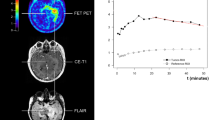
Abstract
Most high-grade gliomas (HGG) recur after initial multimodal therapy and re-irradiation (Re-RT) has been shown to be a valuable re-treatment option in selected patients. We evaluated the prognostic value of dynamic time-to-peak analysis and early static summation images in O-(2-18F-fluoroethyl)-l-tyrosine (18F-FET) PET for patients treated with Re-RT ± concomitant bevacizumab. We retrospectively analyzed 72 patients suffering from recurrent HGG with 18F-FET PET prior to Re-RT. PET analysis revealed the maximal tumor-to-background-ratio (TBRmax), the biological tumor volume, the number of PET-foci and pattern of time-activity-curves (TACs; increasing vs. decreasing). Furthermore, the novel PET parameters early TBRmax (at 5–15 min post-injection) and minimal time-to-peak (TTPmin) were evaluated. Additional analysis was performed for gender, age, KPS, O6-methylguanine-DNA methyltransferase methylation status, isocitrate dehydrogenase 1 mutational status, WHO grade and concomitant bevacizumab therapy. The influence of PET and clinical parameters on post-recurrence survival (PRS) was investigated. Shorter TTPmin was related to shorter PRS after Re-RT with 6 months for TTPmin < 12.5 min, 7 months for TTPmin 12.5–25 min and 11 months for TTPmin >25 min (p = 0.027). TTPmin had a significant impact on PRS both on univariate (p = 0.027; continuous) and multivariate analysis (p = 0.011; continuous). Other factors significantly related to PRS on multivariate analysis were increasing vs. decreasing TACs (p = 0.008) and Karnofsky Performance Score (p = 0.015; <70 vs. ≥70). Early TBRmax as well as the other conventional PET parameters were not significantly related to PRS on univariate analysis. Dynamic 18F-FET PET with TTPmin provides a high prognostic value for recurrent HGG prior to Re-RT, whereas early TBRmax does not. Dynamic 18F-FET PET using TTPmin might help to personalize Re-RT treatment regimens in future through voxelwise TTPmin analysis for dose painting purposes and PET-guided dose escalation.


Similar content being viewed by others
References
Stupp R, Hegi ME, Mason WP, van den Bent MJ, Taphoorn MJ, Janzer RC, Ludwin SK, Allgeier A, Fisher B, Belanger K, Hau P, Brandes AA, Gijtenbeek J, Marosi C, Vecht CJ, Mokhtari K, Wesseling P, Villa S, Eisenhauer E, Gorlia T, Weller M, Lacombe D, Cairncross JG, Mirimanoff RO (2009) Effects of radiotherapy with concomitant and adjuvant temozolomide versus radiotherapy alone on survival in glioblastoma in a randomised phase III study: 5-year analysis of the EORTC-NCIC trial. Lancet Oncol 10:459–466. doi:10.1016/s1470-2045(09)70025-7
Stupp R, Mason WP, van den Bent MJ, Weller M, Fisher B, Taphoorn MJ, Belanger K, Brandes AA, Marosi C, Bogdahn U, Curschmann J, Janzer RC, Ludwin SK, Gorlia T, Allgeier A, Lacombe D, Cairncross JG, Eisenhauer E, Mirimanoff RO (2005) Radiotherapy plus concomitant and adjuvant temozolomide for glioblastoma. N Engl J Med 352:987–996. doi:10.1056/NEJMoa043330
Gebhardt BJ, Dobelbower MC, Ennis WH, Bag AK, Markert JM, Fiveash JB (2014) Patterns of failure for glioblastoma multiforme following limited-margin radiation and concurrent temozolomide. Radiat Oncol 9: 130 doi:10.1186/1748-717x-9-130
Niyazi M, Siefert A, Schwarz SB, Ganswindt U, Kreth F-W, Tonn J-C, Belka C (2011) Therapeutic options for recurrent malignant glioma. Radiother Oncol 98:1–14
Flieger M, Ganswindt U, Schwarz SB, Kreth F-W, Tonn J-C, la Fougère C, Ertl L, Linn J, Herrlinger U, Belka C (2014) Re-irradiation and bevacizumab in recurrent high-grade glioma: an effective treatment option. J Neurooncol 117:337–345
Hundsberger T, Brügge D, Putora PM, Weder P, Weber J, Plasswilm L (2013) Re-irradiation with and without bevacizumab as salvage therapy for recurrent or progressive high-grade gliomas. J Neurooncol 112:133–139
Niyazi M, Jansen NL, Rottler M, Ganswindt U, Belka C (2014) Recurrence pattern analysis after re-irradiation with bevacizumab in recurrent malignant glioma patients. Radiat Oncol 9: 299 doi:10.1186/s13014-014-0299-y
Hartmann C, Hentschel B, Simon M, Westphal M, Schackert G, Tonn JC, Loeffler M, Reifenberger G, Pietsch T, von Deimling A (2013) Long-term survival in primary glioblastoma with versus without isocitrate dehydrogenase mutations. Clin Cancer Res 19:5146–5157
Hegi ME, Diserens AC, Gorlia T, Hamou MF, de Tribolet N, Weller M, Kros JM, Hainfellner JA, Mason W, Mariani L, Bromberg JE, Hau P, Mirimanoff RO, Cairncross JG, Janzer RC, Stupp R (2005) MGMT gene silencing and benefit from temozolomide in glioblastoma. N Engl J Med 352:997–1003. doi:10.1056/NEJMoa043331
Ohgaki H, Kleihues P (2011) Genetic profile of astrocytic and oligodendroglial gliomas. Brain Tumor Pathol 28:177–183
Harris RJ, Cloughesy TF, Pope WB, Nghiemphu PL, Lai A, Zaw T, Czernin J, Phelps ME, Chen W, Ellingson BM (2012) 18F-FDOPA and 18F-FLT positron emission tomography parametric response maps predict response in recurrent malignant gliomas treated with bevacizumab. Neuro Oncol 14:1079–1089. doi:10.1093/neuonc/nos141
Schwarzenberg J, Czernin J, Cloughesy TF, Ellingson BM, Pope WB, Geist C, Dahlbom M, Silverman DH, Satyamurthy N, Phelps ME, Chen W (2012) 3′-deoxy-3′-18F-fluorothymidine PET and MRI for early survival predictions in patients with recurrent malignant glioma treated with bevacizumab. J Nucl Med 53:29–36. doi:10.2967/jnumed.111.092387
Schwarzenberg J, Czernin J, Cloughesy TF, Ellingson BM, Pope WB, Grogan T, Elashoff D, Geist C, Silverman DH, Phelps ME, Chen W (2014) Treatment response evaluation using 18F-FDOPA PET in patients with recurrent malignant glioma on bevacizumab therapy. Clin Cancer Res 20:3550–3559. doi:10.1158/1078-0432.ccr-13-1440
Harris RJ, Cloughesy TF, Hardy AJ, Liau LM, Pope WB, Nghiemphu PL, Lai A, Ellingson BM (2015) MRI perfusion measurements calculated using advanced deconvolution techniques predict survival in recurrent glioblastoma treated with bevacizumab. J Neurooncol 122:497–505. doi:10.1007/s11060-015-1755-8
Huang RY, Rahman R, Ballman KV, Felten SJ, Anderson SK, Ellingson BM, Nayak L, Lee EQ, Abrey LE, Galanis E, Reardon DA, Pope WB, Cloughesy TF, Wen PY (2016) The Impact of T2/FLAIR evaluation per RANO criteria on Response assessment of recurrent glioblastoma patients treated with bevacizumab. Clin Cancer Res 22:575–581. doi:10.1158/1078-0432.ccr-14-3040
Leu K, Enzmann DR, Woodworth DC, Harris RJ, Tran AN, Lai A, Nghiemphu PL, Pope WB, Cloughesy TF, Ellingson BM (2014) Hypervascular tumor volume estimated by comparison to a large-scale cerebral blood volume radiographic atlas predicts survival in recurrent glioblastoma treated with bevacizumab. Cancer Imaging 14:31. doi:10.1186/s40644-014-0031-z
Niyazi M, Geisler J, Siefert A, Schwarz SB, Ganswindt U, Garny S, Schnell O, Suchorska B, Kreth F-W, Tonn J-C, Bartenstein P, Fougère Cl, Belka C (2011) FET–PET for malignant glioma treatment planning. Radiother Oncol 99:44–48. doi:10.1016/j.radonc.2011.03.001
Piroth M, Pinkawa M, Holy R, Stoffels G, Demirel C, Attieh C, Kaiser H, Langen K, Eble M (2009) Integrated-boost IMRT or 3-D-CRT using FET-PET based auto-contoured target volume delineation for glioblastoma multiforme: a dosimetric comparison. Radiat Oncol 4:57
Wyss M, Hofer S, Bruehlmeier M, Hefti M, Uhlmann C, Bärtschi E, Buettner U, Roelcke U (2009) Early metabolic responses in temozolomide treated low-grade glioma patients. J Neurooncol 95:87–93. doi:10.1007/s11060-009-9896-2
Miwa K, Matsuo M, Ogawa S, Shinoda J, Yokoyama K, Yamada J, Yano H, Iwama T (2014) Re-irradiation of recurrent glioblastoma multiforme using 11 C-methionine PET/CT/MRI image fusion for hypofractionated stereotactic radiotherapy by intensity modulated radiation therapy. Radiat Oncol 9: 181 doi:10.1186/1748-717x-9-181
Albert NL, Winkelmann I, Suchorska B, Wenter V, Schmid-Tannwald C, Mille E, Todica A, Brendel M, Tonn JC, Bartenstein P, la Fougere C (2016) Early static (18)F-FET-PET scans have a higher accuracy for glioma grading than the standard 20–40 min scans. Eur J Nucl Med Mol Imaging 43:1105–1114. doi:10.1007/s00259-015-3276-2
Niyazi M, Jansen N, Ganswindt U, Schwarz S, Geisler J, Schnell O, Büsing K, Eigenbrod S, la Fougère C, Belka C (2012) Re-irradiation in recurrent malignant glioma: prognostic value of [18F]FET–PET. J Neurooncol 110:389–395. doi:10.1007/s11060-012-0980-7
Jansen NL, Suchorska B, Wenter V, Schmid-Tannwald C, Todica A, Eigenbrod S, Niyazi M, Tonn J-C, Bartenstein P, Kreth F-W, la Fougère C (2015) Prognostic significance of dynamic 18F-FET PET in newly diagnosed astrocytic high-grade glioma. J Nucl Med 56:9–15. doi:10.2967/jnumed.114.144675
Suchorska B, Tonn JC, Jansen NL (2014) PET imaging for brain tumor diagnostics. Curr Opin Neurol 27:683–688. doi:10.1097/wco.0000000000000143
Hutterer M, Nowosielski M, Putzer D, Waitz D, Tinkhauser G, Kostron H, Muigg A, Virgolini IJ, Staffen W, Trinka E, Gotwald T, Jacobs AH, Stockhammer G (2011) O-(2-18F-fluoroethyl)-L-tyrosine PET predicts failure of antiangiogenic treatment in patients with recurrent high-grade glioma. J Nucl Med 52:856–864. doi:10.2967/jnumed.110.086645
Galldiks N, Rapp M, Stoffels G, Fink GR, Shah NJ, Coenen HH, Sabel M, Langen KJ (2013) Response assessment of bevacizumab in patients with recurrent malignant glioma using [18F]Fluoroethyl-L-tyrosine PET in comparison to MRI. Eur J Nucl Med Mol Imaging 40:22–33. doi:10.1007/s00259-012-2251-4
Wen PY, Macdonald DR, Reardon DA, Cloughesy TF, Sorensen AG, Galanis E, DeGroot J, Wick W, Gilbert MR, Lassman AB, Tsien C, Mikkelsen T, Wong ET, Chamberlain MC, Stupp R, Lamborn KR, Vogelbaum MA, van den Bent MJ, Chang SM (2010) updated response assessment criteria for high-grade gliomas: response assessment in neuro-oncology working group. J Clin Oncol 28:1963–1972. doi:10.1200/jco.2009.26.3541
Jansen NL, Graute V, Armbruster L, Suchorska B, Lutz J, Eigenbrod S, Cumming P, Bartenstein P, Tonn J-C, Kreth FW (2012) MRI-suspected low-grade glioma: is there a need to perform dynamic FET PET? Eur J Nucl Med Mol Imaging 39:1021–1029
Mehrkens J, Pöpperl G, Rachinger W, Herms J, Seelos K, Tatsch K, Tonn J, Kreth F (2008) The positive predictive value of O-(2-[18F] fluoroethyl)-L-tyrosine (FET) PET in the diagnosis of a glioma recurrence after multimodal treatment. J Neurooncol 88:27–35
Suchorska B, Jansen NL, Linn J, Kretzschmar H, Janssen H, Eigenbrod S, Simon M, Popperl G, Kreth FW, la Fougere C, Weller M, Tonn JC (2015) Biological tumor volume in 18FET-PET before radiochemotherapy correlates with survival in GBM. Neurology 84:710–719. doi:10.1212/wnl.0000000000001262
Niyazi M, Ganswindt U, Schwarz SB, Kreth F-W, Tonn J-C, Geisler J, la Fougère C, Ertl L, Linn J, Siefert A (2012) Irradiation and bevacizumab in high-grade glioma retreatment settings. Int J Radiat Oncol Biol Phys 82:67–76
Jager PL, Vaalburg W, Pruim J, de Vries EGE, Langen K-J, Piers DA (2001) Radiolabeled amino acids: basic aspects and clinical applications in oncology. J Nucl Med 42:432–445
Hutterer M, Nowosielski M, Putzer D, Jansen NL, Seiz M, Schocke M, McCoy M, Göbel G, la Fougère C, Virgolini IJ, Trinka E, Jacobs AH, Stockhammer G (2013) [18F]-fluoro-ethyl-l-tyrosine PET: a valuable diagnostic tool in neuro-oncology, but not all that glitters is glioma. Neuro Oncol 15:341–351. doi:10.1093/neuonc/nos300
Piroth MD, Holy R, Pinkawa M, Stoffels G, Kaiser HJ, Galldiks N, Herzog H, Coenen HH, Eble MJ, Langen KJ (2011) Prognostic impact of postoperative, pre-irradiation (18)F-fluoroethyl-l-tyrosine uptake in glioblastoma patients treated with radiochemotherapy. Radiother Oncol 99:218–224. doi:10.1016/j.radonc.2011.03.006
Acknowledgements
We thank Alexander Nieto for proofreading the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Daniel F. Fleischmann and Marcus Unterrainer have contributed equally to this work.
Rights and permissions
About this article
Cite this article
Fleischmann, D.F., Unterrainer, M., Bartenstein, P. et al. 18F-FET PET prior to recurrent high-grade glioma re-irradiation—additional prognostic value of dynamic time-to-peak analysis and early static summation images?. J Neurooncol 132, 277–286 (2017). https://doi.org/10.1007/s11060-016-2366-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11060-016-2366-8