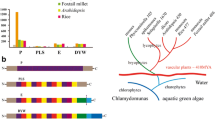
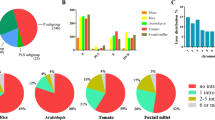
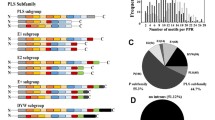
Abstract
Nucleus-encoded pentatricopeptide repeat proteins (PPRs) are a superfamily of ubiquitous RNA-binding factors involved in organellar gene expression. To date, a large number of PPR genes have been identified and some of them have been functionally analyzed in higher plants, while the family in maize [Zea mays (Zm)] has long been unknown. In this study, a genome-scale analysis was conducted to identify all ZmPPR gene members, and exon/intron structure, chromosomal mapping and gene duplication events were analyzed. Then, homology modeling was used to explore structure-function relationships. Finally, publically transcriptome data and real-time PCR were used for studying gene expression. A total of 521 PPR-encoding genes were predicted to occur in the maize genome and unevenly distributed on ten chromosomes. More than 66% PPR genes rarely contain introns and similar exon/intron structural patterns were observed in the same classes/subclasses. Three-dimensional (3D) structural modeling of ZmPPR479 provided insights into the mechanism of RNA recognition. The transcript abundance of 471 genes detected in 60 different developmental stages reveled that some members exhibit tissue-specific expression. Under salt stress, five significantly differentially expressed genes were found in crown root (CR) and ten detected in primary root (PR) and seminal root (SR), respectively. For drought stress, the expressions of seven ZmPPRs showed obvious upregulation in leaf, whereas ten genes were significantly downregulated. Additionally, the expression of 25 and 145 genes were evidently increased and decreased in cob, respectively. Furthermore, the expression patterns of 18 ZmPPRs were confirmed using quantitative real-time PCR.




Similar content being viewed by others
References
Arnal N, Quadrado M, Simon M, Mireau H (2014) A restorer-of-fertility like pentatricopeptide repeat gene directs ribonucleolytic processing within the coding sequence of rps 3-rpl 16 and orf 240a mitochondrial transcripts in Arabidopsis thaliana. Plant journal: for cell and. molecular biology 78(1):134–145
Bartel DP (2009) Micro RNAs: target recognition and regulatory functions. Cell 136(2):215–233
Bentolila S, Alfonso AA, Hanson MR (2002) A pentatricopeptide repeat-containing gene restores fertility to cytoplasmic male-sterile plants. Proc Natl Acad Sci U S A 99(16):10887–10892
Bentolila S, Heller WP, Sun T, Babina AM, Friso G, van Wijk KJ, Hanson MR (2012) RIP1, a member of an Arabidopsis protein family, interacts with the protein RARE1 and broadly affects RNA editing. Proc Natl Acad Sci U S A 109(22):E1453–E1461
Bosacchi M, Gurdon C, Maliga P (2015) Plastid genotyping reveals the uniformity of cytoplasmic male sterile-T maize cytoplasms. Plant Physiol 169(3):2129–2137
Boussardon C, Avon A, Kindgren P, Bond CS, Challenor M, Lurin C, Small I (2014) The cytidine deaminase signature HxE (x) n CxxC of DYW1 binds zinc and is necessary for RNA editing of ndh D-1. New Phytol 203(4):1090–1095
Chateigner-Boutin AL, Colas des Francs-Small C, Fujii S, Okuda K, Tanz SK, Small I (2013) The E domains of pentatricopeptide repeat proteins from different organelles are not functionally equivalent for RNA editing. The Plant journal: for cell and. molecular biology 74(6):935–945
Chateigner-Boutin AL, Ramos-Vega M, Guevara-Garcia A, Andres C, de la Luz G-NM, Cantero A, Delannoy E, Jimenez LF, Lurin C, Small I, Leon P (2008) CLB19, a pentatricopeptide repeat protein required for editing of rpo a and clp P chloroplast transcripts. The Plant journal: for cell and. molecular biology 56(4):590–602
Chen L, Liu YG (2014) Male sterility and fertility restoration in crops. Annu Rev Plant Biol 65:579–606
Coquille S, Filipovska A, Chia T, Rajappa L, Lingford JP, Razif MF, Thore S, Rackham O (2014) An artificial PPR scaffold for programmable RNA recognition. Nat Commun 5:5729
Desloire S, Gherbi H, Laloui W, Marhadour S, Clouet V, Cattolico L, Falentin C, Giancola S, Renard M, Budar F, Small I, Caboche M, Delourme R, Bendahmane A (2003) Identification of the fertility restoration locus, Rfo, in radish, as a member of the pentatricopeptide-repeat protein family. EMBO Rep 4(6):588–594
Ding YH, Liu NY, Tang ZS, Liu J, Yang WC (2006) Arabidopsis GLUTAMINE-RICH PROTEIN23 is essential for early embryogenesis and encodes a novel nuclear PPR motif protein that interacts with RNA polymerase II subunit III. Plant Cell 18(4):815–830
Doniwa Y, Ueda M, Ueta M, Wada A, Kadowaki K, Tsutsumi N (2010) The involvement of a PPR protein of the P subfamily in partial RNA editing of an Arabidopsis mitochondrial transcript. Gene 454(1–2):39–46
Duvick DN, Snyder RJ, Anderson EG (1961) The chromosomal location of Rfl, a restorer gene for cytoplasmic pollen sterile maize. Genetics 46:1245–1252
Finn RD, Clements J, Eddy SR (2011) HMMER web server: interactive sequence similarity searching. Nucleic Acids Res 39(Web Server issue):W29–W37
Fisk DG, Walker MB, Barkan A (1999) Molecular cloning of the maize gene crp 1 reveals similarity between regulators of mitochondrial and chloroplast gene expression. EMBO J 18(9):2621–2630
Fujii S, Sato N, Shikanai T (2013) Mutagenesis of individual pentatricopeptide repeat motifs affects RNA binding activity and reveals functional partitioning of Arabidopsis PROTON gradient regulation 3. Plant Cell 25(8):3079–3088
Geddy R, Brown GG (2007) Genes encoding pentatricopeptide repeat (PPR) proteins are not conserved in location in plant genomes and may be subject to diversifying selection. BMC Genomics 8:130
Griffiths-Jones S, Grocock RJ, van Dongen S, Bateman A, Enright AJ (2006) miRBase: micro RNA sequences, targets and gene nomenclature. Nucleic Acids Res 34(Database issue):D140–D144
Hammani K, des Francs-Small CC, Takenaka M, Tanz SK, Okuda K, Shikanai T, Brennicke A, Small I (2011a) The pentatricopeptide repeat protein OTP87 is essential for RNA editing of nad 7 and atp 1 transcripts in Arabidopsis mitochondria. J Biol Chem 286(24):21361–21371
Hammani K, Gobert A, Hleibieh K, Choulier L, Small I, Giege P (2011b) An Arabidopsis dual-localized pentatricopeptide repeat protein interacts with nuclear proteins involved in gene expression regulation. Plant Cell 23(2):730–740
Hauler A, Jonietz C, Stoll B, Stoll K, Braun HP, Binder S (2013) RNA processing factor 5 is required for efficient 5′ cleavage at a processing site conserved in RNAs of three different mitochondrial genes in Arabidopsis thaliana. The Plant journal: for cell and. molecular biology 74(4):593–604
Hobert O (2008) Gene regulation by transcription factors and micro RNAs. Science 319(5871):1785–1786
Holzle A, Jonietz C, Torjek O, Altmann T, Binder S, Forner J (2011) A restorer of fertility-like PPR gene is required for 5′-end processing of the nad 4 mRNA in mitochondria of Arabidopsis thaliana. The Plant journal: for cell and. molecular biology 65(5):737–744
Jiang SC, Mei C, Liang S, Yu YT, Lu K, Wu Z, Wang XF, Zhang DP (2015) Crucial roles of the pentatricopeptide repeat protein SOAR1 in Arabidopsis response to drought, salt and cold stresses. Plant Mol Biol 88(4–5):369–385
Jimenez-Lopez JC, Gachomo EW, Seufferheld MJ, Kotchoni SO (2010) The maize ALDH protein superfamily: linking structural features to functional specificities. BMC Struct Biol 10:43
Jonietz C, Forner J, Hildebrandt T, Binder S (2011) RNA processing factor3 is crucial for the accumulation of mature ccm C transcripts in mitochondria of Arabidopsis accession Columbia. Plant Physiol 157(3):1430–1439
Jonietz C, Forner J, Holzle A, Thuss S, Binder S (2010) RNA processing factor2 is required for 5′ end processing of nad 9 and cox 3 mRNAs in mitochondria of Arabidopsis thaliana. Plant Cell 22(2):443–453
Kakumanu A, Ambavaram MM, Klumas C, Krishnan A, Batlang U, Myers E, Grene R, Pereira A (2012) Effects of drought on gene expression in maize reproductive and leaf meristem tissue revealed by RNA-seq. Plant Physiol 160(2):846–867
Kong YQM, Elling AA, Chen BB, Deng XW (2010) Differential expression of micro RNAs in maize inbred and hybrid lines during salt and drought stress. Am J Plant Sci 1:69–76
Koprivova A, des Francs-Small CC, Calder G, Mugford ST, Tanz S, Lee BR, Zechmann B, Small I, Kopriva S (2010) Identification of a pentatricopeptide repeat protein implicated in splicing of intron 1 of mitochondrial nad 7 transcripts. J Biol Chem 285(42):32192–32199
Kotera E, Tasaka M, Shikanai T (2005) A pentatricopeptide repeat protein is essential for RNA editing in chloroplasts. Nature 433(7023):326–330
Langewisch T (2012) Localization of the Rf3 restorer-of-fertility gene for maize S-type cytoplasmic male sterility.
Laughnan JR, Gabay-Laughnan S (1983) Cytoplasmic male sterility in maize. Annu Rev Genet 17:27–48
Librado P, Rozas J (2009) DnaSP v5: a software for comprehensive analysis of DNA polymorphism data. Bioinformatics 25(11):1451–1452
Lurin C, Andres C, Aubourg S, Bellaoui M, Bitton F, Bruyere C, Caboche M, Debast C, Gualberto J, Hoffmann B, Lecharny A, Le Ret M, Martin-Magniette ML, Mireau H, Peeters N, Renou JP, Szurek B, Taconnat L, Small I (2004) Genome-wide analysis of Arabidopsis pentatricopeptide repeat proteins reveals their essential role in organelle biogenesis. Plant Cell 16(8):2089–2103
Lv HX, Huang C, Guo GQ, Yang ZN (2014) Roles of the nuclear-encoded chloroplast SMR domain-containing PPR protein SVR7 in photosynthesis and oxidative stress tolerance in Arabidopsis. J Plant Biol 57(5):291–301
Manthey GM, McEwen JE (1995) The product of the nuclear gene PET309 is required for translation of mature mRNA and stability or production of intron-containing RNAs derived from the mitochondrial COX1 locus of Saccharomyces cerevisiae. EMBO J 14(16):4031–4043
Meierhoff K, Felder S, Nakamura T, Bechtold N, Schuster G (2003) HCF152, an Arabidopsis RNA binding pentatricopeptide repeat protein involved in the processing of chloroplast psb B-psb T-psb H-pet B-pet D RNAs. Plant Cell 15(6):1480–1495
Mootha VK, Lepage P, Miller K, Bunkenborg J, Reich M, Hjerrild M, Delmonte T, Villeneuve A, Sladek R, Xu F, Mitchell GA, Morin C, Mann M, Hudson TJ, Robinson B, Rioux JD, Lander ES (2003) Identification of a gene causing human cytochrome c oxidase deficiency by integrative genomics. Proc Natl Acad Sci U S A 100(2):605–610
Okuda K, Hammani K, Tanz SK, Peng L, Fukao Y, Myouga F, Motohashi R, Shinozaki K, Small I, Shikanai T (2010) The pentatricopeptide repeat protein OTP82 is required for RNA editing of plastid ndh B and ndh G transcripts. The Plant journal: for cell and. molecular biology 61(2):339–349
Pfalz J, Liere K, Kandlbinder A, Dietz KJ, Oelmuller R (2006) pTAC2, -6, and -12 are components of the transcriptionally active plastid chromosome that are required for plastid gene expression. Plant Cell 18(1):176–197
Saha D, Prasad AM, Srinivasan R (2007) Pentatricopeptide repeat proteins and their emerging roles in plants. Plant physiology and biochemistry: PPB/Societe francaise de physiologie vegetale 45(8):521–534
Skibbe DS, Liu F, Wen TJ, Yandeau MD, Cui X, Cao J, Simmons CR, Schnable PS (2002) Characterization of the aldehyde dehydrogenase gene families of Zea mays and Arabidopsis. Plant Mol Biol 48(5–6):751–764
Small ID, Peeters N (2000) The PPR motif—a TPR-related motif prevalent in plant organellar proteins. Trends Biochem Sci 25(2):46–47
Soderlund C, Bomhoff M, Nelson WM (2011) SyMAP v3.4: a turnkey synteny system with application to plant genomes. Nucleic Acids Res 39(10):e68
Takenaka M (2010) MEF9, an E-subclass pentatricopeptide repeat protein, is required for an RNA editing event in the nad 7 transcript in mitochondria of Arabidopsis. Plant Physiol 152(2):939–947
Takenaka M, Verbitskiy D, Zehrmann A, Brennicke A (2010) Reverse genetic screening identifies five E-class PPR proteins involved in RNA editing in mitochondria of Arabidopsis thaliana. J Biol Chem 285(35):27122–27129
Tan J, Tan Z, Wu F, Sheng P, Heng Y, Wang X, Ren Y, Wang J, Guo X, Zhang X, Cheng Z, Jiang L, Liu X, Wang H, Wan J (2014) A novel chloroplast-localized pentatricopeptide repeat protein involved in splicing affects chloroplast development and abiotic stress response in rice. Mol Plant 7(8):1329–1349
Toda T, Fujii S, Noguchi K, Kazama T, Toriyama K (2012) Rice MPR25 encodes a pentatricopeptide repeat protein and is essential for RNA editing of nad 5 transcripts in mitochondria. The Plant journal: for cell and. molecular biology 72(3):450–460
Uyttewaal M, Mireau H, Rurek M, Hammani K, Arnal N, Quadrado M, Giege P (2008) PPR336 is associated with polysomes in plant mitochondria. J Mol Biol 375(3):626–636
Verbitskiy D, Zehrmann A, Hartel B, Brennicke A, Takenaka M (2012) Two related RNA-editing proteins target the same sites in mitochondria of Arabidopsis thaliana. J Biol Chem 287(45):38064–38072
Wang YG, An M, Zhou SF, She YH, Li WC, Fu FL (2014) Expression profile of maize micro RNAs corresponding to their target genes under drought stress. Biochem Genet 52(11–12):474–493
Wang Z, Zou Y, Li X, Zhang Q, Chen L, Wu H, Su D, Chen Y, Guo J, Luo D, Long Y, Zhong Y, Liu YG (2006) Cytoplasmic male sterility of rice with boro II cytoplasm is caused by a cytotoxic peptide and is restored by two related PPR motif genes via distinct modes of mRNA silencing. Plant Cell 18(3):676–687
Wei K, Pan S (2014) Maize protein phosphatase gene family: identification and molecular characterization. BMC Genomics 15:773
Wei KF, Chen J, Chen YF, Wu LJ, Xie DX (2012) Molecular phylogenetic and expression analysis of the complete WRKY transcription factor family in maize. DNA research: an international journal for rapid publication of reports on genes and genomes 19(2):153–164
Wei K, Chen Y, Xie D (2013) Genome-scale evolution and phylodynamics of H5N1 influenza virus in China during 1996-2012. Vet Microbiol 167(3–4):383–393
Wei K, Wang Y, Zhong X, Pan S (2014) Protein kinase structure, expression and regulation in maize drought signaling. Mol Breed 34(2):583–602
Wen LY, Chase CD (1999) Mitochondrial gene expression in developing male gametophytes of male-fertile and S male-sterile maize. Sex Plant Reprod 11(6):323–330
Wise RP, Fliss AE, Pring DR, Gengenbach BG (1987) Urf 13-T of T cytoplasm maize mitochondria encodes a 13 kD polypeptide. Plant Mol Biol 9(2):121–126
Xie F, Zhang B (2010) Target-align: a tool for plant micro RNA target identification. Bioinformatics 26(23):3002–3003
Xu XH, Chen H, Sang YL, Wang F, Ma JP, Gao XQ, Zhang XS (2012) Identification of genes specifically or preferentially expressed in maize silk reveals similarity and diversity in transcript abundance of different dry stigmas. BMC Genomics 13:294
Yang Z, Gu S, Wang X, Li W, Tang Z, Xu C (2008) Molecular evolution of the CPP-like gene family in plants: insights from comparative genomics of Arabidopsis and rice. J Mol Evol 67(3):266–277
Yin P, Li Q, Yan C, Liu Y, Liu J, Yu F, Wang Z, Long J, He J, Wang HW, Wang J, Zhu JK, Shi Y, Yan N (2013) Structural basis for the modular recognition of single-stranded RNA by PPR proteins. Nature 504(7478):168–171
Yu QB, Jiang Y, Chong K, Yang ZN (2009) AtECB2, a pentatricopeptide repeat protein, is required for chloroplast transcript acc D RNA editing and early chloroplast biogenesis in Arabidopsis thaliana. The Plant journal: for cell and. molecular biology 59(6):1011–1023
Yuan H, Liu D (2012) Functional disruption of the pentatricopeptide protein SLG1 affects mitochondrial RNA editing, plant development, and responses to abiotic stresses in Arabidopsis. The Plant journal: for cell and. molecular biology 70(3):432–444
Zabala G, Gabay-Laughnan S, Laughnan JR (1997) The nuclear gene Rf3 affects the expression of the mitochondrial chimeric sequence R implicated in S-type male sterility in maize. Genetics 147(2):847–860
Zehrmann A, van der Merwe J, Verbitskiy D, Hartel B, Brennicke A, Takenaka M (2012) The DYW-class PPR protein MEF7 is required for RNA editing at four sites in mitochondria of Arabidopsis thaliana. RNA Biol 9(2):155–161
Zehrmann A, Verbitskiy D, van der Merwe JA, Brennicke A, Takenaka M (2009) A DYW domain-containing pentatricopeptide repeat protein is required for RNA editing at multiple sites in mitochondria of Arabidopsis thaliana. Plant Cell 21(2):558–567
Zhang M, Kong X, Xu X, Li C, Tian H, Ding Z (2015) Comparative transcriptome profiling of the maize primary, crown and seminal root in response to salinity stress. PLoS One 10(3):e0121222
Zhu Q, Dugardeyn J, Zhang C, Muhlenbock P, Eastmond PJ, Valcke R, De Coninck B, Oden S, Karampelias M, Cammue BP, Prinsen E, Van Der Straeten D (2014) The Arabidopsis thaliana RNA editing factor SLO2, which affects the mitochondrial electron transport chain, participates in multiple stress and hormone responses. Mol Plant 7(2):290–310
Zoschke R, Kroeger T, Belcher S, Schottler MA, Barkan A, Schmitz-Linneweber C (2012) The pentatricopeptide repeat-SMR protein ATP4 promotes translation of the chloroplast atp B/E mRNA. The Plant journal: for cell and molecular biology 72(4):547–558
Acknowledgments
We are grateful to the providers who submitted microarray and RNA-seq data to the public expression databases, which can be freely applied.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
The project was supported by the Science and Technology Cooperation Project of Fujian Province, China (Grant No. 2015I0006)
Additional information
These authors have contributed equally to this work
Electronic supplementary material
Fig. S1
Sequence alignment of eight repeats of ZmPPR479. The secondary structure of a typical PPR motif is shown above the alignment, with green and blue bars representing helices. The sequence logo was generated using eight repeats of ZmPPR479 sequence. (PDF 404 kb)
Fig. S2
The map of exon/intron arrangement of ZmPPRs. (PDF 523 kb)
Fig. S3
Circos diagram of PPR gene pairs between maize and rice genomes. Outer two circles distribution of each of the PPR genes and scaled chromosomes for each species in megabase (Mb) units, respectively. Line plot below each chromosome Number of introns of PPR genes: green <5 introns, red ≥10 introns. Boxes represent the syntenic blocks. Colors are assigned to the syntenic regions according to the colors of the corresponding chromosomes. Innermost colored lines interconnect putative orthologous PPR gene pairs between the genomes. (PDF 273 kb)
Fig. S4
Genomic distribution of 520 ZmPPR genes on ten maize chromosomes. Different classes of ZmPPR genes have been marked by different colors. The tandem duplicated gene clusters are joined with vertical lines, and duplication genes are connected by dashed lines in green. (PDF 430 kb)
Fig. S5
Hierarchial clustering display of 471 ZmPPR transcripts detected in 60 distinct tissues at different stages in eleven organs in maize. (PDF 481 kb)
Fig. S6
Differentially expressed analysis of ZmPPRs and organelle genes along maize leaf developmental gradients. E2 enzyme was used as a internal control. Chloroplast and mitochondrial genes were highlighted with green and orange colors respectively. (PDF 538 kb)
Fig. S7
Hierarchical clustering on pairwise correlation coefficients of ZmPPRs and intracellular organelles genes [chloroplast (above) and mitochondria (below)]. (PDF 147 kb)
Fig. S8
The expression profiles of nucleus genes (ZmPPR and ZmRf genes) and intracellular organelle genes across five tissues [immature silks (IMS), mature ovary (MO), mature pollen (MP), mature silk (MS) and seedling (SL)]. (PDF 452 kb)
Fig. S9
Normalized microarray signal intensities of 64 probe sets representing 64 ZmPPRs expressed in U. maydis infected seedling leaves. (PDF 401 kb)
Fig. S10
Expression profiles of 450 ZmPPRs in three root types [primary root (PR), seminal roots (SR) and crown roots (CR)] under salt stress. Log2 based fold changes were used to create the heatmap. White box means that gene only expressed in control or was not expressed both in control and salt stress treatment, while gray box indicates that gene specifically expressed under salt treatment. (PDF 67 kb)
Table S1
The identified maize PPR proteins and their related information. 1 Isoelectric Point of ZmPPRs. 2 Molecular Weight of ZmPPRs. 3 Probability of export to mitochondria and chloroplast. 4 Predicted subcellular localization of ZmPPRs. “C”, “M”, “S”, and “_” are represent as “chloroplast”, “mitochondrion”, “secretory pathway” and “any other location”, respectively. (PDF 245 kb)
Table S2
The Ka/Ks ratios and estimate of the absolute dates for the duplication events between the duplicated ZmPPR genes. (PDF 66 kb)
Table S3
List of 471 detected PPR genes and their expression values in sixty distinct tissues representing eleven major organ systems of inbred line B73. (PDF 420 kb)
Table S4
The expression patterns of ZmPPRs and organelle genes at successive stages in leaf development. (PDF 189 kb)
Table S5
A list of ZmPPRs and their corresponding orthologous genes in Arabidopsis or rice. (PDF 153 kb)
Table S6
The probe sets of ZmPPRs on the maize 18 k GeneChip in this study. (PDF 76 kb)
Table S7
RPKM values of ZmPPRs under salt stress in crown root (CR), primary root (PR), and seminal root (SR). (PDF 130 kb)
Table S8
List of primers used in this study. (PDF 56 kb)
Table S9
List of FPKM values of ZmPPRs in maize reproductive (cob) and vegetative tissue (leaf) under both drought and well-watered conditions. MCC and MCD stand for maize ovary tissue, well watered and drought, respectively; MLC and MLD stand for maize basal leaf meristem, well watered and drought, respectively. Numbers 1 and 2 indicate the two biological replicates. The extent of differential expression is measured in terms of fold change and (−) indicates failure to calculate or undetected values. Values highlighted in red and blue stand for the fold increase and decrease in the drought-stressed tissue, respectively. (PDF 147 kb)
Table S10
Cis-acting elements were predicted from 1.5 kb upstream promoter region of ZmPPRs. (PDF 94 kb)
Table S11
List of predicted miRNA-regulated ZmPPRs. (PDF 153 kb)
Rights and permissions
About this article
Cite this article
Wei, K., Han, P. Pentatricopeptide repeat proteins in maize. Mol Breeding 36, 170 (2016). https://doi.org/10.1007/s11032-016-0596-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11032-016-0596-2