Abstract
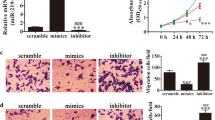
Abnormal cell proliferation is a main driver of tumor formation and development, which involves the deletion, mutation, and downregulation of tumor suppressor genes. One study recently demonstrated that miR-200a plays an oncogenic role by inhibiting phosphatase and tensin homolog deleted on chromosome ten (PTEN) expression. In the human endometrial adenocarcinoma cell line HEC-1B, suppression of miR-200a expression inhibited cell proliferation and promoted apoptosis, whereas its over-expression had no effect on proliferation and apoptosis. Furthermore, inhibition or over-expression of miR-200a increased or reduced the expression of PTEN, respectively, with no change in PTEN mRNA levels. These effects were achieved by directly targeting miR-200a to the 3′ untranslated region of the PTEN mRNA to inhibit its translation. Taken together, we propose that in HEC-1B cells, miR-200a functions as an oncogene, affecting proliferation and apoptosis by regulating the expression of the tumor suppressor PTEN at the translational level.






Similar content being viewed by others
References
Ray M, Fleming G (2009) Management of advanced-stage and recurrent endometrial cancer. Semin Oncol 36:145–154
Okuda T, Sekizawa A, Purwosunu Y, Nagatsuka M, Morioka M, Hayashi M, Okai T (2010) Genetics of endometrial cancers. Obstet Gynecol Int 2010:984013
Jemal A, Siegel R, Ward E, Hao Y, Xu J, Murray T, Thun MJ (2008) Cancer statistics, 2008. CA Cancer J Clin 58:71–96
Lee DY, Jeyapalan Z, Fang L, Yang J, Zhang Y, Yee AY, Li M, Du WW, Shatseva T, Yang BB (2010) Expression of versican 3′-untranslated region modulates endogenous microRNA functions. PLoS One 5:e13599
Stark A, Brennecke J, Bushati N, Russell RB, Cohen SM (2005) Animal MicroRNAs confer robustness to gene expression and have a significant impact on 3′UTR evolution. Cell 123:1133–1146
Bartel DP (2004) MicroRNAs: genomics, biogenesis, mechanism, and function. Cell 116:281–297
Tricoli JV, Jacobson JW (2007) MicroRNA: potential for cancer detection, diagnosis, and prognosis. Cancer Res 67:4553–4555
Negrini M, Ferracin M, Sabbioni S, Croce CM (2007) MicroRNAs in human cancer: from research to therapy. J Cell Sci 120:1833–1840
Kutay H, Bai S, Datta J, Motiwala T, Pogribny I, Frankel W, Jacob ST, Ghoshal K (2006) Downregulation of miR-122 in the rodent and human hepatocellular carcinomas. J Cell Biochem 99:671–678
Gaur A, Jewell DA, Liang Y, Ridzon D, Moore JH, Chen C, Ambros VR, Israel MA (2007) Characterization of microRNA expression levels and their biological correlates in human cancer cell lines. Cancer Res 67:2456–2468
Croce CM, Calin GA (2005) miRNAs, cancer, and stem cell division. Cell 122:6–7
Wu X, Zhao B, Li W, Chen Y, Liang R, Li L, Jin Y, Ruan K (2012) MiR-200a is involved in rat epididymal development by targeting beta-catenin mRNA. Acta Biochim Biophys Sin (Shanghai) 44:233–240
Soubani O, Ali AS, Logna F, Ali S, Philip PA, Sarkar FH (2013) Re-expression of miR-200 by novel approaches regulates the expression of PTEN and MT1-MMP in pancreatic cancer. Carcinogenesis 33:1563–1571
Mateescu B, Batista L, Cardon M, Gruosso T, de Feraudy Y, Mariani O, Nicolas A, Meyniel JP, Cottu P, Sastre-Garau X, Mechta-Grigoriou F (2011) miR-141 and miR-200a act on ovarian tumorigenesis by controlling oxidative stress response. Nat Med 17:1627–1635
Eades G, Yang M, Yao Y, Zhang Y, Zhou Q (2011) miR-200a regulates Nrf2 activation by targeting Keap1 mRNA in breast cancer cells. J Biol Chem 286:40725–40733
Li A, Omura N, Hong SM, Vincent A, Walter K, Griffith M, Borges M, Goggins M (2010) Pancreatic cancers epigenetically silence SIP1 and hypomethylate and overexpress miR-200a/200b in association with elevated circulating miR-200a and miR-200b levels. Cancer Res 70:5226–5237
Pan Q, Chegini N (2008) MicroRNA signature and regulatory functions in the endometrium during normal and disease states. Semin Reprod Med 26:479–493
Myatt SS, Wang J, Monteiro LJ, Christian M, Ho KK, Fusi L, Dina RE, Brosens JJ, Ghaem-Maghami S, Lam EW (2010) Definition of microRNAs that repress expression of the tumor suppressor gene FOXO1 in endometrial cancer. Cancer Res 70:367–377
Lee JW, Park YA, Choi JJ, Lee YY, Kim CJ, Choi C, Kim TJ, Lee NW, Kim BG, Bae DS (2011) The expression of the miRNA-200 family in endometrial endometrioid carcinoma. Gynecol Oncol 120:56–62
Hiroki E, Akahira J, Suzuki F, Nagase S, Ito K, Suzuki T, Sasano H, Yaegashi N (2010) Changes in microRNA expression levels correlate with clinicopathological features and prognoses in endometrial serous adenocarcinomas. Cancer Sci 101:241–249
Torres A, Torres K, Pesci A, Ceccaroni M, Paszkowski T, Cassandrini P, Zamboni G, Maciejewski R (2013) Diagnostic and prognostic significance of miRNA signatures in tissues and plasma of endometrioid endometrial carcinoma patients. Int J Cancer 132:1633–1645
Qian K, Chen H, Wei Y, Hu J, Zhu G (2005) Differentiation of endometrial stromal cells in vitro: down-regulation of suppression of the cell cycle inhibitor p57 by HOXA10? Mol Hum Reprod 11:245–251
Schmittgen TD, Jiang J, Liu Q, Yang L (2004) A high-throughput method to monitor the expression of microRNA precursors. Nucleic Acids Res 32:e43
Megraw M, Sethupathy P, Corda B, Hatzigeorgiou AG (2007) miRGen: a database for the study of animal microRNA genomic organization and function. Nucleic Acids Res 35:D149–D155
Lewis BP, Burge CB, Bartel DP (2005) Conserved seed pairing, often flanked by adenosines, indicates that thousands of human genes are microRNA targets. Cell 120:15–20
Krek A, Grun D, Poy MN, Wolf R, Rosenberg L, Epstein EJ, MacMenamin P, da Piedade I, Gunsalus KC, Stoffel M, Rajewsky N (2005) Combinatorial microRNA target predictions. Nat Genet 37:495–500
Osborne C, Wilson P, Tripathy D (2004) Oncogenes and tumor suppressor genes in breast cancer: potential diagnostic and therapeutic applications. Oncologist 9:361–377
Croce CM (2008) Oncogenes and cancer. N Engl J Med 358:502–511
Leslie NR, Foti M (2011) Non-genomic loss of PTEN function in cancer: not in my genes. Trends Pharmacol Sci 32:131–140
Yu J, Ryan DG, Getsios S, Oliveira-Fernandes M, Fatima A, Lavker RM (2008) MicroRNA-184 antagonizes microRNA-205 to maintain SHIP2 levels in epithelia. Proc Natl Acad Sci USA 105:19300–19305
Zbuk KM, Eng C (2007) Cancer phenomics: RET and PTEN as illustrative models. Nat Rev Cancer 7:35–45
Yan X, Fraser M, Qiu Q, Tsang BK (2006) Over-expression of PTEN sensitizes human ovarian cancer cells to cisplatin-induced apoptosis in a p53-dependent manner. Gynecol Oncol 102:348–355
Wang X, Trotman LC, Koppie T, Alimonti A, Chen Z, Gao Z, Wang J, Erdjument-Bromage H, Tempst P, Cordon-Cardo C, Pandolfi PP, Jiang X (2007) NEDD4-1 is a proto-oncogenic ubiquitin ligase for PTEN. Cell 128:129–139
Trotman LC, Wang X, Alimonti A, Chen Z, Teruya-Feldstein J, Yang H, Pavletich NP, Carver BS, Cordon-Cardo C, Erdjument-Bromage H, Tempst P, Chi SG, Kim HJ, Misteli T, Jiang X, Pandolfi PP (2007) Ubiquitination regulates PTEN nuclear import and tumor suppression. Cell 128:141–156
Schondorf T, Ebert MP, Hoffmann J, Becker M, Moser N, Pur S, Gohring UJ, Weisshaar MP (2004) Hypermethylation of the PTEN gene in ovarian cancer cell lines. Cancer Lett 207:215–220
Blumenthal GM, Dennis PA (2008) PTEN hamartoma tumor syndromes. Eur J Hum Genet 16:1289–1300
Salmena L, Carracedo A, Pandolfi PP (2008) Tenets of PTEN tumor suppression. Cell 133:403–414
Acknowledgments
We thank Prof. Yingxiong Wang for helpful suggestions, and Prof. Junlin He for assistance with the statistical analysis. This study was supported by the National Natural Science Foundation of China (Grant number 31271545). The National Key Clinical Department Funding (201101CKZD). The foundation played no role in the study’s design, the collection or analysis of data, the decision to publish, or the preparation of the manuscript.
Conflict interests
The authors declare no competing interests.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Li, R., He, Jl., Chen, Xm. et al. MiR-200a is involved in proliferation and apoptosis in the human endometrial adenocarcinoma cell line HEC-1B by targeting the tumor suppressor PTEN. Mol Biol Rep 41, 1977–1984 (2014). https://doi.org/10.1007/s11033-014-3045-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11033-014-3045-5