodies. MAbs. 2011;3(5):415–6.
Kontermann RE. Strategies for extended serum half-life of protein therapeutics. Curr Opin Biotechnol. 2011;22(6):868–76.
Wang YM, Sloey B, Wong T, Khandelwal P, Melara R, Sun YN. Investigation of the pharmacokinetics of romiplostim in rodents with a focus on the clearance mechanism. Pharm Res. 2011;28(8):1931–8.
Datta-Mannan A, Chow CK, Dickinson C, Driver D, Lu J, Witcher DR, et al. FcRn affinity-pharmacokinetic relationship of five human IgG4 antibodies engineered for improved in vitro FcRn binding properties in cynomolgus monkeys. Drug Metab Dispos. 2012;40(8):1545–55.
Datta-Mannan A, Witcher DR, Tang Y, Watkins J, Jiang W, Wroblewski VJ. Humanized IgG1 variants with differential binding properties to the neonatal Fc receptor: relationship to pharmacokinetics in mice and primates. Drug Metab Dispos. 2007;35(1):86–94.
Ober RJ, Radu CG, Ghetie V, Ward ES. Differences in promiscuity for antibody-FcRn interactions across species: implications for therapeutic antibodies. Int Immunol. 2001;13(12):1551–9.
Andersen JT, Daba MB, Berntzen G, Michaelsen TE, Sandlie I. Cross-species binding analyses of mouse and human neonatal Fc receptor show dramatic differences in immunoglobulin G and albumin binding. J Biol Chem. 2010;285(7):4826–36.
Vaughn DE, Bjorkman PJ. High-affinity binding of the neonatal Fc receptor to its IgG ligand requires receptor immobilization. Biochemistry. 1997;36(31):9374–80.
Schwartz AL. Trafficking of asialoglycoproteins and the asialoglycoprotein receptor. Target Diagn Ther. 1991;4:3–39.
Stahl PD, Ezekowitz RA. The mannose receptor is a pattern recognition receptor involved in host defense. Curr Opin Immunol. 1998;10(1):50–5.
Egrie JC, Browne JK. Development and characterization of novel erythropoiesis stimulating protein (NESP). Nephrol Dial Transplant. 2001;16 Suppl 3:3–13.
Meier W, Gill A, Rogge M, Dabora R, Majeau GR, Oleson FB, et al. Immunomodulation by LFA3TIP, an LFA-3/IgG1 fusion protein: cell line dependent glycosylation effects on pharmacokinetics and pharmacodynamic markers. Ther Immunol. 1995;2(3):159–71.
Stefanich EG, Ren S, Danilenko DM, Lim A, Song A, Iyer S, et al. Evidence for an asialoglycoprotein receptor on nonparenchymal cells for O-linked glycoproteins. J Pharmacol Exp Ther. 2008;327(2):308–15.
ACKNOWLEDGMENTS AND DISCLOSURES
The authors would like to thank members of the Strain Development, Fermentation, Purification and Analytical groups of Biologics Discovery (GlycoFi, Merck), that supported this study. We would also like to thank Tamara Pittman and Scott Faulty for their in vivo support, and Dr Paul Peloso for reviewing this manuscript. All funding for this project was provided by Merck & Co., Inc.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Liu, L., Gomathinayagam, S., Hamuro, L. et al. The Impact of Glycosylation on the Pharmacokinetics of a TNFR2:Fc Fusion Protein Expressed in Glycoengineered Pichia Pastoris . Pharm Res 30, 803–812 (2013). https://doi.org/10.1007/s11095-012-0921-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11095-012-0921-3