Abstract
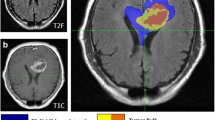
There is a distinct diversity between the appearance of every glioblastoma multiforme (GBM) on pretreatment magnetic resonance imaging (MRI) with a potential impact on clinical outcome and survival of the patients. The object of this study was to determine the impact of 10 different single nucleotide polymorphisms (SNPs) on various volumetric parameters in patients harboring a GBM. We prospectively analyzed 20 steroid-naïve adult patients who had been treated for newly diagnosed GBM. The volumetry was performed using MRI with the help of a semiautomated quantitative software measuring contrast enhancing tumor volume including necrosis, central necrosis alone and peritumoral edema (PTE). We calculated ratios between the tumor volume and edema (ETR), respectively necrosis (NTR). SNP analysis was done using genomic DNA extracted from peripheral blood genotyped via PCR and sequencing. There was a strong correlation between tumor volume and PTE (p < 0.001), necrosis (p < 0.001) and NTR (p = 0.003). Age and sex had no influence on volumetric data. The Aquaporin 4-31G > A SNP had a significant influence on the ETR (p = 0.042) by decreasing the measured edema compared with the tumor volume. The Interleukin 8-251A > T SNP was significantly correlated with an increased tumor (p = 0.048), PTE (p = 0.033) and necrosis volume (p = 0.028). We found two SNPs with a distinct impact on pretreatment tumor characteristics, presenting a potential explanation for the individual diversity of GBM appearance on MRI and influence on survival.

Similar content being viewed by others
References
Ostrom QT, Bauchet L, Davis FG et al (2014) The epidemiology of glioma in adults: a “state of the science” review. Neuro Oncol 16(7):896–913
Iliadis G, Kotoula V, Chatzisotiriou A et al (2012) Volumetric and MGMT parameters in glioblastoma patients: survival analysis. BMC Cancer 12(3):3
Crawford FW, Khayal IS, McGue C et al (2009) Relationship of pre-surgery metabolic and physiological MR imaging parameters to survival for patients with untreated GBM. J Neurooncol 91(3):337–351
Nestler U, Lutz K, Pichlmeier U, Stummer W et al (2015) Anatomic features of glioblastoma and their potential impact on survival. Acta Neurochir (Wien) 157(2):179–186
Liu SY, Mei WZ, Lin ZX (2013) Pre-operative peritumoral edema and survival rate in glioblastoma multiforme. Onkologie 36(11):679–684
Dubois LG, Campanati L, Righy C et al (2014) Gliomas and the vascular fragility of the blood brain barrier. Front Cell Neurosci 8:418
Nico B, Ribatti D (2011) Role of aquaporins in cell migration and edema formation in human brain tumors. Exp Cell Res 317(17):2391–2396
Saadoun S, Papadopoulos MC, Davies DC, Bell BA, Krishna S (2002) Increased aquaporin 1 water channel expression in human brain tumours. Br J Cancer 87(6):621–623
Saadoun S, Papadopoulos MC, Davies DC, Krishna S, Bell BA (2002) Aquaporin-4 expression is increased in oedematous human brain tumours. J Neurol Neurosurg Psychiatry 72(2):262–265
Warth A, Simon P, Capper D et al (2007) Expression pattern of the water channel aquaporin-4 in human gliomas is associated with blood-brain barrier disturbance but not with patient survival. J Neurosci Res 85(6):1336–1346
Kim J, Jung Y (2011) Different expressions of AQP1, AQP4, eNOS, and VEGF proteins in ischemic versus non-ischemic cerebropathy in rats: potential roles of AQP1 and eNOS in hydrocephalic and vasogenic edema formation. Anat Cell Biol 44(4):295–303
Lee MD, Bhakta KY, Raina S et al (1996) The human Aquaporin-5 gene. Molecular characterization and chromosomal localization. J Biol Chem 271(15):8599–8604
Weyerbrock A, Walbridge S, Saavedra JE, Keefer LK, Oldfield EH (2011) Differential effects of nitric oxide on blood-brain barrier integrity and cerebral blood flow in intracerebral C6 gliomas. Neuro Oncol 13(2):203–211
Yeung YT, McDonald KL, Grewal T, Munoz L (2013) Interleukins in glioblastoma pathophysiology: implications for therapy. Br J Pharmacol 168(3):591–606
Carneiro-Lobo TC, Lima MT, Mariano-Oliveira A et al (2014) Expression of tissue factor signaling pathway elements correlates with the production of vascular endothelial growth factor and interleukin-8 in human astrocytoma patients. Oncol Rep 31(2):679–686
Poirier J, Miron J, Picard C et al (2014) Apolipoprotein E and lipid homeostasis in the etiology and treatment of sporadic Alzheimer’s disease. Neurobiol Aging 35(suppl 2):S3–S10
Horsburgh K, McCarron MO, White F, Nicoll JA (2000) The role of apolipoprotein E in Alzheimer’s disease, acute brain injury and cerebrovascular disease: evidence of common mechanisms and utility of animal models. Neurobiol Aging 21(2):245–255
Nicoll JA, Zunarelli E, Rampling R, Murray LS, Papanastassiou V, Stewart J (2003) Involvement of apolipoprotein E in glioblastoma: immunohistochemistry and clinical outcome. NeuroReport 14(15):1923–1926
Louis DN, Ohgaki H, Wiestler OD et al (2007) The 2007 WHO classification of tumours of the central nervous system. Acta Neuropathol 114(2):97–109
Sawaya R, Hammoud M, Schoppa D et al (1998) Neurosurgical outcomes in a modern series of 400 craniotomies for treatment of parenchymal tumors. Neurosurgery 42(5):1044–1055
Rigau V, Zouaoui S, Mathieu-Daudé H et al (2011) French brain tumor database: 5-year histological results on 25 756 cases. Brain Pathol 21(6):633–644
Lacroix M, Abi-Said D, Fourney DR et al (2001) A multivariate analysis of 416 patients with glioblastoma multiforme: prognosis, extent of resection, and survival. J Neurosurg 95(2):190–198
Grabowski MM, Recinos PF, Nowacki AS et al (2014) Residual tumor volume versus extent of resection: predictors of survival after surgery for glioblastoma. J Neurosurg 121(5):1115–1123
Bennet AM, Di Angelantonio E, Ye Z, Wensley F, Dahlin A, Ahlbom A, Keavney B, Collins R, Wiman B, de Faire U, Danesh J (2007) Association of apolipoprotein E genotypes with lipid levels and coronary risk. JAMA 298(11):1300–1311
Papadopoulos MC, Verkman AS (2013) Aquaporin water channels in the nervous system. Nat Rev Neurosci 14(4):265–277
Papadopoulos MC, Verkman AS (2005) Aquaporin-4 gene disruption in mice reduces brain swelling and mortality in pneumococcal meningitis. J Biol Chem 280(14):13906–13912
Zador Z, Stiver S, Wang V, Manley GT (2009) Role of aquaporin-4 in cerebral edema and stroke. Handb Exp Pharmacol 190:159–170
Abreu-Rodríguez I, Sánchez Silva R, Martins AP et al (2011) Functional and transcriptional induction of aquaporin-1 gene by hypoxia; analysis of promoter and role of Hif-1α. PLoS One 6(12):e28385
Brat DJ, Bellail AC, Van Meir EG (2005) The role of interleukin-8 and its receptors in gliomagenesis and tumoral angiogenesis. Neuro Oncol 7(2):122–133
Cooper LA, Gutman DA, Chisolm C et al (2012) The tumor microenvironment strongly impacts master transcriptional regulators and gene expression class of glioblastoma. Am J Pathol 180(5):2108–2119
Anand M, Brat DJ (2012) Oncogenic regulation of tissue factor and thrombosis in cancer. Thromb Res 129(suppl 1):S46–S49
Mou K, Chen M, Mao Q et al (2010) AQP-4 in peritumoral edematous tissue is correlated with the degree of glioma and with expression of VEGF and HIF-alpha. J Neurooncol 100(3):375–383
Sorani MD, Zador Z, Hurowitz E, Yan D, Giacomini KM, Manley GT (2008) Novel variants in human Aquaporin-4 reduce cellular water permeability. Hum Mol Genet 17(15):2379–2389
Kleffner I, Bungeroth M, Schiffbauer H, Schäbitz WR, Ringelstein EB, Kuhlenbäumer G (2008) The role of aquaporin-4 polymorphisms in the development of brain edema after middle cerebral artery occlusion. Stroke 39(4):1333–1335
Chaichana KL, Cabrera-Aldana EE, Jusue-Torres I et al (2014) When gross total resection of a glioblastoma is possible, how much resection should be achieved? World Neurosurg 82(1–2):257–265
Kelly PJ, Daumas-Duport C, Kispert DB, Kall BA, Scheithauer BW, Illig JJ (1987) Imaging-based stereotaxic serial biopsies in untreated intracranial glial neoplasms. J Neurosurg 66(6):865–874
Sun S, Wang Q, Giang A et al (2011) Knockdown of CypA inhibits interleukin-8 (IL-8) and IL-8-mediated proliferation and tumor growth of glioblastoma cells through down-regulated NF-κB. J Neurooncol 101(1):1–14
Rak J, Milsom C, Magnus N, Yu J (2009) Tissue factor in tumour progression. Best Pract Res Clin Haematol 22(1):71–83
Ahn MH, Park BL, Lee SH et al (2011) A promoter SNP rs4073T > A in the common allele of the interleukin 8 gene is associated with the development of idiopathic pulmonary fibrosis via the IL-8 protein enhancing mode. Respir Res 12:73
Lee WP, Tai DI, Lan KH et al (2005) The -251T allele of the interleukin-8 promoter is associated with increased risk of gastric carcinoma featuring diffuse-type histopathology in Chinese population. Clin Cancer Res 11(18):6431–6441
Hammoud MA, Sawaya R, Shi W, Thall PF, Leeds NE (1996) Prognostic significance of preoperative MRI scans in glioblastoma multiforme. J Neurooncol 27(1):65–73
Acknowledgments
We thank Dr. Maryam Sherman for her kind help in editing our draft.
Funding
No financial support was provided for this study.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
No conflicts of interest to disclose.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Rights and permissions
About this article
Cite this article
Henker, C., Kriesen, T., Fürst, K. et al. Effect of 10 different polymorphisms on preoperative volumetric characteristics of glioblastoma multiforme. J Neurooncol 126, 585–592 (2016). https://doi.org/10.1007/s11060-015-2005-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11060-015-2005-9