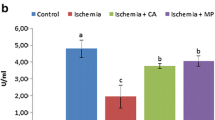
Abstract
In the present study, we investigated the protective effects of heme oxygenase (HO-1) against ischemic damage in motor neurons of the rabbit spinal cord. A PEP-1-HO-1 fusion protein was made to and confirmed the effective the penetration of HO-1 into spinal cord neurons at 8 h after treatment. Transient spinal cord ischemia was induced by occlusion of the abdominal aorta for 15 min. Vehicle (glycerol) or 0.375 mg/kg PEP-1-HO-1 was administered intraperitoneally to rabbits immediately after ischemia/reperfusion. Animals were sacrificed 15 min after reperfusion to measure lactate levels; 24 h after reperfusion to measure caspase 3 and myeloperoxidase levels, lipid peroxidation, and the activity of Cu,Zn-superoxide dismutase (SOD1) and catalase (CAT); or 72 h after reperfusion to assess neuronal survival and measure the levels of brain-derived neurotrophic factor (BDNF) in spinal cord homogenates. Administration of PEP-1-HO-1 did not significantly alter arterial blood gases (PaCO2 and PaO2), pH, or blood glucose levels before ischemia, 10 min after occlusion, or 10 min after reperfusion. Mean arterial pressure was selectively reduced 10 min after occlusion. Administration of PEP-1-HO-1 improved the rabbit Tarlov scores, and increased neuronal survival, as assessed by NeuN immunohistochemical staining 72 h after ischemia/reperfusion. In addition, administration of PEP-1-HO-1 significantly ameliorated lactate accumulation 15 min after reperfusion, and the increases in caspase 3, myeloperoxidase, and lipid peroxidation 24 h after reperfusion. PEP-1-HO-1 administration significantly mitigated the decrease in SOD1 and CAT 24 h after reperfusion, and reversed the decrease in BDNF levels in spinal cord homogenates 72 h after ischemia/reperfusion. These results suggest that PEP-1-HO-1 can protect against neuronal damage after transient spinal cord ischemia by limiting early lactic acidosis and increasing SOD1, CAT, and BDNF levels.







Similar content being viewed by others
References
Conrad MF, Ye JY, Chung TK, Davison JK, Cambria RP (2008) Spinal cord complications after thoracic aortic surgery: long-term survival and functional status varies with deficit severity. J Vasc Surg 48:47–53
DeGirolami U, Zivin JA (1982) Neuropathology of experimental spinal cord ischemia in the rabbit. J Neuropathol Exp Neurol 41:129–149
Weir CJ, Zivin JA, Lyden PD (2002) Inter-relationships between spinal cord blood flow, neuronal death and neurological function in rabbit spinal cord ischemia. Brain Res 946:43–51
Hayashi T, Sakurai M, Abe K, Sadahiro M, Tabayashi K, Itoyama Y (1998) Apoptosis of motor neurons with induction of caspases in the spinal cord after ischemia. Stroke 29:1007–1012
Mackey ME, Wu Y, Hu R et al (1997) Cell death suggestive of apoptosis after spinal cord ischemia in rabbits. Stroke 28:2012–2017
Chen WF, Sung CS, Jean YH et al (2010) Suppressive effects of intrathecal granulocyte colony-stimulating factor on excessive release of excitatory amino acids in the spinal cerebrospinal fluid of rats with cord ischemia: role of glutamate transporters. Neuroscience 165:1217–1232
Hwang I, Lee JC, Yoo KY et al (2007) Transient ischemia-induced changes of excitatory amino acid carrier 1 in the ventral horn of the lumbar spinal cord in rabbits. Neurol Res 29:310–316
Izumi S, Okada K, Hasegawa T et al (2010) Augmentation of systemic blood pressure during spinal cord ischemia to prevent postoperative paraplegia after aortic surgery in a rabbit model. J Thorac Cardiovasc Surg 139:1261–1268
Lin R, Roseborough G, Dong Y, Williams GM, Wei C (2003) DNA damage and repair system in spinal cord ischemia. J Vasc Surg 37:847–858
Marsala M, Sorkin LS, Yaksh TL (1994) Transient spinal ischemia in rat: characterization of spinal cord blood flow, extracellular amino acid release, and concurrent histopathological damage. J Cereb Blood Flow Metab 14:604–614
Mori A, Ueda T, Nakamichi T et al (1997) Detrimental effects of exogenous glutamate on spinal cord neurons during brief ischemia in vivo. Ann Thorac Surg 63:1057–1062
Tufan K, Oztanir N, Ofluoglu E et al (2008) Ultrastructure protection and attenuation of lipid peroxidation after blockade of presynaptic release of glutamate by lamotrigine in experimental spinal cord injury. Neurosurg Focus 25:E6
Kumar S, Bandyopadhyay U (2005) Free heme toxicity and its detoxification systems in human. Toxicol Lett 157:175–188
Kikuchi G, Yoshida T, Noguchi M (2005) Heme oxygenase and heme degradation. Biochem Biophys Res Commun 338:558–567
Ryter SW, Alam J, Choi AM (2006) Heme oxygenase-1/carbon monoxide: from basic science to therapeutic applications. Physiol Rev 86:583–650
de Sousa Oliveira Vanderlei E, de Araújo IW, Quinderé AL et al (2011) The involvement of the HO-1 pathway in the anti-inflammatory action of a sulfated polysaccharide isolated from the red seaweed Gracilaria birdiae. Inflamm Res 60:1121–1130
Geddes JW, Pettigrew LC, Holtz ML, Craddock SD, Maines MD (1996) Permanent focal and transient global cerebral ischemia increase glial and neuronal expression of heme oxygenase-1, but not heme oxygenase-2, protein in rat brain. Neurosci Lett 210:205–208
Matsuoka Y, Okazaki M, Kitamura Y (1999) Induction of inducible heme oxygenase (HO-1) in the central nervous system: is HO-1 helpful or harmful? Neurotox Res 1:113–117
Takeda A, Onodera H, Sugimoto A, Itoyama Y, Kogure K, Shibahara S (1994) Increased expression of heme oxygenase mRNA in rat brain following transient forebrain ischemia. Brain Res 666:120–124
Takizawa S, Hirabayashi H, Matsushima K, Tokuoka K, Shinohara Y (1998) Induction of heme oxygenase protein protects neurons in cortex and striatum, but not in hippocampus, against transient forebrain ischemia. J Cereb Blood Flow Metab 18:559–569
Youn JK, Kim DW, Kim ST et al (2014) PEP-1-HO-1 prevents MPTP-induced degeneration of dopaminergic neurons in a Parkinson’s disease mouse model. BMB Rep 47:569–574
Lin Y, Vreman HJ, Wong RJ, Tjoa T, Yamauchi T, Noble-Haeusslein LJ (2007) Heme oxygenase-1 stabilizes the blood–spinal cord barrier and limits oxidative stress and white matter damage in the acutely injured murine spinal cord. J Cereb Blood Flow Metab 27:1010–1021
Mautes AE, Bergeron M, Sharp FR et al (2000) Sustained induction of heme oxygenase-1 in the traumatized spinal cord. Exp Neurol 166:254–265
Lee JC, Kim IH, Park JH et al (2015) Ischemic preconditioning protects hippocampal pyramidal neurons from transient ischemic injury via the attenuation of oxidative damage through upregulating heme oxygenase-1. Free Radic Biol Med 79:78–90
Panahian N, Yoshiura M, Maines MD (1999) Overexpression of heme oxygenase-1 is neuroprotective in a model of permanent middle cerebral artery occlusion in transgenic mice. J Neurochem 72:1187–1203
Qi D, Ouyang C, Wang Y et al (2014) HO-1 attenuates hippocampal neurons injury via the activation of BDNF–TrkB–PI3K/Akt signaling pathway in stroke. Brain Res 1577:69–76
Kwon SW, Sohn EJ, Kim DW et al (2011) Anti-inflammatory effect of transduced PEP-1-heme oxygenase-1 in Raw 264.7 cells and a mouse edema model. Biochem Biophys Res Commun 411:354–359
Reagan-Shaw S, Nihal M, Ahmad N (2008) Dose translation from animal to human studies revisited. FASEB J 22:659–661
Kiyoshima T, Fukuda S, Matsumoto M et al (2003) Lack of evidence for apoptosis as a cause of delayed onset paraplegia after spinal cord ischemia in rabbits. Anesth Analg 96:839–846
Murakami H, Tsukube T, Kawanishi Y, Okita Y (2004) Transcranial myogenic motor-evoked potentials after transient spinal cord ischemia predicts neurologic outcome in rabbits. J Vasc Surg 39:207–213
Tarlov IM (1957) Spinal cord compression; mechanism of paralysis and treatment. In: Thomas CC (ed) Blackwell Scientific Publication, Oxford, p 147
Huang Y, Xie K, Li J et al (2011) Beneficial effects of hydrogen gas against spinal cord ischemia-reperfusion injury in rabbits. Brain Res 1378:125–136
Jacobs TP, Kempski O, McKinley D, Dutka AJ, Hallenbeck JM, Feuerstein G (1992) Blood flow and vascular permeability during motor dysfunction in a rabbit model of spinal cord ischemia. Stroke 23:367–373
Moore WM Jr, Hollier LH (1991) The influence of severity of spinal cord ischemia in the etiology of delayed-onset paraplegia. Ann Surg 213:427–432
Wisselink W, Patetsios P, Panetta TF et al (1998) Medium molecular weight pentastarch reduces reperfusion injury by decreasing capillary leak in an animal model of spinal cord ischemia. J Vasc Surg 27:109–116
Moon SM, Kim W, Chung JY et al (2014) Neuroprotective effects of adipose-derived stem cells are maintained for 3 weeks against ischemic damage in the rabbit spinal cord. Biomed Res Int 2014:539051
Robertson CS, Goodman JC, Grossman RG, Priessman A (1990) Reduction in spinal cord postischemic lactic acidosis and functional improvement with dichloroacetate. J Neurotrauma 7:1–12
Grochot-Przeczek A, Dulak J, Jozkowicz A (2012) Haem oxygenase-1: non-canonical roles in physiology and pathology. Clin Sci (Lond) 122:93–103
Morse D, Choi AM (2002) Heme oxygenase-1: the “emerging molecule” has arrived. Am J Respir Cell Mol Biol 27:8–16
Morris MC, Depollier J, Mery J, Heitz F, Divita G (2001) A peptide carrier for the delivery of biologically active proteins into mammalian cells. Nat Biotechnol 19:1173–1176
Denicourt C, Dowdy SF (2003) Protein transduction technology offers novel therapeutic approach for brain ischemia. Trends Pharmacol Sci 24:216–218
Eum WS, Kim DW, Hwang IK et al (2004) In vivo protein transduction: biologically active intact pep-1-superoxide dismutase fusion protein efficiently protects against ischemic insult. Free Radic Biol Med 37:1656–1669
Yune TY, Lee JY, Jiang MH, Kim DW, Choi SY, Oh TH (2008) Systemic administration of PEP-1-SOD1 fusion protein improves functional recovery by inhibition of neuronal cell death after spinal cord injury. Free Radic Biol Med 45:1190–1200
Kim W, Kim DW, Yoo DY et al (2012) Neuroprotective effects of PEP-1-Cu, Zn-SOD against ischemic neuronal damage in the rabbit spinal cord. Neurochem Res 37:307–313
van der Knaap MS, van der Voorn P, Barkhof F et al (2003) A new leukoencephalopathy with brainstem and spinal cord involvement and high lactate. Ann Neurol 53:252–258
Gürer B, Kertmen H, Kasim E et al (2015) Neuroprotective effects of testosterone on ischemia/reperfusion injury of the rabbit spinal cord. Injury 46:240–248
Kertmen H, Gürer B, Yılmaz ER et al (2013) The protective effect of low-dose methotrexate on ischemia-reperfusion injury of the rabbit spinal cord. Eur J Pharmacol 714:148–156
Sanli AM, Serbes G, Sargon MF et al (2012) Methothrexate attenuates early neutrophil infiltration and the associated lipid peroxidation in the injured spinal cord but does not induce neurotoxicity in the uninjured spinal cord in rats. Acta Neurochir (Wien) 154:1045–1054
Taoka Y, Okajima K, Uchiba M et al (1997) Role of neutrophils in spinal cord injury in the rat. Neuroscience 79:1177–1182
Yilmaz ER, Kertmen H, Dolgun H et al (2012) Effects of darbepoetin-α in spinal cord ischemia-reperfusion injury in the rabbit. Acta Neurochir (Wien) 154:1037–1043
Jazwa A, Cuadrado A (2010) Targeting heme oxygenase-1 for neuroprotection and neuroinflammation in neurodegenerative diseases. Curr Drug Targets 11:1517–1531
Konrad FM, Knausberg U, Höne R, Ngamsri KC, Reutershan J (2015) Tissue heme oxygenase-1 exerts anti-inflammatory effects on LPS-induced pulmonary inflammation. Mucosal Immunol. doi:10.1038/mi.2015.39
Shah ZA, Nada SE, Doré S (2011) Heme oxygenase 1, beneficial role in permanent ischemic stroke and in Gingko biloba (EGb 761) neuroprotection. Neuroscience 180:248–255
Ilhan A, Koltuksuz U, Ozen S, Uz E, Ciralik H, Akyol O (1999) The effects of caffeic acid phenethyl ester (CAPE) on spinal cord ischemia/reperfusion injury in rabbits. Eur J Cardiothorac Surg 16:458–463
Ilhan A, Yilmaz HR, Armutcu F, Gurel A, Akyol O (2004) The protective effect of nebivolol on ischemia/reperfusion injury in rabbit spinal cord. Prog Neuropsychopharmacol Biol Psychiatry 28:1153–1160
Morita K, Lee MS, Her S (2009) Possible relation of hemin-induced HO-1 expression to the upregulation of VEGF and BDNF mRNA levels in rat C6 glioma cells. J Mol Neurosci 38:31–40
Kiprianova I, Freiman TM, Desiderato S et al (1999) Brain-derived neurotrophic factor prevents neuronal death and glial activation after global ischemia in the rat. J Neurosci Res 56:21–27
Shi Q, Zhang P, Zhang J et al (2009) Adenovirus-mediated brain-derived neurotrophic factor expression regulated by hypoxia response element protects brain from injury of transient middle cerebral artery occlusion in mice. Neurosci Lett 465:220–225
Acknowledgments
This work was supported by Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Education (NRF-2013R1A1A2059364) and also supported by Priority Research Centers Program grant from the National Research Foundation (NRF-2009-0093812) funded by the Ministry of Science, ICT & Future Planning in the Republic of Korea.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Dae Young Yoo and Dae Won Kim have contributed equally to this article.
Rights and permissions
About this article
Cite this article
Jung, H.Y., Kim, D.W., Yim, H.S. et al. Heme Oxygenase-1 Protects Neurons from Ischemic Damage by Upregulating Expression of Cu,Zn-Superoxide Dismutase, Catalase, and Brain-Derived Neurotrophic Factor in the Rabbit Spinal Cord. Neurochem Res 41, 869–879 (2016). https://doi.org/10.1007/s11064-015-1764-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11064-015-1764-1