Abstract
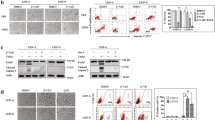
Here we aimed to evaluate the effects of DAXX subcellular localization on ox-LDL induced macrophages apoptosis. Cytoplasmic localization vector DAXX-W621A and nuclear localization vector DAXX-S667A were constructed by point mutation in DAXX. Blank vector, full length DAXX, DAXX-W621A, DAXX-S667A was transfect into RAW264.7 cells, respectively. Then the cells were incubated with 100 mg/ml ox-LDL for 48 h. Immunofluorescent assay was used to assay the localization of DAXX. MTT and Flow cytometry was used to determine cellular viability and apoptosis. RT-PCR and Western blot were used to analyze the expression levels. A significantly increased expression of DAXX was found in transfected cells of DAXX. The content of DAXX in nucleus was significantly increased in DAXX(S667A), and DAXX was significantly increased in cytoplasm of DAXX(W621A). Besides, we found DAXX was mainly expressed in nucleus with a low-level expression in cytoplasm through immunofluorescence. However in DAXX(W621A) group, the DAXX began to transferred to cytoplasm, which exhibited significant florescence. After treated with ox-LDL, the cytoactive of DAXX-W621A exhibited significantly decreased level when compared DAXX group. However, after added inhibitor LMB, the inhibition was relieved. The cell viability was also significantly increased in DAXX-S667A group. The results of apoptosis rates were similar in each group. Furthermore, we found the expression of ASK1 and JNK was also consistent with the apoptosis rates. Cytoplasmic localization of DAXX can increase injury sensitivity of ox-LDL on cells, and nuclear localization can antagonise the effect of ox-LDL. Besides, it is certified ox-LDL induced apoptosis is mainly through ASK1-JNK pathway.





Similar content being viewed by others
References
Libby P, Ridker PM, Hansson GK (2011) Progress and challenges in translating the biology of atherosclerosis. Nature 473(7347):317–325
Libby P, Okamoto Y, Rocha VZ, Folco E (2010) Inflammation in atherosclerosis. Circ J 74:213–220
Kawaguchi H, Yamada T, Miura N, Ayaori M, Uto-Kondo H, Ikegawa M, Noguchi M, Wang KY, Izumi H, Tanimoto A (2013) Rapid Development of Atherosclerosis in the World’s Smallest Microminipig Fed a High-Fat/High-Cholesterol Diet. J Atheroscler Thromb 21(3):186–203
Thorp E, Li Y, Bao L, Yao PM, Kuriakose G, Rong J, Fisher EA, Tabas I (2009) Brief report: increased apoptosis in advanced atherosclerotic lesions of Apoe −/− mice lacking macrophage Bcl-2. Arterioscler Thromb Vasc Biol 29(2):169–172
Huang Z, Liu Q, Li W, Wang R, Wang D, Zhang Y, Zhang F, Chi Y, Liu Z, Matsuura E (2010) 7-Ketocholesterol induces cell apoptosis by activation of nuclear factor kappa B in mouse macrophages. Acta Med Okayama 64(2):85–93
Geng Y-J, Phillips JE, Mason RP, Casscells SW (2003) Cholesterol crystallization and macrophage apoptosis: implication for atherosclerotic plaque instability and rupture. Biochem Pharmacol 66(8):1485–1492
Lim WS, Timmins JM, Seimon TA, Sadler A, Kolodgie FD, Virmani R, Tabas I (2008) Signal transducer and activator of transcription-1 is critical for apoptosis in macrophages subjected to endoplasmic reticulum stress in vitro and in advanced atherosclerotic lesions in vivo. Circulation 117(7):940–951
Tabas I (2005) Consequences and therapeutic implications of macrophage apoptosis in atherosclerosis the importance of lesion stage and phagocytic efficiency. Arterioscler Thromb Vasc Biol 25(11):2255–2264
Schwartz CJ, Valente AJ, Sprague EA, Kelley J, Nerem R (1991) The pathogenesis of atherosclerosis: an overview. Clin Cardiol 14(S1):1–16
Aviram M (2000) Review of human studies on oxidative damage and antioxidant protection related to cardiovascular diseases. Free Radic Res 33:S85
He QZ, Tuo QH, Zeng HC, Zhu BY, Rang WQ, Tang XQ, Liao DF (2010) Daxx mediates oxidized low-density lipoprotein-induced cholesterol accumulation and apoptosis in macrophages by upregulating caveolin-1 expression. Prog Biochem Biophy 37(8):881–890
Yao PM, Tabas I (2000) Free cholesterol loading of macrophages induces apoptosis involving the fas pathway. J Biol Chem 275(31):23807–23813
Tibbetts MD, Zheng L, Lenardo MJ (2003) The death effector domain protein family: regulators of cellular homeostasis. Nat Immunol 4(5):404–409
Salomoni P, Khelifi AF (2006) Daxx: death or survival protein? Trends Cell Biol 16(2):97–104
Qiu H, Fan Y, Joyee AG, Wang S, Han X, Bai H, Jiao L, Van Rooijen N, Yang X (2008) Type I IFNs enhance susceptibility to Chlamydia muridarum lung infection by enhancing apoptosis of local macrophages. J Immunol 181(3):2092–2102
Khelifi A, D’Alcontres MS, Salomoni P (2005) Daxx is required for stress-induced cell death and JNK activation. Cell Death Differ 12(7):724–733
Kitamura T, Fukuyo Y, Inoue M, Horikoshi NT, Shindoh M, Rogers BE, Usheva A, Horikoshi N (2009) Mutant p53 disrupts the stress MAPK activation circuit induced by ASK1-dependent stabilization of Daxx. Cancer Res 69(19):7681–7688
Roubille F, Combes S, Leal-Sanchez J, Barrère C, Cransac F, Sportouch-Dukhan C, Gahide G, Serre I, Kupfer E, Richard S (2007) Myocardial expression of a dominant-negative form of Daxx decreases infarct size and attenuates apoptosis in an in vivo mouse model of ischemia/reperfusion injury. Circulation 116(23):2709–2717
Ma Q, Devarajan P (2008) Induction of proapoptotic Daxx following ischemic acute kidney injury. Kidney Int 74(3):310–318
Song JJ, Lee YJ (2004) Tryptophan 621 and serine 667 residues of Daxx regulate its nuclear export during glucose deprivation. J Biol Chem 279(29):30573–30578
Tabas I (2004) Apoptosis and plaque destabilization in atherosclerosis: the role of macrophage apoptosis induced by cholesterol. Cell Death Differ 11:S12–S16
Kolodgie F, Virmani R, Burke A, Farb A, Weber D, Kutys R, Finn A, Gold H (2004) Pathologic assessment of the vulnerable human coronary plaque. Heart 90(12):1385–1391
Su B, Yang YB, Tuo QH, Zhu BY, Lei XY, Yin W, Liao DF (2007) Anti-apoptotic effects of probucol are associated with downregulation of Daxx expression in THP-1 macrophage. Cardiovasc Drugs Ther 21(1):37–45
Curtin JF, Cotter TG (2003) Live and let die: regulatory mechanisms in Fas-mediated apoptosis. Cell Signal 15(11):983–992
Yamada S, Ding Y, Tanimoto A, Wang KY, Guo X, Li Z, Tasaki T, Nabesima A, Murata Y, Shimajiri S (2011) Apoptosis signal-regulating kinase 1 deficiency accelerates hyperlipidemia-induced atheromatous plaques via suppression of macrophage apoptosis. Arterioscler Thromb Vasc Biol 31(7):1555–1564
Zhong S, Salomoni P, Ronchetti S, Guo A, Ruggero D, Pandolfi PP (2000) Promyelocytic leukemia protein (PML) and Daxx participate in a novel nuclear pathway for apoptosis. J Exp Med 191(4):631–640
Jung YS, Kim HY, Lee YJ, Kim E (2007) Subcellular localization of Daxx determines its opposing functions in ischemic cell death. FEBS Lett 581(5):843–852
Acknowledgments
This work was supported by Grants from the National Natural Science Foundation of China (31371161, 81173047 and 30971267), Hunan Province universities innovation platform of Open Fund Project (12K096), Hunan Province foundation for distinguished young scholar (14JJ1024) and the 12th Five Year Plan Key Discipline.
Conflict of interest
None.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Guozuo Xiong and Lin Li contributed equally to this work.
Rights and permissions
About this article
Cite this article
Xiong, G., Li, L., Sun, S. et al. Subcellular localization of DAXX influence ox-LDL induced apoptosis in macrophages. Mol Biol Rep 41, 7183–7190 (2014). https://doi.org/10.1007/s11033-014-3601-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11033-014-3601-z