Abstract
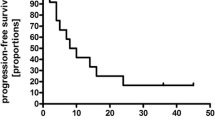
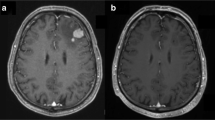
Treatment for patients with refractory or relapsed primary CNS lymphoma (PCNSL) remains unsatisfactory. Topotecan is an intravenous topoisomerase I inhibitor with good CSF penetration and documented efficacy in patients with relapsed systemic non-Hodgkin’s lymphoma. In this study 15 patients with refractory or relapsed PCNSL were treated with intravenous topotecan (1.5 mg/m2) for five consecutive days during each 21-day cycle. All 15 patients had measurable, contrast-enhancing tumor on cranial MRI at the time of relapse. Three (20%) patients achieved a complete response after one, three and four cycles, respectively, while three (20%) patients achieved a partial response after two cycles each, for a total response proportion of 40%. Three patients had stable disease at the end of topotecan treatment. Six patients (40%) had progressive disease during treatment. Median overall survival was 981 days (95% CI: 275, NA) and median progression free survival was 60 days (95% CI: 46, 945). Three out of 15 patients had grade 3 thrombocytopenia. Six out of 15 patients had grade 3 neutropenia, while 5/15 patients had grade 4 neutropenia, and 13/15 patients received g-CSF at some point during treatment. There were no deaths directly related to treatment toxicity. Our study shows that topotecan, as a salvage therapy in patients with relapsed or refractory PCNSL, is associated with an overall response proportion of 40% and should be considered in patients who have failed prior methotrexate-based chemotherapy and/or whole brain irradiation. However, progression is frequent and early and most patients required growth factor support due to myelotoxicity.
Similar content being viewed by others
References
CBTRUS, Central Brain Tumor Registry of the United States (2005–2006) Primary brain tumors in the United States. Statistical Report 1998–2002
Batchelor T, Carson K, O’Neill A et al (2003) Treatment of primary CNS lymphoma with methotrexate and deferred radiotherapy: a report of NABTT 96-07. J Clin Oncol 21:1044–1049
Macdonald DR, Cascino TL et al (1990) Response criteria of phase II studies for supratentorial malignant glioma. J Clin Oncol 8(7):1277–1280
Nelson DF, Martz KL, Bonner H, Nelson JS, Newall J, Kerman HD, Thomson LW, Murray KJ (1992) Non-Hodgkin’s lymphoma of the brain: can high dose, large volume radiation therapy improve survival? Report on prospective trial by the Radiation Therapy Oncology Group (RTOG): RTOG 8315. Int J Radiat Onc Biol Phys 23:9–17
Abrey LE, Yahalom J, DeAngelis LM (1997) Relapse and late neurotoxicity in primary central nervous system lymphoma. Neurology 48:A18
Nguyen PL, Chakravarti A, Finkelstein DM, Hochberg FH, Batchelor TT, Loeffler JS (2005) Results of whole-brain radiation as salvage of methotrexate failure for immunocompetent patients with primary CNS lymphoma. J Clin Oncol 23(7):1507–1513
Plotkin SR, Betensky RA, Hochberg FH, Grossman SA, Lesser GJ, Nabors LB, Chon B, Batchelor TT (2004) Treatment of relapsed central nervous system lymphoma with high-dose methotrexate. Clin Cancer Res 10(17):5643–5646
Herrlinger U, Brugger W, Bamberg M et al (2000) PCV salvage therapy for recurrent primary CNS lymphoma. Neurology 54:1707–1708
Reni M, Mason W, Zaja F et al (2004) Salvage chemotherapy with temozolomide in primary CNS lymphomas: preliminary results of a phase II trial. Eur J Cancer 40:1682–1688
Enting RH, Demopoulos A, DeAngelis LM, Abrey LE (2004) Salvage therapy for primary CNS lymphoma with a combination of temozolomide and rituximab. Neurology 63:901–903
Fischer L et al (2006) Prospective trial on topotecan salvage therapy in primary CNS lymphoma. Ann Oncol 17:1141–1145
Soussain C, Hoang-Xuan K, Levy V (2004) Results of intensive chemotherapy followed by hematopoietic stem-cell rescue in 22 patients with refractory or recurrent primary CNS lymphoma or intraocular lymphoma. Bull Cancer 91(2):189–192
Tyson RM, Siegal T, Doolittle ND, Lacy C, Kraemer DF, Neutwelt EA (2003) Current status and future of relapsed primary central nervous system lymphoma (PCNSL). Leuk Lymphoma 44(4):627–633
Bashir R, Freedman A, Harris N, Bain K, Nadler L, Hochberg F (1989) Immunophenotypic profile of CNS lymphoma: a review of eighteen cases. J Neurooncol 7(3):249–254
Raizer JJ, DeAngelis L, Zelenetz A, Abrey L (2000) Activity of rituximab in primary central nervous system lymphoma. Proc Am Soc Clin Oncol 19:166a
Sung C, Blaney SM, Cole DE et al (1994) A pharmacokinetic model of topotecan clearance from plasma and cerebrospinal fluid. Cancer Res 54:5118–5122
Preti HA, Plunkett W, Sarris AH, Younes A, Hagemeister F, Rodriguez MA, Romaguera J, McLaughlin P, Bachier C, Cabanillas F (1995) Preliminary results of a phase II trial of topotecan in patients with relapsing lymphoma. Blood 86(10, Suppl 1):820A
Wong ET, Berkenblit A (2004) The role of topotecan in the treatment of brain metastases. Oncologist 9(1):68–79
Voloschin A, Wen P, Hochberg F, Batchelor T (2004) Topotecan as salvage therapy for refractory or relapsed primary central nervous system lymphoma: final report. Neurology 62:A478
Acknowledgments
This study was supported by the Richard and Nancy Simches Fund for Brain Tumor Research.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Voloschin, A.D., Betensky, R., Wen, P.Y. et al. Topotecan as salvage therapy for relapsed or refractory primary central nervous system lymphoma. J Neurooncol 86, 211–215 (2008). https://doi.org/10.1007/s11060-007-9464-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11060-007-9464-6