Abstract
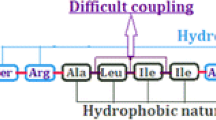
A chemical synthesis of the 37 residue polypeptide human amylin using microwave enhanced solid phase peptide chemistry is described. An optimised protocol used only a single pseudoproline derivative, a chemically pure resin and single couplings of all amino acids to deliver non-oxidised amylin in high yield. Oxidation of the crude peptide to form the disulfide bond was accomplished in 20 min using 2,2′-dipyridyl disulfide in dimethyl sulphoxide giving human amylin that was fully functional in a cAMP assay.






Similar content being viewed by others
References
Abedini A, Raleigh DP (2005) Incorporation of pseudoproline derivatives allows the facile synthesis of human IAPP, a highly amyloidogenic and aggregation-prone polypeptide. Org Lett 7(4):693–696. doi:10.1021/ol047480+
Abedini A, Singh G, Raleigh DP (2006) Recovery and purification of highly aggregation-prone disulfide-containing peptides: application to islet amyloid polypeptide. Anal Biochem 351(2):181–186. doi:10.1016/J.Ab.2005.11.029
Bacsa B, Horvati K, Bosze S, Andreae F, Kappe CO (2008) Solid-phase synthesis of difficult peptide sequences at elevated temperatures: a critical comparison of microwave and conventional heating technologies. J Org Chem 73(19):7532–7542. doi:10.1021/Jo8013897
Bouillon I, Soural M, Miller MJ, Krchnak V (2009) Resins with identical specifications are not identical. Identifying a useful solid-phase resin. J Comb Chem 11(2):213–215. doi:10.1021/Cc800143e
Cao P, Raleigh DP (2010) Ester to amide switch peptides provide a simple method for preparing monomeric islet amyloid polypeptide under physiologically relevant conditions and facilitate investigations of amyloid formation. J Am Chem Soc 132(12):4052–4053. doi:10.1021/Ja910763m
Cline DJ, Thorpe C, Schneider JP (2004) General method for facile intramolecular disulfide formation in synthetic peptides. Anal Biochem 335(1):168–170. doi:10.1016/J.Ab.2004.07.015
Coin I (2010) The depsipeptide method for solid-phase synthesis of difficult peptides. J Pept Sci 16(5):223–230. doi:10.1002/Psc.1224
Collins JM, Leadbeater NE (2007) Microwave energy: a versatile tool for the biosciences. Org Biomol Chem 5(8):1141–1150. doi:10.1039/B617084f
Cooper GJS, Willis AC, Clark A, Turner RC, Sim RB, Reid KBM (1987) Purification and characterization of a peptide from amyloid-rich pancreases of type-2 diabetic-patients. Proc Natl Acad Sci USA 84(23):8628–8632
Cooper GJS, Day AJ, Willis AC, Roberts AN, Reid KBM, Leighton B (1989) Amylin and the amylin gene—structure, function and relationship to islet amyloid and to diabetes–mellitus. Biochim Biophys Acta 1014(3):247–258
Deng FK, Mandal K, Luisier S, Kent SBH (2010) Synthesis and comparative properties of two amide-generating resin linkers for use in solid phase peptide synthesis. J Pept Sci 16(10):545–550. doi:10.1002/Psc.1279
Gingell JJ, Qi T, Bailey RJ, Hay DL (2010) A key role for tryptophan 84 in receptor activity-modifying protein 1 in the amylin 1 receptor. Peptides 31(7):1400–1404. doi:10.1016/j.peptides.2010.03.027
Harris PWR, Brimble MA (2009) Synthesis of an Arginine Tagged [Cys(155)-Arg(180)] Fragment of NY-ESO-1: elimination of an undesired by-product using ‘in house’ resins. Synthesis-Stuttgart 20:3460–3466. doi:10.1055/s-0029-1216952
Harris PWR, Yang SH, Brimble MA (2011) An improved procedure for the preparation of aminomethyl polystyrene resin and its use in solid phase (peptide) synthesis. Tetrahedron Lett 52(45):6024–6026. doi:10.1016/j.tetlet.2011.09.010
Hood CA, Fuentes G, Patel H, Page K, Menakuru M, Park JH (2008) Fast conventional Fmoc solid-phase peptide synthesis with HCTU. J Pept Sci 14(1):97–101. doi:10.1002/Psc.921
Ludvik B, KautzkyWiller A, Prager R, Thomaseth K, Pacini G (1997) Amylin: history and overview. Diabet Med 14(6):S9–S13
Lutz TA (2012) Effects of amylin on eating and adiposity. In: Joost H-G (ed) Appetite control, vol 209. Handbook of experimental pharmacology. Springer–Berlin, Heidelberg, pp 231–250. doi:10.1007/978-3-642-24716-3_10
Marek P, Woys AM, Sutton K, Zanni MT, Raleigh DP (2010) Efficient microwave-assisted synthesis of human islet amyloid polypeptide designed to facilitate the specific incorporation of labeled amino acids. Org Lett 12(21):4848–4851. doi:10.1021/Ol101981b
Maruyama K, Nagasawa H, Suzuki A (1999) 2,2′-Bispyridyl disulfide rapidly induces intramolecular disulfide bonds in peptides. Peptides 20(7):881–884
Muthusamy K, Albericio F, Arvidsson PI, Govender P, Kruger HG, Maguire GEM, Govender T (2010) Microwave assisted SPPS of amylin and its toxicity of the pure product to RIN-5F cells. Biopolymers 94(3):323–330. doi:10.1002/Bip.21370
Mutter M, Nefzi A, Sato T, Sun X, Wahl F, Wohr T (1995) Pseudo–Prolines (Psi–Pro) for accessing inaccessible peptides. Pept Res 8(3):145–153
NovaBiochem (2004) NovaBiochem innovations 1/4 guidelines for the use of pseudoproline dipeptides. http://www.emdmillipore.com/life-science-research/novabiochem-innovations/c_nZGb.s1OBzUAAAEj2WxumA4O. Accessed Aug 2012
Opie EL (1901) The relation of diabetes mellitus to lesions of the pancreas. Hyaline degeneration of the islands of Langerhans. J Exp Med 5(5):527–540
Page K, Hood CA, Patel H, Fuentes G, Menakuru M, Park JH (2007) Fast Fmoc synthesis of hAMYLIN(1–37) with pseudoproline assisted on-resin disulfide formation. J Pept Sci 13(12):833–838
Pedersen SL, Tofteng AP, Malik L, Jensen KJ (2012) Microwave heating in solid-phase peptide synthesis. Chem Soc Rev 41(5):1826–1844
Reda TK, Geliebter A, Pi-Sunyer FX (2002) Amylin, food intake, and obesity. Obes Res 10(10):1087–1091
Roth JD, Roland BL, Cole RL, Trevaskis JL, Weyer C, Koda JE, Anderson CM, Parkes DG, Baron AD (2008) Leptin responsiveness restored by amylin agonism in diet-induced obesity: evidence from nonclinical and clinical studies. Proc Natl Acad Sci USA 105(20):7257–7262. doi:10.1073/pnas.0706473105
Roth JD, Erickson MR, Chen S, Parkes DG (2012) GLP-1R and amylin agonism in metabolic disease: complementary mechanisms and future opportunities. Br J Pharmacol 166(1):121–136. doi:10.1111/j.1476-5381.2011.01537.x
Smith SR, Aronne LJ, Burns CM, Kesty NC, Halseth AL, Weyer C (2008) Sustained weight loss following 12-month pramlintide treatment as an adjunct to lifestyle intervention in obesity. Diabetes Care 31(9):1816–1823. doi:10.2337/Dc08-0029
Sohma Y, Sasaki M, Hayashi Y, Kimura T, Kiso Y (2004) Novel and efficient synthesis of difficult sequence-containing peptides through O-N intramolecular acyl migration reaction of O-acyl E isopeptides. Chem Commun (Camb) 1:124–125. doi:10.1039/B312129a
Sohma Y, Yoshiya T, Taniguchi A, Kimura T, Hayashi Y, Kiso Y (2007) Development of O-acyl isopeptide method. Biopolymers 88(2):253–262. doi:10.1002/Bip.20683
Tam JP, Wu CR, Liu W, Zhang JW (1991) Disulfide bond formation in peptides by dimethyl-sulfoxide—scope and applications. J Am Chem Soc 113(17):6657–6662
Trevaskis JL, Coffey T, Cole R, Lei C, Wittmer C, Walsh B, Weyer C, Koda J, Baron AD, Parkes DG, Roth JD (2008) Amylin-mediated restoration of leptin responsiveness in diet-induced obesity: magnitude and mechanisms. Endocrinology 149(11):5679–5687. doi:10.1210/En.2008-0770
Weichselbaum A, Stangl E (1901) Zur Kenntnis der feineren Veranderungen des Pankreas bie Diabetes Mellitus. Wein Klin Wochenschr 14:968–972
Westermark P, Wernstedt C, Obrien TD, Hayden DW, Johnson KH (1987) Islet amyloid in type-2 human diabetes–mellitus and adult diabetic cats contains a novel putative polypeptide hormone. Am J Pathol 127(3):414–417
Westermark P, Engstrom U, Johnson KH, Westermark GT, Betsholtz C (1990) Islet amyloid polypeptide—pinpointing amino-acid-residues linked to amyloid fibril formation. Proc Natl Acad Sci USA 87(13):5036–5040
Wohr T, Wahl F, Nefzi A, Rohwedder B, Sato T, Sun XC, Mutter M (1996) Pseudo-prolines as a solubilizing, structure-disrupting protection technique in peptide synthesis. J Am Chem Soc 118(39):9218–9227
Yonemoto IT, Kroon GJA, Dyson HJ, Balch WE, Kelly JW (2008) Amylin proprotein processing generates progressively more amyloidogenic peptides that initially sample the helical state†. Biochemistry 47(37):9900–9910. doi:10.1021/bi800828u
Yoshiya T, Higa A, Abe N, Fukao F, Kuruma T, Toda Y, Sohma Y, Kiso Y (2011) Click peptide concept: O-acyl isopeptide of islet amyloid polypeptide as a nonaggregative precursor molecule. ChemBioChem 12(8):1216–1222. doi:10.1002/cbic.201100025
Young A (2005) Amylin and the integrated control of nutrient influx. In: Andrew Y (ed) Advances in pharmacology, vol 52. Academic Press, New York, pp 67–77. doi:10.1016/s1054-3589(05)52004-0
Younk LM, Mikeladze M, Davis SN (2011) Pramlintide and the treatment of diabetes: a review of the data since its introduction. Expert Opin Pharmacother 12(9):1439–1451. doi:10(1517/14656566),2011,581663
Acknowledgments
The authors wish to thank The Maurice Wilkins Centre for Molecular Biodiscovery for financial support (PWRH).
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Harris, P.W.R., Kowalczyk, R., Hay, D.L. et al. A Single Pseudoproline and Microwave Solid Phase Peptide Synthesis Facilitates an Efficient Synthesis of Human Amylin 1–37. Int J Pept Res Ther 19, 147–155 (2013). https://doi.org/10.1007/s10989-012-9325-9
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10989-012-9325-9