Abstract
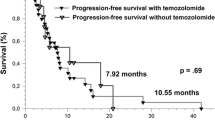
The current best standard care for glioblastoma still has limitations and unsatisfactory outcomes in patients with an unmethylated O-6-methylguanine-DNA methyltransferase (MGMT) promoter. Whether the effects of temozolomide are primarily due to its concomitant use with radiotherapy or are also mediated by their independent use in the adjuvant phase remain unclear. To validate the concomitant use of temozolomide in the standard protocol, we compared the overall survival of two prospective patient groups: one treated with radiotherapy alone followed by adjuvant temozolomide (RT → TMZ group) and the other treated with concomitant radiotherapy and temozolomide followed by adjuvant temozolomide (CCRT-TMZ group). Each patient in the RT → TMZ group (n = 25) was matched with two patients in the CCRT-TMZ group (n = 50) with respect to age, extent of resection, MGMT promoter methylation status, and postsurgical performance status to minimize the influence of confounding factors. In patients with MGMT promoter methylation, the CCRT-TMZ group showed superior overall survival (OS; median, 41.0 months) and progression-free survival (PFS; median, 24.0 months) compared with the RT → TMZ group. However, the OS and PFS did not differ between the CCRT-TMZ and the RT → TMZ groups in the patients without MGMT promoter methylation. Although this data is from a retrospective analysis using small number of patients, the study might indicate that concomitant use of temozolomide with radiotherapy is a crucial step in the standard treatment for glioblastoma patients with MGMT promoter methylation. And the use of temozolomide, either concurrently or by adjuvant after radiotherapy, remains a questionable value for those with an unmethylated MGMT promoter.



Similar content being viewed by others
References
Stupp R, Mason WP, van den Bent MJ et al (2005) Radiotherapy plus concomitant and adjuvant temozolomide for glioblastoma. N Engl J Med 352:987–996
Cohen MH, Johnson JR, Pazdur R (2005) Food and Drug Administration Drug approval summary: temozolomide plus radiation therapy for the treatment of newly diagnosed glioblastoma multiforme. Clin Cancer Res 11:6767–6771
Athanassiou H, Synodinou M, Maragoudakis E et al (2005) Randomized phase II study of temozolomide and radiotherapy compared with radiotherapy alone in newly diagnosed glioblastoma multiforme. J Clin Oncol: Off J Am Soc Clin Oncol 23:2372–2377
Lanzetta G, Campanella C, Rozzi A et al (2003) Temozolomide in radio-chemotherapy combined treatment for newly-diagnosed glioblastoma multiforme: phase II clinical trial. Anticancer res 23:5159–5164
Stupp R, Dietrich PY, Ostermann Kraljevic S et al (2002) Promising survival for patients with newly diagnosed glioblastoma multiforme treated with concomitant radiation plus temozolomide followed by adjuvant temozolomide. J Clin Oncol: Off J Am Soc Clin Oncol 20:1375–1382
Stupp R, Hegi ME, Mason WP et al (2009) Effects of radiotherapy with concomitant and adjuvant temozolomide versus radiotherapy alone on survival in glioblastoma in a randomised phase III study: 5-year analysis of the EORTC-NCIC trial. Lancet Oncol 10:459–466
Sher DJ, Henson JW, Avutu B et al (2008) The added value of concurrently administered temozolomide versus adjuvant temozolomide alone in newly diagnosed glioblastoma. J Neurooncol 88:43–50
Kim IH, Park CK, Heo DS et al (2011) Radiotherapy followed by adjuvant temozolomide with or without neoadjuvant ACNU-CDDP chemotherapy in newly diagnosed glioblastomas: a prospective randomized controlled multicenter phase III trial. J Neurooncol 103:595–602
Macdonald DR, Cascino TL, Schold SC Jr et al (1990) Response criteria for phase II studies of supratentorial malignant glioma. J Clin Oncol: Off J Am Soc Clin Oncol 8:1277–1280
Wen PY, Macdonald DR, Reardon DA et al (2010) Updated response assessment criteria for high-grade gliomas: response assessment in neuro-oncology working group. J Clin Oncol: Off J Am Soc Clin Oncol 28:1963–1972
Park CK, Kim J, Yim SY et al (2011) Usefulness of MS-MLPA for detection of MGMT promoter methylation in the evaluation of pseudoprogression in glioblastoma patients. Neuro-oncology 13:195–202
Hegi ME, Diserens AC, Gorlia T et al (2005) MGMT gene silencing and benefit from temozolomide in glioblastoma. N Engl J Med 352:997–1003
Chakravarti A, Erkkinen MG, Nestler U et al (2006) Temozolomide-mediated radiation enhancement in glioblastoma: a report on underlying mechanisms. Clin Cancer Res 12:4738–4746
Koukourakis GV, Kouloulias V, Zacharias G et al (2009) Temozolomide with radiation therapy in high grade brain gliomas: pharmaceuticals considerations and efficacy; a review article. Molecules 14:1561–1577
Kil WJ, Cerna D, Burgan WE et al (2008) In vitro and in vivo radiosensitization induced by the DNA methylating agent temozolomide. Clin Cancer Res 14:931–938
Wedge SR, Porteous JK, Glaser MG et al (1997) In vitro evaluation of temozolomide combined with X-irradiation. Anticancer Drugs 8:92–97
Hofer S, Lassman AB (2010) Molecular markers in gliomas: impact for the clinician. Target oncol 5:201–210
Acknowledgments
This research was supported by Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Education, Science and Technology (2012R1A1A2003779). No author received financial support in conjunction with the generation of this manuscript. The authors have no personal financial or institutional interest in any of the material described in this article.
Disclosure
No financial support or relationships that may pose a conflict of interest exist regarding this work.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Park, CK., Lee, SH., Kim, T.M. et al. The value of temozolomide in combination with radiotherapy during standard treatment for newly diagnosed glioblastoma. J Neurooncol 112, 277–283 (2013). https://doi.org/10.1007/s11060-013-1060-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11060-013-1060-3