Abstract
In this paper we explore the question of whether there is an optimal set up for a putative prebiotic system leading to open-ended evolution (OEE) of the events unfolding within this system. We do so by proposing two key innovations. First, we introduce a new index that measures OEE as a function of the likelihood of events unfolding within a universe given its initial conditions. Next, we apply this index to a variant of the graded autocatalysis replication domain (GARD) model, Segre et al. (P Natl Acad Sci USA 97(8):4112-4117, 2000; Markovitch and Lancet Artif Life 18(3), 2012), and use it to study - under a unified and concise prebiotic evolutionary framework - both a variety of initial conditions of the universe and the OEE of species that evolve from them.
Similar content being viewed by others
Open-Ended Evolution
Open-ended evolution may be thought of as referring to a “system in which components continue to evolve new forms continuously, rather than grinding to a halt when some sort of ‘optimal’ or stable position is reached” (Taylor 1999). Notably, open-ended evolution does not necessarily imply evolutionary progress or complexification. Yet, a system in which complexity increases along the evolutionary time axis fulfills a sufficient (even if not necessary) condition for open-endedness.
Indeed, evolution of complexity and the related concept of open-ended evolution have been a topic of scientific enquiry since Darwin and Wallace introduced the Theory of Evolution by Natural Selection. There is no doubt that a complexification process took place over the extended evolutionary time frame, with some endosymbiotic events (Neef et al. 2011). With the advent of powerful computational tools in which one could seamlessly run “what-if” scenarios about the origins of life, the questions of how complexity emerges from evolution-like processes and how open-ended the emergent processes have gained renewed impetus (Bedau et al. 2000). Researchers have proposed multiple definitions of both open-ended evolution and (pre)biotic complexity and have applied these measures to several, more or less convoluted, “Artificial Life” and prebiotic systems. Common definitions of open-ended evolution consider an increase in the internal complexity of species (McMullin 2000; Ruiz-Mirazo et al. 2004) or species occupying ever more diverse niches of the natural design space (Mcshea 1994; Korb and Dorin 2011). A difference between the two approaches is apparent when considering the case of a few species in an ecosystem becoming more and more complex vs. the emergence of multiple species that might each be relatively simple but overall occupy a relatively large portion of the natural possible design space. The former definition can be viewed as “species-centric” whereas the latter as more “system-centric” (Korb and Dorin 2011).
Korb and Dorin discuss at length the various attempts made at measuring open-endedness and suggest a two-part measure based on message minimum length required for conveying (or encoding) information (MML). They propose that a measure that considers the complexity of events of species evolving (part 1) and of the related hypothesis (part 2) would be a better measure for evolutionary complexity as it is a relative measure that takes into account not only the end product but the context in which these are produced. Building on this concept, we address the question of whether there is an optimal set up for a putative prebiotic universe that leads to greater OEE of the species evolving within it. We define an index of the excess-complexity of species (event, E) in relation to the universe in which they evolve (U), as a proxy for OEE. This index is ec(P(E|U), P(U)), that relates the probability of observing events (P(E|U)) to the probability of the initial conditions (P(U). Our index, like suggested by Korb and Dorin (2011), is a two part index but with the additional advantage of having the following properties:
-
1.
P(E|U) and P(U) are normalized such that 0 ≤ P(E|U), P(U) ≤ 1.
-
2.
P(E|U) → 0 and → 1 respectively represents improbable and probable outcomes unraveling from the initial conditions U. Similarly, P(U) → 0 and → 1 represents improbable and probable initial universe conditions.
-
3.
ec ≥ 0 and can grow arbitrarily large. The larger the value of ec, the more complex are the unfolding events in relation to a given universe.
-
4.
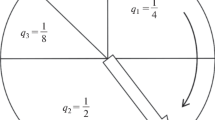
lim P(E|U)→1,P(U)→1 ec(P(E|U), P(U) ) = 0, that is, probable initial conditions that lead to probable events receive the lowest rank (i.e. no surprises can be expected from this universe under the given initial conditions). This is marked as Scenario A in Fig. 1.
-
5.
lim P(E|U)→1,P(U)→0 ec(P(E|U), P(U) ) = K; K > 0, that is, improbable initial conditions that lead to probable events are ranked slightly higher than 0 (Scenario B).
-
6.
lim P(E|U)→0,P(U)→0 ec(P(E|U), P(U) ) = L; L > K > 0, that is, an improbable initial state that leads to improbable events ranks even higher as this clearly represents an unexpected observation emerging from an unexpected initial condition (“Garden of Eden”, scenario C).
-
7.
lim P(E|U)→0,P(U)→1 ec(P(E|U), P(U) ) = M; M > L > K > 0, that is, a probable set of initial conditions throws out surprising outputs thus ranking at the top of the scale (“Elegant Garden of Eden”, scenario D).
We now define ec (Eq. 1) with exactly the above characteristics:
where the first part is an embodiment of MML and is scale invariant, and the second part is different from zero only when added value in the complexity of events has occurred (i.e. U is simpler than E). An increase in ec during a simulation will serve as a proxy to identify OEE, as an increase in complexity is generally considered to be indicative of OEE. Moreover, following the previous discussion, ec(E,U) is a species-centric measure of OEE but can easily be “system-shifted” by encompassing all outcomes E.
In Fig. 1 we identify the four extreme ec values a simulated universe might receive. As one moves from A, to B, to C and finally to D the level of ec increases, thus OEE is observed. In fact, as the likelihood of the initial conditions increases (P(U) → 1) and the likelihood of the events decreases (P(E|U) → 0) the level grows, potentially without limit.
Universe-GARD
In order to address the question of whether there is an optimal set up for a putative prebiotic “universe” with events unfolding inside such that OEE is observed, we extend the well-studied lipid-world framework, the GARD model (Segre et al. 2000). GARD simulates molecular assemblies formed by accretion from an immediate buffered environment containing a defined set of N G amphiphilic compound types, where the rate of accretion is biased by a network of mutually catalytic interactions, β. The assembly is kept out of equilibrium by imposing a fission action when it reaches a predefined size, producing two progeny of the same size, one of which grows again generating a growth-fission cycle of consecutive generations. GARD dynamics portray compotypes, sets of faithfully replicating compositional states that may be regarded as analogous to species or quasispecies (Eigen and Schuster 1977; Shenhav et al. 2007; Markovitch and Lancet 2012). Typically up to eight different compotypes are being cycled, suggesting that GARD does not appear to display high levels of ec to begin with, which impends on its OEE. The new proposed model, to be termed universe-GARD (U-GARD), will allow us to systematically study the tradeoff between the initial conditions of the universe and the emerging compotypes (i.e. map the ec surface). In U-GARD, the immediate environment is embedded in a larger “universe” with N U (≥ N G) different compound types, instances of which are continually being exchanged with the immediate environment (Fig. 2). This is physicochemically feasible, as exemplified by the immediate environment being absorbed to a mineral surface, contained in a mineral pore or constituting an ineffectively stirred sub-region of a larger prebiotic aquatic body. As a compotype constitutes a set of molecules that function better as a whole in their particular environment and thus faithfully replicate, the organization of a compotype is also assumed to protect its constituting molecular types from being “washed out” to the larger universe.
ec in U-GARD Simulations
When a simulation starts, the universe is defined by the initial configuration of the N U compounds. E and U respectively represent the compositions of compotypes encountered and the initial-environment. P(E|U) and P(U) will be measured as the probabilities of getting E and U by random picking. It is also possible to measure P(E|U) as the probability of finding new compotypes, never encountered before in that simulation, after a sufficiently long time course. OEE will be identified when a universe will exhibit an increase in ec. Different universes with different N U, N G and β parameters can be compared by using the expected value of ec:
in order to find the optimal set of parameters exhibiting the highest <ec>.
Conclusions
We have presented a new version of the GARD model that, through a simple extension, allows us to address the question of whether there is an optimal level of open-ended evolution that can be expected from a prebiotic world. To quantify this we have proposed a new index that incorporates several intuitive expectations of what “open-endedness” should look like. Namely, predictable and simple universes producing predictable and simple outputs are at the bottom of the scale and unlikely universes producing simple output score only slightly higher. Next on the scale we find the unlikely universe producing unlikely and complex output (Garden of Eden scenario) and finally, at the pinnacle, close to where true-emergence lies, we find those probable prebiotic universes that throw out unlikely and complex events (Elegant Garden of Eden scenarios, a hallmark of emergence). In our future work we plan to systematically chart what kind of dynamics GARD and U-GARD provide and how they map into ec.
References
Bedau MA, McCaskill JS, Packard NH, Rasmussen S, Adami C, Green DG, Ikegami T, Kaneko K, Ray TS (2000) Open problems in artificial life. Artif Life 6(4):363–376
Eigen M, Schuster P (1977) Hypercycle - principle of natural self-organization. A. Emergence of hypercycle. Naturwissenschaften 64(11):541–565
Korb KB, Dorin A (2011) Evolution unbound: releasing the arrow of complexity. Biol Philos 26(3):317–338. doi:10.1007/s10539-011-9254-6
Markovitch O, Lancet D (2012) Excess mutual catalysis is required for effective evolvability. Artif Life 18(3)
McMullin B (2000) John von Neumann and the evolutionary growth of complexity: looking backward, looking forward …. Artif Life 6(4):347–361
Mcshea DW (1994) Mechanisms of large-scale evolutionary trends. Evolution 48(6):1747–1763
Neef A, Latorre A, Pereto J, Silva FJ, Pignatelli M, Moya A (2011) Genome economization in the endosymbiont of the wood roach cryptocercus punctulatus Due to drastic loss of amino acid synthesis capabilities. Genome Biol Evol 3:1437–1448. doi:10.1093/Gbe/Evr118
Ruiz-Mirazo K, Pereto J, Moreno A (2004) A universal definition of life: autonomy and open-ended evolution. Origins Life Evol B 34(3):323–346
Segre D, Ben-Eli D, Lancet D (2000) Compositional genomes: prebiotic information transfer in mutually catalytic noncovalent assemblies. P Natl Acad Sci USA 97(8):4112–4117
Shenhav B, Oz A, Lancet D (2007) Coevolution of compositional protocells and their environment. Phil Trans R Soc B-Biol Sci 362(1486):1813–1819
Taylor TJ (1999) From artificial evolution to artificial life. Unpublished Ph.D. Thesis. University of Edinburg, Edinburg
Acknowledgements
NK would like to thank the UK’s Engineering and Physical Sciences Research Council for Grant EP/J004111/1 & EP/G042462/1. Parts of this work were done while he was a recipient of a Weizmann Institute of Science’s “Morris Belkin” visiting professorship. This work is partly supported by EU-FP7 project MATCHIT and by the Crown Human Genome Center at the Weizmann Institute of Science.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Markovitch, O., Sorek, D., Lui, L.T. et al. Is There an Optimal Level of Open-Endedness in Prebiotic Evolution?. Orig Life Evol Biosph 42, 469–474 (2012). https://doi.org/10.1007/s11084-012-9309-y
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11084-012-9309-y