Abstract
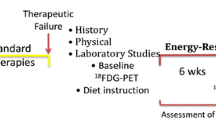
Retrospective data suggests that low serum glucose levels during the treatment of glioblastoma multiforme (GBM) may improve clinical outcomes. As such, many patients are implementing a ketogenic diet (KD) in order to decrease serum glucose flux while simultaneously elevating circulating ketones during radiation therapy and chemotherapy for the treatment of GBM. With IRB approval, a retrospective review of patients with high-grade glioma treated with concurrent chemoradiotherapy and adjuvant chemotherapy was carried out from August 2010 to April 2013. Serum glucose and ketone levels, dexamethasone dose, and toxicity of patients undergoing a KD during treatment were also assessed. Blood glucose levels were compared between patients on an unspecified/standard diet and a KD. Toxicity was assessed by Common Terminology Criteria for Adverse Events version 4. In total, 53 patients were analyzed. Six underwent a KD during treatment. The diet was well tolerated with no grade III toxicity and one episode of grade II fatigue. No episodes of symptomatic hypoglycemia were experienced. Four patients are alive at a median follow-up of 14 months. The mean blood glucose of patients on a standard diet was 122 versus 84 mg/dl for those on a KD. Based on this retrospective study, a KD appears safe and well tolerated during the standard treatment of GBM. Dietary restriction of carbohydrates through a KD reduces serum glucose levels significantly, even in conjunction with high dose steroids, which may affect the response to standard treatment and prognosis. Larger prospective trials to confirm this relationship are warranted.

Similar content being viewed by others
References
Stupp R, Mason WP, van den Bent MJ, Weller M, Fisher B, Taphoorn MJB, Belanger K, Brandes AA, Marosi C, Bogdahn U, Curschmann J, Janzer RC, Ludwin SK, Gorlia T, Allgeier A, Lacombe D, Cairncross JG, Eisenhauer E, Mirimanoff RO (2005) Radiotherapy plus concomitant and adjuvant temozolomide for glioblastoma. N Engl J Med 352:987–996. doi:10.1056/NEJMoa043330
Warburg O (1956) On the origin of cancer cells. Science 123:309–314
Yamamoto T, Seino Y, Fukumoto H, Koh G, Yano H, Inagaki N, Yamada Y, Inoue K, Manabe T, Imura H (1990) Over-expression of facilitative glucose transporter genes in human cancer. Biochem Biophys Res Commun 170:223–230
Mathupala SP, Ko YH, Pedersen PL (2009) Hexokinase-2 bound to mitochondria: cancer’s stygian link to the “Warburg Effect” and a pivotal target for effective therapy. Semin Cancer Biol 19:17–24. doi:10.1016/j.semcancer.2008.11.006
Kubicek GJ, Champ C, Fogh S, Wang F, Reddy E, Intenzo C, Dusing RW, Machtay M (2010) FDG-PET staging and importance of lymph node SUV in head and neck cancer. Head Neck Oncol 2:19. doi:10.1186/1758-3284-2-19
Maurer GD, Brucker DP, Bahr O, Harter PN, Hattingen E, Walenta S, Mueller-Klieser W, Steinbach JP, Rieger J (2011) Differential utilization of ketone bodies by neurons and glioma cell lines: a rationale for ketogenic diet as experimental glioma therapy. BMC Cancer 11:315. doi:10.1186/1471-2407-11-315
Seyfried TN, Kiebish M, Mukherjee P, Marsh J (2008) Targeting energy metabolism in brain cancer with calorically restricted ketogenic diets. Epilepsia 49:114–116. doi:10.1111/j.1528-1167.2008.01853.x
Derr RL, Ye X, Islas MU, Desideri S, Saudek CD, Grossman SA (2009) Association between hyperglycemia and survival in patients with newly diagnosed glioblastoma. J Clin Oncol 27:1082–1086. doi:10.1200/JCO.2008.19.1098
McGirt MJ, Chaichana KL, Gathinji M, Attenello F, Than K, Ruiz AJ, Olivi A, Quinones-Hinojosa A (2008) Persistent outpatient hyperglycemia is independently associated with decreased survival after primary resection of malignant brain astrocytomas. Neurosurgery 63:286–291. doi:10.1227/01.NEU.0000315282.61035.48
Fine EJ, Segal-Isaacson CJ, Feinman RD, Herszkopf S, Romano MC, Tomuta N, Bontempo AF, Negassa A, Sparano JA (2012) Targeting insulin inhibition as a metabolic therapy in advanced cancer: a pilot safety and feasibility dietary trial in 10 patients. Nutrition 28:1028–1035. doi:10.1016/j.nut.2012.05.001
Leino RL, Gerhart DZ, Duelli R, Enerson BE, Drewes LR (2001) Diet-induced ketosis increases monocarboxylate transporter (MCT1) levels in rat brain. Neurochem Int 38:519–527. doi:10.1016/s0197-0186(00)00102-9
Nehlig A (2004) Brain uptake and metabolism of ketone bodies in animal models. Prostaglandins Leukot Essent Fatty Acids 70:265–275. doi:10.1016/j.plefa.2003.07.006
Simanek R, Vormittag R, Hassler M, Roessler K, Schwarz M, Zielinski C, Pabinger I, Marosi C (2007) Venous thromboembolism and survival in patients with high-grade glioma. Neuro-oncology 9:89–95. doi:10.1215/15228517-2006-035
Den Heijer M, Lewington S, Clarke R (2005) Homocysteine, MTHFR and risk of venous thrombosis: a meta-analysis of published epidemiological studies. J Thromb Haemost 3:292–299. doi:10.1111/j.1538-7836.2005.01141.x
Amiel SA, Archibald HR, Chusney G, Williams AJ, Gale EA (1991) Ketone infusion lowers hormonal responses to hypoglycaemia: evidence for acute cerebral utilization of a non-glucose fuel. Clin Sci (Lond) 81:189–194
Volek J, Phinney S, Forsythe C, Quann E, Wood R, Puglisi M, Kraemer W, Bibus D, Fernandez M, Feinman R (2009) Carbohydrate restriction has a more favorable impact on the metabolic syndrome than a low fat diet. Lipids 44:297–309. doi:10.1007/s11745-008-3274-2
Fine EJ, Segal-Isaacson CJ, Feinman RD, Herszkopf S, Romano MC, Tomuta N, Bontempo AF, Negassa A, Sparano JA (2012) Targeting insulin inhibition as a metabolic therapy in advanced cancer: a pilot safety and feasibility dietary trial in 10 patients. Nutrition 28(10):1028–1035. doi:10.1016/j.nut.2012.05.001
Champ CE, Baserga R, Mishra MV, Jin L, Sotgia F, Lisanti MP, Pestell RG, Dicker AP, Simone NL (2013) Nutrient restriction and radiation therapy for cancer treatment: when less is more. Oncologist 18:97–103. doi:10.1634/theoncologist.2012-0164
Stafford P, Abdelwahab M, Kim D, Preul M, Rho J, Scheck A (2010) The ketogenic diet reverses gene expression patterns and reduces reactive oxygen species levels when used as an adjuvant therapy for glioma. Nutr Metab 7:74
Abdelwahab MG, Fenton KE, Preul MC, Rho JM, Lynch A, Stafford P, Scheck AC (2012) The ketogenic diet is an effective adjuvant to radiation therapy for the treatment of malignant glioma. PLoS ONE 7:e36197. doi:10.1371/journal.pone.0036197
Zuccoli G, Marcello N, Pisanello A, Servadei F, Vaccaro S, Mukherjee P, Seyfried T (2010) Metabolic management of glioblastoma multiforme using standard therapy together with a restricted ketogenic diet: case report. Nutr Metab 7:33
Nebeling LC, Lerner E (1995) Implementing a ketogenic diet based on medium-chain triglyceride oil in pediatric patients with cancer. J Am Diet Assoc 95:693–697
Baserga R (2000) The contradictions of the insulin-like growth factor 1 receptor. Oncogene 19:5574–5581. doi:10.1038/sj.onc.1203854
Resnicoff M, Coppola D, Sell C, Rubin R, Ferrone S, Baserga R (1994) Growth inhibition of human melanoma cells in nude mice by antisense strategies to the type 1 insulin-like growth factor receptor. Cancer Res 54:4848–4850
Macaulay VM (1992) Insulin-like growth factors and cancer. Br J Cancer 65:311–320
Perer ES, Madan AK, Shurin A, Zakris E, Romeguera K, Pang Y, Beech DJ (2000) Insulin-like growth factor I receptor antagonism augments response to chemoradiation therapy in colon cancer cells. J Surg Res 94:1–5. doi:10.1006/jsre.2000.5923S0022-4804(00)95923-7
Andrews DW, Resnicoff M, Flanders AE, Kenyon L, Curtis M, Merli G, Baserga R, Iliakis G, Aiken RD (2001) Results of a pilot study involving the use of an antisense oligodeoxynucleotide directed against the insulin-like growth factor type I receptor in malignant astrocytomas. J Clin Oncol 19:2189–2200
Turner BC, Haffty BG, Narayanan L, Yuan J, Havre PA, Gumbs AA, Kaplan L, Burgaud JL, Carter D, Baserga R, Glazer PM (1997) Insulin-like growth factor-I receptor overexpression mediates cellular radioresistance and local breast cancer recurrence after lumpectomy and radiation. Cancer Res 57:3079–3083
Champ CE, Volek JS, Siglin J, Jin L, Simone NL (2012) Weight gain, metabolic syndrome, and breast cancer recurrence: are dietary recommendations supported by the data? Int J Breast Cancer 2012:9. doi:10.1155/2012/506868
Klement RJ, Kammerer U (2011) Is there a role for carbohydrate restriction in the treatment and prevention of cancer? Nutr Metab (Lond) 8:75. doi:10.1186/1743-7075-8-75
Simone BA, Champ CE, Rosenberg AL, Berger AC, Monti DA, Dicker AP, Simone NL (2013) Selectively starving cancer cells through dietary manipulation: methods and clinical implications. Future Oncol 9:959–976. doi:10.2217/fon.13.31
Conflict of interest
None declared.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Champ, C.E., Palmer, J.D., Volek, J.S. et al. Targeting metabolism with a ketogenic diet during the treatment of glioblastoma multiforme. J Neurooncol 117, 125–131 (2014). https://doi.org/10.1007/s11060-014-1362-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11060-014-1362-0