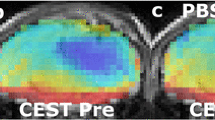
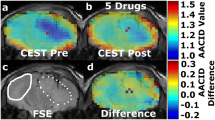
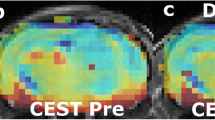
Abstract
Reversal of the intracellular/extracellular pH gradient is a hallmark of malignant tumors and is an important consideration in evaluating tumor growth potential and the effectiveness of anticancer therapies. Glioblastoma multiforme (GBM) brain tumors have increased expression of the carbonic anhydrase (CA) isozymes CAII, CAIX and CAXII that contribute to the altered regulation of intracellular pH (pHi). The anti-epileptic drug topiramate (TPM) inhibits CA action and may acidify the tumor intracellular compartment. In-vivo detection of acute tumor acidification could aid in cancer diagnosis and monitoring treatment response. Chemical exchange saturation transfer (CEST) magnetic resonance imaging (MRI) has been used to measure tissue pH. Using a recently developed CEST-MRI method called amine/amide concentration independent detection (AACID), we have previously shown intracellular acidification caused by single dose of lonidamine. The current study aims to evaluate the intracellular acidification induced by a single dose of the clinically approved drug TPM. Brain tumors were induced in NU/NU mice by injecting 105 U87 human glioblastoma multiforme cells into the right frontal lobe. Using a 9.4T MRI scanner AACID measurements were acquired, before and after administration of TPM (dose: 120 mg/kg, intraperitoneal), 15 ± 2 days after tumor cell implantation. TPM administration induced acute intracellular acidification (average ± SD: baseline AACID = 1.14 ± 0.05; post AACID = 1.19 ± 0.05, paired ttest p = 0.02) in implanted brain tumors. In contrast, contralateral tissue showed no change in AACID value. These results suggest that topiramate can rapidly induce a tumor specific physiological change detectable by AACID CEST. This pH challenge paradigm could be exploited to aid in tumor detection and monitoring treatment response.





Similar content being viewed by others
References
Kanu OO, Mehta A, Di C, Lin N, Bortoff K, Bigner DD, Yan H, Adamson DC (2009) Glioblastoma multiforme: a review of therapeutic targets. Expert Opin Ther Targets 13:701–718. doi:10.1517/14728220902942348
Surawicz TS, Davis F, Freels S, Laws ER Jr, Menck HR (1998) Brain tumor survival: results from the National Cancer Data Base. J Neurooncol 40:151–160
Adamson C, Kanu OO, Mehta AI, Di C, Lin N, Mattox AK, Bigner DD (2009) Glioblastoma multiforme: a review of where we have been and where we are going. Expert Opin Investig Drugs 18:1061–1083. doi:10.1517/13543780903052764
Mason WP, Maestro RD, Eisenstat D, Forsyth P, Fulton D, Laperriere N, Macdonald D, Perry J, Thiessen B (2007) Canadian recommendations for the treatment of glioblastoma multiforme. Curr Oncol 14:110–117
Parsons DW, Jones S, Zhang X, Lin JC, Leary RJ, Angenendt P, Mankoo P, Carter H, Siu IM, Gallia GL, Olivi A, McLendon R, Rasheed BA, Keir S, Nikolskaya T, Nikolsky Y, Busam DA, Tekleab H, Diaz LA Jr, Hartigan J, Smith DR, Strausberg RL, Marie SK, Shinjo SM, Yan H, Riggins GJ, Bigner DD, Karchin R, Papadopoulos N, Parmigiani G, Vogelstein B, Velculescu VE, Kinzler KW (2008) An integrated genomic analysis of human glioblastoma multiforme. Science 321:1807–1812
Stupp R, Hegi ME, Mason WP, van den Bent MJ, Taphoorn MJ, Janzer RC, Ludwin SK, Allgeier A, Fisher B, Belanger K, Hau P, Brandes AA, Gijtenbeek J, Marosi C, Vecht CJ, Mokhtari K, Wesseling P, Villa S, Eisenhauer E, Gorlia T, Weller M, Lacombe D, Cairncross JG, Mirimanoff RO (2009) Effects of radiotherapy with concomitant and adjuvant temozolomide versus radiotherapy alone on survival in glioblastoma in a randomised phase III study: 5-year analysis of the EORTC-NCIC trial. Lancet Oncol 10:459–466
Wen PY, Kesari S (2008) Malignant gliomas in adults. N Engl J Med 359:492–507
Kumar AJ, Leeds NE, Fuller GN, Van Tassel P, Maor MH, Sawaya RE, Levin VA (2000) Malignant gliomas: MR imaging spectrum of radiation therapy- and chemotherapy-induced necrosis of the brain after treatment. Radiology 217:377–384. doi:10.1148/radiology.217.2.r00nv36377
Webb BA, Chimenti M, Jacobson MP, Barber DL (2011) Dysregulated pH: a perfect storm for cancer progression. Nat Rev Cancer 11:671–677
Simon S, Roy D, Schindler M (1994) Intracellular pH and the control of multidrug resistance. Proc Natl Acad Sci USA 91:1128–1132
Neri D, Supuran CT (2011) Interfering with pH regulation in tumours as a therapeutic strategy. Nat Rev Drug Discov 10:767–777
Swietach P, Hulikova A, Vaughan-Jones RD, Harris AL (2010) New insights into the physiological role of carbonic anhydrase IX in tumour pH regulation. Oncogene 29:6509–6521
Supuran T, Claudiu (2003) Carbonic Anhydrase Inhibitors. Med Res Rev 23:146–189. doi:10.1002/med.10025
Chiche J, Ilc K, Laferriere J, Trottier E, Dayan F, Mazure NM, Brahimi-Horn MC, Pouyssegur J (2009) Hypoxia-inducible carbonic anhydrase IX and XII promote tumor cell growth by counteracting acidosis through the regulation of the intracellular pH. Cancer Res 69:358–368
Parkkila AK, Herva R, Parkkila S, Rajaniemi H (1995) Immunohistochemical demonstration of human carbonic anhydrase isoenzyme II in brain tumours. Histochem J 27:974–982
Haapasalo J, Nordfors K, Jarvela S, Bragge H, Rantala I, Parkkila AK, Haapasalo H, Parkkila S (2007) Carbonic anhydrase II in the endothelium of glial tumors: a potential target for therapy. Neuro-oncology 9:308–313
Vandenberg JI, Carter ND, Bethell HW, Nogradi A, Ridderstrale Y, Metcalfe JC, Grace AA (1996) Carbonic anhydrase and cardiac pH regulation. Am J Physiol 271:C1838–C1846
Parks SK, Chiche J, Pouyssegur J (2011) pH control mechanisms of tumor survival and growth. J Cell Physiol 226:299–308. doi:10.1002/jcp.22400
Gieling RG, Parker CA, De Costa LA, Robertson N, Harris AL, Stratford IJ, Williams KJ (2013) Inhibition of carbonic anhydrase activity modifies the toxicity of doxorubicin and melphalan in tumour cells in vitro. J Enzyme Inhib Med Chem 28:360–369. doi:10.3109/14756366.2012.736979
Parkkila S, Rajaniemi H, Parkkila AK, Kivela J, Waheed A, Pastorekova S, Pastorek J, Sly WS (2000) Carbonic anhydrase inhibitor suppresses invasion of renal cancer cells in vitro. Proc Natl Acad Sci USA 97:2220–2224. doi:10.1073/pnas.040554897
Mardor Y, Kaplan O, Sterin M, Ruiz-Cabello J, Ash E, Roth Y, Ringel I, Cohen JS (2000) Noninvasive real-time monitoring of intracellular cancer cell metabolism and response to lonidamine treatment using diffusion weighted proton magnetic resonance spectroscopy. Cancer Res 60:5179–5186
McVicar N, Li AX, Goncalves DF, Bellyou M, Meakin SO, Prado MA, Bartha R (2014) Quantitative tissue pH measurement during cerebral ischemia using amine and amide concentration-independent detection (AACID) with MRI. J Cereb Blood Flow Metab 34:690–698. doi:10.1038/jcbfm.2014.12
Desmond K, Stanisz G (2013) pH mapping based on the ratiometric amide and amine relationship from endogenous CEST. Contrast Media Mol Imaging doi:10.1002/cmmi.1522
Zong X, Wang P, Kim SG, Jin T (2013) Sensitivity and source of amine-proton exchange and amide-proton transfer magnetic resonance imaging in cerebral ischemia. Magn Reson Med. doi:10.1002/mrm.24639
Zhou J, Payen J-F, Wilson DA, Traystman RJ, van Zijl PCM (2003) Using the amide proton signals of intracellular proteins and peptides to detect pH effects in MRI. Nat Med 9:1085–1090
Zhou J, Lal B, Wilson DA, Laterra J, van Zijl PC (2003) Amide proton transfer (APT) contrast for imaging of brain tumors. Magn Reson Med 50:1120–1126. doi:10.1002/mrm.10651
Murray RK, Granner DK (2003) Membranes: structure & function. In: Murray RK, Granner DK, Mayes PA, Rodwell VW (eds) Harper’s illustrated biochemistry, 26th edn. McGraw-Hill Companies, Inc., New York, pp 415–433
McVicar N, Li AX, Meakin SO, Bartha R (2015) Imaging chemical exchange saturation transfer (CEST) effects following tumor-selective acidification using lonidamine. NMR Biomed 28:566–575. doi:10.1002/nbm.3287
Aggarwal M, Kondeti B, McKenna R (2013) Anticonvulsant/antiepileptic carbonic anhydrase inhibitors: a patent review. Expert opinion on therapeutic patents 23:717–724. doi:10.1517/13543776.2013.782394
Kim M, Gillen J, Landman BA, Zhou J, van Zijl PC (2009) Water saturation shift referencing (WASSR) for chemical exchange saturation transfer (CEST) experiments. Magn Reson Med 61:1441–1450. doi:10.1002/mrm.21873
Kogan F, Hariharan H, Reddy R (2013) Chemical exchange saturation transfer (CEST) imaging: description of technique and potential clinical applications. Curr Radiol Rep 1: 102–114 doi:10.1007/s40134-013-0010-3
Zhou J, Payen JF, Wilson DA, Traystman RJ, van Zijl PC (2003) Using the amide proton signals of intracellular proteins and peptides to detect pH effects in MRI. Nat Med 9:1085–1090. doi:10.1038/nm907
Haapasalo JA, Nordfors KM, Hilvo M, Rantala IJ, Soini Y, Parkkila AK, Pastorekova S, Pastorek J, Parkkila SM, Haapasalo HK (2006) Expression of carbonic anhydrase IX in astrocytic tumors predicts poor prognosis. Clin Cancer Res 12:473–477
Haapasalo J, Hilvo M, Nordfors K, Haapasalo H, Parkkila S, Hyrskyluoto A, Rantala I, Waheed A, Sly WS, Pastorekova S, Pastorek J, Parkkila AK (2008) Identification of an alternatively spliced isoform of carbonic anhydrase XII in diffusely infiltrating astrocytic gliomas. Neuro. Oncol 10:131–138
Rich IN, Worthington-White D, Garden OA, Musk P (2000) Apoptosis of leukemic cells accompanies reduction in intracellular pH after targeted inhibition of the Na(+)/H(+) exchanger. Blood 95:1427–1434
Nath K, Nelson DS, Ho AM, Lee SC, Darpolor MM, Pickup S, Zhou R, Heitjan DF, Leeper DB, Glickson JD (2013) (31) P and (1) H MRS of DB-1 melanoma xenografts: lonidamine selectively decreases tumor intracellular pH and energy status and sensitizes tumors to melphalan. NMR Biomed 26:98–105. doi:10.1002/nbm.2824
Cianchi F, Vinci MC, Supuran CT, Peruzzi B, De Giuli P, Fasolis G, Perigli G, Pastorekova S, Papucci L, Pini A, Masini E, Puccetti L (2010) Selective inhibition of carbonic anhydrase IX decreases cell proliferation and induces ceramide-mediated apoptosis in human cancer cells. J Pharmacol Exp Ther 334:710–719
Song CW, Lyons JC, Griffin RJ, Makepeace CM, Cragoe EJ Jr (1993) Increase in thermosensitivity of tumor cells by lowering intracellular pH. Cancer Res 53:1599–1601
Kim GE, Lyons JC, Song CW (1991) Effects of amiloride on intracellular pH and thermosensitivity. Int J Radiat Oncol Biol Phys 20:541–549
Ma B, Xiang Y, Li T, Yu HM, Li XJ (2004) Inhibitory effect of topiramate on Lewis lung carcinoma metastasis and its relation with AQP1 water channel. Acta Pharmacol Sin 25:54–60
Lyons JC, Kim GE, Song CW (1992) Modification of intracellular pH and thermosensitivity. Radiat Res 129:79–87
Morgan PE, Supuran CT, Casey JR (2004) Carbonic anhydrase inhibitors that directly inhibit anion transport by the human Cl-/HCO3- exchanger, AE1. Mol Membr Biol 21:423–433
Leniger T, Thone J, Wiemann M (2004) Topiramate modulates pH of hippocampal CA3 neurons by combined effects on carbonic anhydrase and Cl-/HCO3- exchange. Br J Pharmacol 142:831–842. doi:10.1038/sj.bjp.0705850
Lou Y, McDonald PC, Oloumi A, Chia S, Ostlund C, Ahmadi A, Kyle A, Auf dem Keller U, Leung S, Huntsman D, Clarke B, Sutherland BW, Waterhouse D, Bally M, Roskelley C, Overall CM, Minchinton A, Pacchiano F, Carta F, Scozzafava A, Touisni N, Winum JY, Supuran CT, Dedhar S (2011) Targeting tumor hypoxia: suppression of breast tumor growth and metastasis by novel carbonic anhydrase IX inhibitors. Cancer Res 71:3364–3376
Pacchiano F, Carta F, McDonald PC, Lou Y, Vullo D, Scozzafava A, Dedhar S, Supuran CT (2011) Ureido-substituted benzenesulfonamides potently inhibit carbonic anhydrase IX and show antimetastatic activity in a model of breast cancer metastasis. J Med Chem 54:1896–1902. doi:10.1021/jm101541x
Weller M, Stupp R, Wick W (2012) Epilepsy meets cancer: when, why, and what to do about it? Lancet Oncol 13:e375–e382
Harguindey S, Arranz JL, Orozco JDP, Rauch C, Fais S, Cardone RA, Reshkin SJ (2013) Cariporide and other new and powerful NHE1 inhibitors as potentially selective anticancer drugs–an integral molecular/biochemical/metabolic/clinical approach after one hundred years of cancer research. J Transl Med 11:282. doi:10.1186/1479-5876-11-282
Miranda-Goncalves V, Honavar M, Pinheiro C, Martinho O, Pires MM, Pinheiro C, Cordeiro M, Bebiano G, Costa P, Palmeirim I, Reis RM, Baltazar F (2013) Monocarboxylate transporters (MCTs) in gliomas: expression and exploitation as therapeutic targets. Neuro-oncology 15:172–188. doi:10.1093/neuonc/nos298
Perez-Sayans M, Somoza-Martin JM, Barros-Angueira F, Rey JM, Garcia-Garcia A (2009) V-ATPase inhibitors and implication in cancer treatment. Cancer Treat Rev 35:707–713. doi:10.1016/j.ctrv.2009.08.003
Acknowledgments
We gratefully thank technical support of C. O’Neil. This work was funded by the Ontario Institute of Cancer Research (OICR) Smarter Imaging Program, and a Discovery Grant from the National Science and Engineering Research Council of Canada (NSERC).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declares no conflict of interest.
Ethical approval
All applicable national and institutional guidelines for the care and use of animals were followed. All procedures performed in studies involving animals were in accordance with the ethical standards of the institution or practice at which the studies were conducted.
Rights and permissions
About this article
Cite this article
Marathe, K., McVicar, N., Li, A. et al. Topiramate induces acute intracellular acidification in glioblastoma. J Neurooncol 130, 465–472 (2016). https://doi.org/10.1007/s11060-016-2258-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11060-016-2258-y