Abstract
We normally think of enzymes as being proteins; however, the RNA world hypothesis suggests that the earliest biological catalysts may have been composed of RNA. One of the oldest surviving RNA enzymes we are aware of is the peptidyl transferase centre (PTC) of the large ribosomal RNA, which joins amino acids together to form proteins. Recent evidence indicates that the enzymatic activity of the PTC is principally due to ribose 2′-OHs. Many other reactions catalyzed by RNA and/or in which RNA is a substrate similarly utilize ribose 2′-OHs, including phosphoryl transfer reactions that involve the cleavage and/or ligation of the ribose-phosphate backbone. It has recently been proposed by Yakhnin (2013) that phosphoryl transfer reactions were important in the prebiotic chemical evolution of RNA, by enabling macromolecules composed of polyols joined by phosphodiester linkages to undergo recombination reactions, with the reaction energy supplied by the phosphodiester bond itself. The almost unique juxtaposition of the ribose 2′-hydroxyl and 3′-oxygen in ribose-containing polymers such as RNA, which gives ribose the ability to catalyze such reactions, may have been an important factor in the selection of ribose as a component of the first biopolymer. In addition, the juxtaposition of hydroxyl groups in free ribose: (i) allows coordination of borate ions, which could have provided significant and preferential stabilization of ribose in a prebiotic environment; and (ii) enhances the rate of permeation by ribose into a variety of lipid membrane systems, possibly favouring its incorporation into early metabolic pathways and an ancestral ribose-phosphate polymer. Somewhat more speculatively, hydrogen bonds formed by juxtaposed ribose hydroxyl groups may have stabilized an ancestral ribose-phosphate polymer against degradation (Bernhardt and Sandwick 2014). I propose that the almost unique juxtaposition of ribose hydroxyl groups constitutes the root of both biological catalysis and the RNA world.
Similar content being viewed by others
Of all the evidence in favour of an ancestral world dominated by RNA, perhaps the most compelling is the likelihood that the vast majority of proteins that have ever existed were made by one – an RNA, that is. And the evidence from ribosome X-ray crystallography that this remains the case (Nissen et al. 2000) emphasizes the continuing indispensable role of RNA catalysis in the biological realm. Logically, coded ribosomal protein synthesis must have preceded the appearance of complex proteins, and therefore the ribosomal peptidyl transferase centre (PTC), which catalyzes peptide synthesis, is one of the most ancient enzymes that we know of (Steitz and Moore 2003), despite the fact that we usually think of enzymes as being proteins. As such, the way in which the PTC catalyzes peptide synthesis potentially provides us with a window into the origins of biological catalysis. After more than three billion years of evolution, has the modern PTC retained any features of the ancestral catalytic mechanism?
The PTC catalyzes peptide bond formation between amino acids attached to the ribose 3′-OHs of the adenosine A76 residues of the A and P site tRNAs. Although some controversy still remains, there is a growing consensus that catalysis by the PTC is mainly due to its ribose 2′-OHs (Erlacher et al. 2006; Trobro and Åqvist 2005). Substrate-assisted catalysis by the ribose 2′-OH of A76 of the P site tRNA also plays an important part (Zaher et al. 2011; Rodnina 2013); the critical roles of the juxtaposed 2′/3′-OHs of the tRNA A76 in all aspects of translation have been reviewed previously (Weinger and Strobel 2006; Safro and Klipcan 2013). What is more, the importance of ribose hydroxyls appears to be a general theme in RNA catalysis beyond the PTC, and more generally in reactions in which RNA is a substrate, including substrate-assisted catalysis in a number of other RNA enzyme-catalyzed reactions (Smith and Pace 1993) and even in cases where the principle catalyst is a protein enzyme (Minajigi and Francklyn 2008).
Substrate-assisted catalysis would seem like a sensible place to look for the origins of biological catalysis, as preprogramming by substrate chemistry would have reduced the requirement for a complex active site in a prebiotic environment; in fact, Safro and Klipcan (2013) have proposed that the activity of the juxtaposed 2′/3′-OHs of the tRNA A76 ribose qualifies tRNA as a ribozyme. However, in the modern reactions in which substrate-assisted catalysis by ribose hydroxyls plays a role – and the PTC is a good example of this – such catalysis still occurs within a highly complex active site. Is catalysis possible in the absence of such complexity, as would appear to have been necessary in the prebiotic situation? A possible answer to this question is provided by phosphoryl transfer reactions – the most common reactions catalyzed by contemporary RNA enzymes – which involve the cleavage and/or ligation of the RNA phosphodiester backbone. The active site of such reactions often consists of no more than a single RNA strand, with the sole requirement being the juxtaposition of the ribose 2′-hydroxyl and 3′-oxygen (Oivanen et al. 1998 and references therein). Yakhnin (2013) has proposed that phosphoryl transfer played a critical role in the chemical evolution of RNA, by enabling recombination reactions that resulted in selection of the most stable phosphodiester bond-containing molecules. Although Yakhnin has proposed that such reactions could have been catalyzed by hydroxyl groups belonging to a range of different polyols, it seems likely that ribose has been performing this function from the beginning, due to the catalytic ability of its juxtaposed 2′-hydroxyl and 3′-oxygen (Bernhardt and Sandwick 2014).
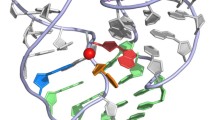
In addition to the important contribution of the ribose 2′-OH to catalysis, the juxtaposition of hydroxyl groups in free ribose may have played a critical role in stabilizing it against degradation, as well as in enabling its faster permeation of prebiotic lipid vesicles. The prebiotic synthesis of ribose is generally thought to have occurred from formaldehyde via the formose reaction pathway; however, this pathway produces a myriad of products, of which ribose is only one of many (Benner et al. 2012). The five-membered ring form of ribose (ribofuranose) can adopt two conformations, with the 1′-OH pointing either above or below the plane of the ring, termed β- and α-conformers, respectively. In α-ribofuranose, the 1′-OH points below the plane of the ring in the same direction as the 2′- and 3′-OHs, with the result that the three hydroxyls point in the same direction (Fig. 1a). This arrangement of hydroxyl groups allows the coordination of borate ions by either the 1′/2′- or 2′/3′-OHs, with the former being more energetically favourable (Chapelle and Verchere 1988). Benner and colleagues have shown that the presence of borate-containing minerals is both compatible with ribose synthesis by the formose reaction, and also protects ribose from degradation for 2 months at 45 °C and pH~12 (Ricardo et al. 2004), suggesting borate may have played a critical role in the preferential stabilization of ribose in a prebiotic environment. In addition – and again as a result of the juxtaposition of its hydroxyl groups – ribose could have more quickly permeated a variety of prebiotic vesicles. In work carried out by Sacerdote and Szostak (2005), ribose was shown to permeate a range of different lipid membrane systems 3- to 10-fold faster than xylose and arabinose, two similar pentoses. Free ribose in aqueous solution normally adopts predominantly the six-membered ring (ribopyranose) conformation, of which the lowest energy and most populated conformer is the β-ribopyranose 4 C 1 chair conformation (Franks et al. 1989) in which the 1′-OH points slightly above the plane of the ring. Wei and Pohorille (2009) have shown using molecular modeling that the four ribose hydroxyls in this conformation are able to form an intramolecular chain of hydrogen bonds (Fig. 1b) that reduces the effective hydrophilicity of the hydroxyl groups, increasing the solubility of ribose in the lipid phase and enabling it to traverse lipid membranes more quickly than other pentoses. This difference appears to be due to the fact that, in xylose and arabinose, the 2′- and 3′-OH groups point in opposite directions, making formation of a similar chain of hydrogen bonds impossible (Wei and Pohorille 2009).
The role of juxtaposed ribose hydroxyl groups in the coordination of borate and in hydrogen bonding interactions, with carbon skeletons and hydrogen bonds between numbered hydroxyls indicated (a) α-ribofuranose coordinating borate (black) (b) β-ribopyranose in 4 C 1 chair conformation with proposed intramolecular chain of hydrogen bonds (c) α-ribofuranose as part of a proposed ancestral ribose-phosphate polymer; some ribose hydrogens have been omitted for clarity. Based on naked ribose zipper nIL (Tamura and Holbrook 2002) from PDB 1S72 (Klein et al. 2004)
We have previously proposed that RNA evolved from an abasic ribose-phosphate polymer by a process of chemical evolution occurring through a series of spontaneous (nonenzymatic) reactions at the ribose C1 carbon, which ultimately produced the purine nucleobases, with the pyrimidine nucleobases evolving later (Bernhardt and Sandwick 2014). In a ribose-phosphate polymer with 5′–3′-phosphodiester linkages, the 5′-phosphate constrains ribose into the ribofuranose conformation. Similarly, ribose-5-phosphate in aqueous solution exists as an equilibrium mixture of mainly α- (~36 %) and β- (~64 %) ribofuranose (Pierce et al. 1985), with the α-conformer having the 1′-OH pointing below the plane of the ring in the same direction as the 2′- and 3′-OHs, the same conformation that coordinates borate (Fig. 1a). We have proposed that evolution of the purine nucleobase precursors was driven by their ability to progressively stabilize the ribose-phosphate backbone to which they were attached, with the earliest stabilizing interactions possibly consisting of hydrogen bonds between ribose 1′- and 2′-OH groups, analogous to naked ribose-zipper interactions that occur between ribose 2′-OH groups in modern RNA (Tamura and Holbrook 2002; Ulyanov and James 2010). The fact that the 1′ and 2′-OHs point in the same direction in the α-ribofuranose conformation potentially would have allowed the formation of two hydrogen bonds by each ribose as part of the proposed ribose-phosphate polymer. If this bonding provided sufficient free energy for duplex formation (as depicted in Fig. 1c), this could have stabilized the proposed abasic ribose-phosphate backbone against degradation, as has been shown to be the case for RNA, where duplexes are much more resistant than single stranded regions to phosphodiester bond cleavage (Mikkola et al. 2001).
Somewhat ironically, the way in which our understanding of the catalysis of protein synthesis has developed over the last 30 years appears to mirror the evolution of biological catalysis, only in reverse. With the demise of the belief that a histidine residue belonging to a ribosomal protein catalyzed the reaction (Sumpter et al. 1991), it was next assumed that an adenine nucleobase with an altered pK a must be responsible (Muth et al. 2000), until finally we have arrived at the concept of catalysis by ribose 2′-OHs (Erlacher et al. 2006; Trobro and Åqvist 2005). In addition to its critical role in catalysis, the almost unique juxtaposition of hydroxyl groups may have stabilized ribose against degradation through coordination of borate, and enhanced its movement across membranes in a prebiotic environment. In turn, and somewhat more speculatively, the faster permeation of ribose into prebiotic lipid vesicles may have been utilized serendipitously by its conversion to a charged or polymerized derivative, possibly ribose-5-phosphate or even a ribose-phosphate polymer in which the juxtaposition of ribose 1′- and 2′-hydroxyls and 3′-oxygen may have enabled both catalysis of recombination reactions as well as the formation of a lattice of hydrogen bonds that stabilized the ribose-phosphate backbone against cleavage, ultimately leading to the evolution of RNA as the first biopolymer of life. It seems that, in discovering the catalytic mechanism of the PTC, we may have stumbled upon the roots of biological catalysis and the RNA world, which, as is often the case, was in the last (and seemingly most unlikely) place that we looked.
References
Benner SA, Kim HJ, Carrigan MA (2012) Asphalt, water, and the prebiotic synthesis of ribose, ribonucleosides, and RNA. Acc Chem Res 45:2025–2034. doi:10.1021/ar200332w
Bernhardt HS, Sandwick RK (2014) Purine biosynthetic intermediate-containing ribose phosphate polymers as evolutionary precursors to RNA. J Mol Evol 79:91–104. doi:10.1007/s00239-014-9640-1
Chapelle S, Verchere J-F (1988) A 11B and 13C nmr determination of the structures of borate complexes of pentoses and related sugars. Tetrahedron 44:4469–4482. doi:10.1016/S0040-4020(01)86149-4
Erlacher MD, Lang K, Wotzel B, Rieder R, Micura R, Polacek N (2006) Efficient ribosomal peptidyl transfer critically relies on the presence of the ribose 2′-OH at A2451 of 23S rRNA. J Am Chem Soc 128:4453–4459
Franks F, Lillford PJ, Robinson G (1989) Isomeric equilibria of monosaccharides in solution. J Chem Soc Faraday Trans 1(85):2417–2426
Klein DJ, Moore PB, Steitz TA (2004) The roles of ribosomal proteins in the structure assembly, and evolution of the large ribosomal subunit. J Mol Biol 340:141–177
Mikkola S, Kaukinen U, Lönnberg H (2001) The effect of secondary structure on cleavage of the phosphodiester bonds of RNA. Cell Biochem Biophys 34:95–119
Minajigi A, Francklyn CS (2008) RNA-assisted catalysis in a protein enzyme: the 2′-hydroxyl of tRNA(Thr) A76 promotes aminoacylation by threonyl-tRNA synthetase. Proc Natl Acad Sci U S A 105:17748–17753. doi:10.1073/pnas.0804247105
Muth GW, Ortoleva-Donnelly L, Strobel SA (2000) A single adenosine with a neutral pK a in the ribosomal peptidyl transferase center. Science 289:947–950
Nissen P, Hansen J, Ban N, Moore PB, Steitz TA (2000) The structural basis of ribosome activity in peptide bond synthesis. Science 289:920–930
Oivanen M, Kuusela S, Lönnberg H (1998) Kinetics and mechanisms for the cleavage and isomerization of the phosphodiester bonds of RNA by Brønsted acids and bases. Chem Rev 98:961–990
Pierce J, Serianni AS, Barker R (1985) Anomerization of furanose sugars and sugar phosphates. J Am Chem Soc 107:2448–2456. doi:10.1021/ja00294a041
Ricardo A, Carrigan MA, Olcott AN, Benner SA (2004) Borate minerals stabilize ribose. Science 303:196
Rodnina MV (2013) The ribosome as a versatile catalyst: reactions at the peptidyl transferase center. Curr Opin Struct Biol 23:595–602. doi:10.1016/j.sbi.2013.04.012
Sacerdote MG, Szostak JW (2005) Semipermeable lipid bilayers exhibit diastereoselectivity favoring ribose. Proc Natl Acad Sci U S A 102:6004–6008
Safro M, Klipcan L (2013) The mechanistic and evolutionary aspects of the 2′- and 3′-OH paradigm in biosynthetic machinery. Biol Direct 8:17. doi:10.1186/1745-6150-8-17
Smith D, Pace NR (1993) Multiple magnesium ions in the ribonuclease P reaction mechanism. Biochemistry 32:5273–5281
Steitz TA, Moore PB (2003) RNA, the first macromolecular catalyst: the ribosome is a ribozyme. Trends Biochem Sci 28:411–418
Sumpter VG, Tate WP, Nowotny P, Nierhaus KH (1991) Modification of histidine residues on proteins from the 50S subunit of the Escherichia coli ribosome. Effects on subunit assembly and peptidyl transferase centre activity. Eur J Biochem 196:255–260
Tamura M, Holbrook SR (2002) Sequence and structural conservation in RNA ribose zippers. J Mol Biol 320:455–474
Trobro S, Åqvist J (2005) Mechanism of peptide bond synthesis on the ribosome. Proc Natl Acad Sci U S A 102:12395–12400
Ulyanov NB, James TL (2010) RNA structural motifs that entail hydrogen bonds involving sugar-phosphate backbone atoms of RNA. New J Chem 34:910–917. doi:10.1039/b9nj00754g
Wei C, Pohorille A (2009) Permeation of membranes by ribose and its diastereomers. J Am Chem Soc 131:10237–10245. doi:10.1021/ja902531k
Weinger JS, Strobel SA (2006) Participation of the tRNA A76 hydroxyl groups throughout translation. Biochemistry 45:5939–5948
Yakhnin AV (2013) A model for the origin of life through rearrangements among prebiotic phosphodiester polymers. Orig Life Evol Biosph 43:39–47. doi:10.1007/s11084-012-9321-2
Zaher HS, Shaw JJ, Strobel SA, Green R (2011) The 2′-OH group of the peptidyl-tRNA stabilizes an active conformation of the ribosomal PTC. EMBO J 30:2445–2453. doi:10.1038/emboj.2011.142
Acknowledgments
Chenyu Wei for supplying the PDB file of β-ribopyranose used to produce Fig. 1b. This work was supported by a Fast Start Grant from the Marsden Fund of New Zealand, the Department of Biochemistry, University of Otago, and by a travel grant from the Origins 2014 conference organizers.
Conflict of interest
The author declares that he has no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Bernhardt, H.S. The Juxtaposition of Ribose Hydroxyl Groups: The Root of Biological Catalysis and the RNA World?. Orig Life Evol Biosph 45, 15–19 (2015). https://doi.org/10.1007/s11084-015-9403-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11084-015-9403-z