Abstract
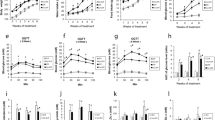
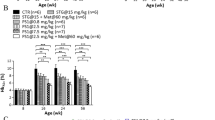
Type 2 Diabetes mellitus (T2DM) is characterized by peripheral insulin resistance, impaired insulin secretion, and reduced β-cell mass. Mechanisms that underlie β-cell failure include glucotoxicity, lipotoxicity, endoplasmic reticulum (ER) stress, and oxidative stress. This study was designed to assess the protective effect of trigonelline and diosgenin against changes in ER stress-associated apoptotic proteins CHOP, Caspase12, and Caspase3 and antioxidant levels in pancreas as well as adipose tissue PPARγ mRNA in T2DM rats. Markers of diabetes and obesity such as serum glucose, insulin, free fatty acid (FFA), TNF-α, IL-6, and leptin were also assessed. T2DM rats showed significantly elevated levels of pancreatic ER stress proteins and lipid peroxidation, while the antioxidants were significantly reduced. Histological examination also confirmed T2DM-associated damage in pancreas. In addition, a significant increase in serum FFA, TNF-α, IL-6, and decrease in leptin levels along with significantly decreased adipose mass and reduced PPARγ expression were observed in T2DM rats. On the other hand, trigonelline and diosgenin treatment independently brought about significant improvement in serum parameters, decrease in apoptotic ER stress proteins, and reinforced antioxidant status in pancreas. Histological examination of pancreas showed normal morphology. Treated groups also showed increased adipose tissue mass and enhanced PPARγ expression. Data from docking studies indicated good interaction of both compounds with PPARγ, and diosgenin showed better binding efficiency. These findings suggest that the insulin-sensitizing effects of trigonelline and diosgenin are mediated through moderation of ER stress and oxidative stress in pancreas as well as by PPARγ activation in adipose tissue.





Similar content being viewed by others
Abbreviations
- CHOP:
-
CCAAT/enhancer-binding protein (C/EBP) homologous protein (CHOP)
- CPCSEA:
-
Control and supervision of Experiments on Animals
- eIF2a:
-
Eukaryotic initiation factor 2a
- ELISA:
-
Enzyme-linked immunosorbent assay
- ER:
-
Endoplasmic reticulum
- ERO1:
-
Endoplasmic reticulum oxidoreductin 1
- FFA:
-
Free fatty acid
- GADD34:
-
Growth arrest and DNA damage-inducible protein 34
- GOD–PAP:
-
Glucose oxidase–phenol + aminophenazone
- GPx:
-
Glutathione peroxidase
- GSH:
-
Reduced glutathione
- H&E:
-
Haematoxylin and eosin
- HFD:
-
High-fat diet
- HOMA-B:
-
Homeostasis model assessment of β‐cell function
- HOMA-IR:
-
Homeostasis model assessment of insulin resistance
- IL-6:
-
Interleukin-6
- LPO:
-
Lipid peroxidation
- NBF:
-
Neutral buffered formaline
- NPD:
-
Normal pelleted diet
- PDI:
-
Protein disulfide isomerase
- PPARγ:
-
Peroxisome proliferated-activated receptor γ
- PUFA:
-
Polyunsaturated fatty acid
- QUICKI:
-
Quantitative insulin sensitivity check index
- ROS:
-
Reactive oxygen species
- SDS-PAGE:
-
Sodium dodecyl sulfate polyacrylamide gel electrophoresis
- SOD:
-
Superoxide dismutase
- STZ:
-
Streptozotocin
- T2DM:
-
Type 2 diabetes mellitus
- TBS:
-
Tris buffered saline
- TMB/H2O2 :
-
3,3′,5,5′-Tetramethylbenzidine/hydrogen peroxide
- TNF-α:
-
Tumor necrosis factor-α
- UPR:
-
Unfolded protein response
References
Hossain P, Kawar B, El Nahas M (2007) Obesity and diabetes in the developing world-a growing challenge. N Engl J Med 356:213–215
Khan SE, Hull RL, Utzschneider KM (2006) Mechanism linking obesity to insulin resistance and type 2 diabetes. Nature 444:840–846
Robertson RP (2004) Chronic oxidative stress as a central mechanism for glucose toxicity in pancreatic islet beta cells in diabetes. J Biol Chem 279:42351–42354
Ozcan U, Cao Q, Yilmaz E, Lee AH, Iwakoshi NN, Ozdelen E, Tuncman G, Görgün C, Glimcher LH, Hotamisligil GS (2004) Endoplasmic reticulum stress links obesity, insulin action, and type 2 diabetes. Science 306:457–461
Wang H, Kouri G, Wollheim CB (2005) ER stress and SREBP-1 activation are implicated in beta-cell glucolipotoxicity. J Cell Sci 118:3905–3915
Nakatani Y, Kaneto H, Kawamori D, Yoshiuchi K, Hatazaki M, Matsuoka TA, Ozawa K, Ogawa S, Hori M, Yamasaki Y, Matsuhisa M (2005) Involvement of ER stress in insulin resistance and diabetes. J Biol Chem 280:847–851
Warne JP (2003) Tumour necrosis factor alpha: a key regulator of adipose tissue mass. J Endocrinol 177:351–355
Margoni A, Perrea DN, Vlachos I, Prokopaki G, Pantopoulou A (2011) Serum leptin, adiponectin and tumor necrosis factor-α in hyperlipidemic rat with/without concomitant diabetes mellitus. Mol Med 17(1–2):36–40
Bays H, Mandarino L, Defronzo R (2004) Mechanisms of endocrine disease. Role of adipocyte, free fatty acids and ectopic fat in pathogenesis of type II diabetes mellitus: peroxisomal proliferator—activated receptor agonists provide a rational approach. The. J Clin Endocrinol Metab 89(2):463–478
Autuna-Puente B, Feve B, Fellahi S, Bastard JP (2008) Adipokines: the missing link between insulin resistance and obesity. Diabetes Metab 34:2–11
Bensinger SJ, Tontonoz P (2008) Integration of metabolism and inflammation by lipid-activated nuclear receptors. Nature 454:470–477
Kintscher U, Law RE (2005) PPAR gamma- mediated insulin sensitization: the importance of fat versus muscle. Am J Physiol Endocrinol Metab 288:E287–E291
Yoshinari O, Igarashi K (2010) Anti-diabetic effect of trigonelline and nicotinic acid, on KK-A(y) mice. Curr Med Chem 17(20):2196–2202
Zhou J, Zhou S, Zeng S (2011) Experimental diabetes treated with trigonelline: effect on beta cell and pancreatic oxidative parameters. Fundam Clin Pharmacol. doi:10.1111/j.1472-8206.2011.01022.x
McAnuff MA, Omoruyi FO, Harding WW, Jacobs H, Morrison EY, Asemota HN (2005) Hypoglycemic effects of steroidal sapogenins isolated from Jamaican bitter yam, (Dioscorea polygonoides). Food Chem Toxicol 43:1667–1672
Patel Kanika, Gadewar Manoj, Tahilyani Vijay, Patel Dinesh Kumar (2012) A review on pharmacological and analytical aspects of diosgenin: a concise report. Nat Prod Bioprospcet 2:46–52
Kim J, Kwak HJ, Cha JY, Jeong YS, Rhee SD, Cheon HG (2014) The role of prolyl hydroxylase domain protein (PHD) during rosiglitazone-induced adipocyte differentiation. J Biol Chem 289:2755–2764
Srinivasan K, Viswanad B, Asrat L, Kaul CL, Ramarao P (2005) Combination of high-fat diet and low dose streptozotocin-treated rat: a model for type II diabetes and pharmacological screening. Pharmacol Res 52:313–320
Falholt K, Lund B, Falholt W (1973) An easy colorimetric method for routine determination of free fatty acids in plasma. Clin Chim Act 46(2):105–111
Matthews DR, Hosker JP, Rudenski AS, Naylor BA, Treacher DF, Turner RC (1985) Homeostasis model assessment: insulin resistance and beta cell function from fasting plasma glucose and insulin concentrations in man. Diabetologia 28:412–419
Ohkawa H, Ohishi N, Yagi K (1979) Assay for lipid peroxides in animal tissues by thiobarbituric acid reaction. Anal Biochem 95:351–358
Ellman GL (1973) Tissue sulfhydryl groups. Arch Biochem Biophys 179:588–590
Marklund S, Marklund G (1974) Involvement of the superoxide anion radical in the autoxidation of pyrogallol and a convenient assay for superoxide dismutase. Eur J Biochem 47:69–74
Claiborne A (1985) Handbook of methods for oxygen radical research. CRC Press, Boca Raton, pp 283–284
Rotruck JT, Pope AL, Ganther HE, Swanson AB, Hateman DG, Hoekstra WG (1973) Selenium: biochemical role as a component of glutathione peroxidase. Science 179:588–590
Luna LG (1968) Manual of histological staining methods of the Armed Forces Institute of Pathology. McGraw-Hill, New York
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2−∆∆Ct method. Methods 25:402–408
Xu HE, Lambert MH, Montana VG, Plunket KD, Moore LB, Collins JL, Oplinger JA, Kliewer SA, Gampe RT Jr, McKee DD, Moore JT, Willson TM (2001) Structural determinants of ligand binding selectivity between the peroxisome proliferator-activated receptors. Proc Natl Acad Sci USA 98(24):13919–13924
Friesner RA, Banks JL, Murphy RB, Halgren TA, Klicic JJ, Mainz DT, Repasky MP, Knoll EH, Shelley M, Perry JK, Shaw DE, Francis P, Shenkin PS (2004) Glide: a new approach for rapid, accurate docking and scoring. 1. Method and assessment of docking accuracy. J Med Chem 47(7):1739–1749
Friesner RA, Murphy RB, Repasky MP, Frye LL, Greenwood JR, Halgren TA, Sanschagrin PC, Mainz DT (2006) Extra precision glide: docking and scoring incorporating a model of hydrophobic enclosure for protein–ligand complexes. J Med Chem 49(21):6177–6196
Søndergaard CR, Olsson MHM, Rostkowski M, Jensen JH (2011) Improved treatment of ligands and coupling effects in empirical calculation and rationalization of pKa values. J Chem Theory Comput 7(7):2284–2295
Liu YQ, Jetton TL, Leahy JL (2002) Β-cell adaptation to insulin resistance. Increased pyruvate carboxylate and malate-pyruvate shuttle activity in islets of nondiabetic Zucker fatty rats. J Biol Chem 277:39163–39168
Hotamisligil GS (2010) Endoplasmic reticulum stress and the inflammatory basis of metabolic disease. Cell 140:900–917
Mother KJ, Hunt AE, Steinberg HO, Paradisi G, Hook G, Katz A, Quon MJ, Baron AD (2001) Repeatability characteristics of simple indices of insulin resistance: implication for research application. J Clin Endocrinol Metab 86:5457–5464
Chen H, Sullivan G, Quon MJ (2005) Assessing the predictive accuracy of QUICKI as a surrogate index for insulin sensitivity using a calibration model. Diabetes 54:1914–1925
Qatanani M, Lazar MA (2007) Mechanisms of obesity-associated insulin resistance: many choices on the menu. Genes Dev 21:1443–1455
Uysal KT, Wiesbrock SM, Marino MW, Hotamisligil GS (1997) Protection from obesity-induced insulin resistance in mice lacking TNF-α function. Nature 389:610–614
Wauters M, Considine RV, Yudkin JS, Peiffer F, De Leeuw I, Van Gaal LF (2003) Leptin levels in Type 2 diabetes: association with measures of insulin resistance and insulin secretion. Horm Metab Res 35(2):92–96
Havel PJ (2002) Control of energy homeostasis and insulin action by adipocyte hormones: leptin, acylation stimulating protein and adiponectin. Curr Opin Lipidol 13:51–59
Schoonjans K, Staels B, Auwerx J (1996) The peroxisome proliferator-activated receptors (PPARs) and their effects on lipid metabolism and adipocyte differentiation. Biochim Biophys Acta 1302:93–109
Desvergne B, Wahli W (1999) Peroxisome proliferator-activated receptors: nuclear control of metabolism. Endocr Rev 20:649–688
Braissant O, Foufellie F, Scotto C, Dauca M, Wahli W (1996) Differential expression of Peroxisome proliferator-activated receptors (PPARs): tissue distribution of PPAR-a, -b and -g in the adult rat. Endocrinology 137:354–366
Aoyama T, Peters JM, Iritani N, Nakajima T, Furihata K, Hashimoto T, Gonzalez FJ (1998) Altered constitutive expression of fatty acid metabolizing enzymes in mice lacking the peroxisome proliferator-activated receptor-a (PPAR-a). J Biol Chem 273:5678–5684
Jain S, Pulikuri S, Zhu Y, Qi C, Kanwar YS, Yeldandi AV, Rao MS, Reddy JK (1998) Differential expression of the peroxisome proliferator-activated receptor g (PPAR g) and its coactivators steroid receptor coactivator-1 and PPAR-binding protein PBP in the brown fat, urinary bladder, colon and breast of the mouse. Am J Pathol 153:349–354
Rosen ED, Sarraf P, Troy AE, Bradwin G, Moore K, Milstone DS, Spiegelman BM, Mortensen RM (1999) PPARγ is required for the differentiation of adipose tissue in vivo and in vitro. Mol Cell 4:611–617
Barroso I, Gurnell M, Crowley VE, Agostini M, Schwabe JW, Soos MA, Maslen GL, Williams TD, Lewis H, Schafer AJ, Chatterjee VK, O’Rahilly S (1999) Dominant negative mutations in human PPARgamma associated with severe insulin resistance, diabetes mellitus and hypertension. Nature 402:880–883
Berthiaume M, Sell H, Lalonde J, Gélinas Y, Tchernof A, Richard D, Deshaies Y (2004) Actions of PPARgamma agonism on adipose tissue remodeling, insulin sensitivity, and lipemia in absence of glucocorticoids. Am J Physiol Regul Integr Comp Physiol 287(5):R1116–R1123
He W, Barak Y, Hevener A, Olson P, Liao D, Le J, Nelson M, Ong E, Olefsky JM, Evans RM (2003) Adipose-specific peroxisome proliferator-activated receptor gamma knockout causes insulin resistance in fat and liver but not in muscle. Proc Natl Acad Sci USA 100(26):15712–15717
Yki-Jarvinen H (2004) Thiazolidinediones. N Engl J Med 351(11):1106–1118
Kozutsumi Y, Segal M, Normington K, Gething M-J, Sambrook J (1988) The presence of malfolded proteins in the endoplasmic reticulum signals the induction of glucose-regulated proteins. Nature 332:462–464
Bernales S, McDonald KL, Walter P (2006) Autophagy counterbalances endoplasmic reticulum expansion during the unfolded protein response. PLoS Biol 4:e423
Rao RV, Ellerby HM, Bredesen DE (2004) Coupling endoplasmic reticulum stress to the cell death program. Cell Death Differ 11:372–380
Eizirik DL, Cardozo AK, Cnop M (2008) The role for endoplasmic reticulum stress in diabetes mellitus. Endocr Rev 29(1):42–61
Ohoka N, Yoshii S, Hattori T, Onozaki K, Hayashi H (2005) TRB3, a novel ER stress-inducible gene, is induced via ATF4-CHOP pathway and is involved in cell death. EMBO J 24:1243–1255
Nagy G, Kardon T, Wunderlich L, Szarka A, Kiss A, Schaff Z, Bánhegyi G, Mandl J (2007) Acetaminophen induces ER dependent signaling in mouse liver. Arch Biochem Biophys 459:273–279
Harding HP, Zeng H, Zhang Y, Jungries R, Chung P, Plesken H, Sabatini DD, Ron D (2001) Diabetes mellitus and exocrine pancreatic dysfunction in perk −/− mice reveals a role for translational control in secretory cell survival. Mol Cell 7:1153–1163
Sanges D, Marigo V (2006) Cross-talk between two apoptotic pathways activated by endoplasmic reticulum stress: differential contribution of caspase-12 and AIF. Apoptosis 11:1629–1641
Nakagawa T, Zhu H, Morishima N, Li E, Xu J, Yankner BA, Yuan J (2000) Caspase-12 mediates endoplasmic-reticulum-specific apoptosis and cytotoxicity by amyloid-beta. Nature 403:98–103
Cullinan SB, Diehl JA (2006) Coordination of ER and oxidative stress signaling: the PERK/Nrf2 signaling pathway. Int J Biochem Cell Biol 38:317–332
Papa FR (2012) Endoplasmic reticulum stress, pancreatic β-cell degeneration and diabetes. Cold Spring Harb Perspect Med 2(9):a007666
Vattem KM, Wek RC (2004) Reinitiation involving upstream ORFs regulates ATF4 mRNA translation in mammalian cells. Proc Natl Acad Sci USA 101:11269–11274
Garg MC, Ojha S, Bansal DD (1996) Antioxidant status of streptozotocin diabetic rats. Indian J Exp Biol 34:264–266
Acknowledgments
The financial support provided by the University Grants Commission, India, [MRP F. No. 36-191/2008(SR)] and the DST-FIST support is gratefully acknowledged.
Conflict of interest
We hereby state that we do not have any conflict of interest in the present work.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Tharaheswari, M., Jayachandra Reddy, N., Kumar, R. et al. Trigonelline and diosgenin attenuate ER stress, oxidative stress-mediated damage in pancreas and enhance adipose tissue PPARγ activity in type 2 diabetic rats. Mol Cell Biochem 396, 161–174 (2014). https://doi.org/10.1007/s11010-014-2152-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11010-014-2152-x