Abstract
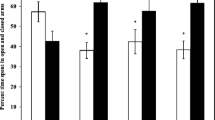
The effect of Arsenosugar on motor function and contextual memory-related to place and event; the extent of DNA damage and oxidative stress in male swiss albino mice was investigated. Passive avoidance test was used for memory test; rota motor test was used for motor function. Several biochemical parameters were used for assessing oxidative stress due to arsenosugar ingestion. Decreased passive avoidance time and decreased retention time in rotating rod indicated disruption of normal neurobehavior. Significant dose-dependent DNA damage was found in mice blood and brain. Decreased super oxide dismutase, increased lipid peroxidation, decreased protein sulfohydryl content, increased protein carbonyl content in blood and hippocampal tissue; glutathione in blood and glutathione peroxidase in hippocampal tissue indicated the ability of arsenosugar to cause oxidative stress. This study concludes with evidence that arsenosugar ingestion causes higher oxidative stress, increases DNA damage in the blood and hippocampus in vivo. This might be responsible for the dysfunction of cognitive and motor functions. However, further investigation is suggested for deciphering the biomolecular mechanism.


Similar content being viewed by others
References
Shimbo S, Hayase A, Murakami M, Hatai I, Higashikawa K, Moon CS, Zhang ZW, Watanabe T, Iguchi H, Ikeda M (1996) Use of a food composition database to estimate daily dietary intake of nutrient or trace elements in Japan, with reference to its limitation. Food Addit Contam 13:775–786
Hansen HR, Raab A, Francesconi KA, Feldmann J (2003) Metabolism of arsenic by sheep chronically exposed to arseno-sugars as a normal part of their diet: quantitative intake, uptake and excretion. Environ Sci Technol 37:845–851
Andrewes P, Demarini DM, Funasaka K, Wallace K, Lai VW, Sun H, Cullen WR, Kitchin KT (2004) Do arsenosugars pose a risk to human health? the comparative toxicities of a trivalent and pentavalent arsenosugar. Environ Sci Technol 38:4140–4148
WHO (1989) Evaluation of certain food additives and contaminants; 33rd Report of the Joint FAO/WHO Expert Committee on Food Additives; WHO: Geneva
Francesconi KA, Tanggaar R, McKenzie CJ, Goessler W (2002) Arsenic metabolites in human urine after ingestion of an arsenosugar. Clin Chem 48:92–101
Ma M, Le XC (1998) Effect of arsenosugar ingestion on urinary arsenic speciation. Clin Chem 44:539–550
Le XC, Cullen WR, Reimer KJ (1994) Human urinary arsenic excretion after one-time ingestion of seaweed, crab, and shrimp. Clin Chem 40:617–624
Wei C, Li W, Zhang C, Van Hulle M, Cornelis R, Zhang X (2003) Safety evaluation of organoarsenical species in edible porphyra from the China Sea. J Agric Food Chem 51:5176–5182
Erickson BE (2003) New concerns about arsenosugars in seaweed and shellfish. Environ Sci Technol 37:84A
Koch I, McPherson K, Smith P, Easton L, Doe KG, Reimer KJ (2007) Arsenic bioaccessibility and speciation in clams and seaweed from a contaminated marine environment. Mar Pollut Bull 54:586–594
Ha SH, Yeun JH, Kim J, Joo JD, Lee LY (2009) New synthetic method of natural arsenosugar. Bull Korean Chem Soc 30:997–998
World Medical Association Declaration of Helsinki. Ethical Principles for Medical Research Involving Human Subjects. Adopted by the 18th WMA General Assembly, Helsinki, Finland, June 1964, and amended by the 59th WMA General Assembly Seoul, South Korea, October, 2008. http://www.wma.net/en/30publications/10policies/b3/index.html. Accessed 1 August, 2012
Piala JJ, High JP, Hessert JLJ, Burke JC, Crower BN (1959) Pharmacological and acute toxicological comparisons of trifluoropromazine and chlorpromazine. J Pharmacol Exp Ther 127:55–65
Kuribara H, Higuchi Y, Tadokoro S (1977) Effects of central depressants on rotarod and tractionperformances in mice. Jpn J Pharmacol 27:117–126
Dunham NW, Meya TS (1957) A note on simple apparatus for detecting neurological defects in rats and mice. J Am Pharm Assoc 46:208–209
Singh NP, McCoy MT, Tice RR, Schneider EL (1988) A simple technique for quantitation of low levels of DNA damage in individual cells. Exp Cell Res 175:184–191
Nadin SB, Vargas-Roig LM, Ciocca DR (2001) A silver staining method for single cell gel assay. J Histochem Cytochem 49:1183–1186
Beauchamp C, Fridovich I (1971) Superoxide dismutase: improved assays and an assay applicable to acrylamide gels. Anal Biochem 44:276–287
Samir M, el-Kholy NM (1999) Thiobarbituric acid reactive substances in patients with laryngeal cancer. Clin Otolaryngol Allied Sci 24:232–234
Ohkawa H, Ohishi N, Yagi K (1979) Assay for lipid peroxides in animal tissues by thiobarbituric acid reaction. Anal Biochem 95:351–358
Ellman GL (1959) Tissue sulfhydryl groups. Arch Biochem Biophys 82:70–77
Levine RL, Garland D, Oliver CN, Amici A, Climent I, Lenz AG et al (1990) Determination of carbonyl content in oxidatively modified proteins. Methods Enzymol 186:464–478
Flora SJS, Bhadauria S, Panta SC, Dhaked RK (2005) Arsenic induced blood and brain oxidative stress and its response to some thiol chelators in rats. Life Sci 77:2324–2337
Jollow DJ, Mitchell JR, Zamppaglione Z, Gillette JR (1974) Bromobenzene induced liver necrosis. Protective role of glutathione and evidence for 3,4-bromobenzene oxide as the hepatotoxic metabolites. Pharmacology 11:151–157
Klaassen CD, Watkins JB (2003) Casarett and Doull’s essentials of toxicology. McGraw-Hill: New York. p 512. ISBN. 978-0-07-138914–3
McAdam DP, Perera AMA, Stick RV (1987) The synthesis of (R)-2′,3′-dihydroxypropyl 5-deoxy-5-dimethylarsinyl-β-D-riboside, a naturally occurring arsenic-containing carbohydrate. Aust J Chem 40:1901–1908
Stick RV, Stubbs KA, Tilbrook DMG (2001) An improved synthesis of (R)-2,3-dihydroxypropyl 5-deoxy-5-dimethylarsinyl-β-D-riboside, a common marine arsenical. Aust J Chem 54:181–183
Cullen WR, McBride BC, Reglinski J (1984) The reduction of trimethylarsine oxide to trimethylarsine by thiols: a mechanistic model for the biological reduction of arsenical. J Inorg Biochem 21:45–60
Kannan GM, Tripathi N, Dube SN, Gupta M, Flora SJ (2001) Toxic effects of arsenic (III) on some hematopoietic and central nervous system variables in rats and guinea pigs. J Toxicol Clin Toxicol 39:675–682
Warburton DM (1975) Brain, behaviour and drugs. Wiley, London
Jomova K, Jenisova Z, Feszterova M, Baros S, Liska J, Hudecova D, Rhodesd CJ, Valko M (2011) Arsenic: toxicity, oxidative stress and human disease. J Appl Toxicol 31:95–107
Münch G, Mayer S, Michaelis J, Hipkiss AR, Riederer P, Müller R, Neumann A, Schinzel R, Cunningham AM (1997) Influence of advanced glycation end-products and AGE-inhibitors on nucleation-dependent polymerization of beta-amyloid peptide. Biochim Biophys Acta 1360:17–29
Münch G, Deuther-Conrad W, Gasic-Milenkovic J (2002) Glycoxidative stress creates a vicious cycle of neurodegeneration in alzheimer’s disease–a target for neuroprotective treatment strategies? J Neural Transm Suppl 620:303–307
Gong G, O’Bryant SE (2010) The arsenic exposure hypothesis for alzheimer disease. Alzheimer Dis Assoc Disord 24:311–316
Acknowledgments
The first and corresponding author (Muhammad Shahdaat Bin Sayeed) was awarded travel grant for presenting partial result of the study in the 35th Annual Meeting of Japan Neuroscience Society, 18-21 September, 2012 in Nagoya, Japan.
Conflict of interest
None.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Bin Sayeed, M.S., Ratan, M., Hossen, F. et al. Arsenosugar Induced Blood and Brain Oxidative Stress, DNA Damage and Neurobehavioral Impairments. Neurochem Res 38, 405–412 (2013). https://doi.org/10.1007/s11064-012-0934-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11064-012-0934-7