Abstract
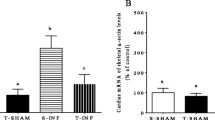
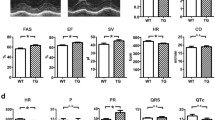
Cardiac hypertrophy (CH) is an adaptive process that exists in two distinct forms and allows the heart to adequately respond to an organism’s needs. The first form of CH is physiological, adaptive and reversible. The second is pathological, irreversible and associated with fibrosis and cardiomyocyte death. CH involves multiple molecular mechanisms that are still not completely defined but it is now accepted that physiological CH is associated more with the PI3-K/Akt pathway while the main signaling cascade activated in pathological CH involves the Calcineurin-NFAT pathway. It was recently demonstrated that the TRPM4 channel may act as a negative regulator of pathological CH by regulating calcium entry and thus the Cn-NFAT pathway. In this study, we examined if the TRPM4 channel is involved in the physiological CH process. We evaluated the effects of 4 weeks endurance training on the hearts of Trpm4 +/+ and Trpm4 −/− mice. We identified an elevated functional expression of the TRPM4 channel in cardiomyocytes after endurance training suggesting a potential role for the channel in physiological CH. We then observed that Trpm4 +/+ mice displayed left ventricular hypertrophy after endurance training associated with enhanced cardiac function. By contrast, Trpm4 −/− mice did not develop these adaptions. While Trpm4 −/− mice did not develop gross cardiac hypertrophy, the cardiomyocyte surface area was larger and associated with an increase of Tunel positive cells. Endurance training in Trpm4 +/+ mice did not increase DNA fragmentation in the heart. Endurance training in Trpm4 +/+ mice was associated with activation of the classical physiological CH Akt pathway while Trpm4 −/− favored the Calcineurin pathway. Calcium studies demonstrated that TRPM4 channel negatively regulates calcium entry providing support for activation of the Cn-NFAT pathway in Trpm4 −/− mice. In conclusion, we provide evidence for the functional expression of TRPM4 channel in response to endurance training. This expression may help to maintain the balance between physiological and pathological hypertrophy.






Similar content being viewed by others
References
Abriel H, Syam N, Sottas V et al (2012) TRPM4 channels in the cardiovascular system: physiology, pathophysiology, and pharmacology. Biochem Pharmacol 84:873–881. doi:10.1016/j.bcp.2012.06.021
Barbet G, Demion M, Moura IC et al (2008) The calcium-activated nonselective cation channel TRPM4 is essential for the migration but not the maturation of dendritic cells. Nat Immunol 9:1148–1156. doi:10.1038/ni.1648
Bernardo BC, Weeks KL, Pretorius L, McMullen JR (2010) Molecular distinction between physiological and pathological cardiac hypertrophy: experimental findings and therapeutic strategies. Pharmacol Ther 128:191–227. doi:10.1016/j.pharmthera.2010.04.005
Bers DM (2008) Calcium cycling and signaling in cardiac myocytes. Annu Rev Physiol 70:23–49. doi:10.1146/annurev.physiol.70.113006.100455
Bush EW, Hood DB, Papst PJ et al (2006) Canonical transient receptor potential channels promote cardiomyocyte hypertrophy through activation of calcineurin signaling. J Biol Chem 281:33487–33496. doi:10.1074/jbc.M605536200
Cho C-H, Kim E, Lee Y-S et al (2014) Depletion of 14-3-3γ reduces the surface expression of transient receptor potential melastatin 4b (TRPM4b) channels and attenuates TRPM4b-mediated glutamate-induced neuronal cell death. Mol Brain 7:52. doi:10.1186/s13041-014-0052-3
Cho GW, Altamirano F, Hill JA (2016) Chronic heart failure: Ca(2+), catabolism, and catastrophic cell death. Biochim Biophys Acta 1862:763–777. doi:10.1016/j.bbadis.2016.01.011
Colquhoun D, Neher E, Reuter H, Stevens CF (1981) Inward current channels activated by intracellular Ca in cultured cardiac cells. Nature 294:752–754
Dai W, Bai Y, Hebda L et al (2014) Calcium deficiency-induced and TRP channel-regulated IGF1R-PI3K-Akt signaling regulates abnormal epithelial cell proliferation. Cell Death Differ 21:568–581. doi:10.1038/cdd.2013.177
Dalal MD, Morgans CW, Duvoisin RM et al (2013) Diagnosis of occult melanoma using transient receptor potential melastatin 1 (TRPM1) autoantibody testing: a novel approach. Ophthalmology 120:2560–2564. doi:10.1016/j.ophtha.2013.07.037
DeBosch B, Treskov I, Lupu TS et al (2006) Akt1 is required for physiological cardiac growth. Circulation 113:2097–2104. doi:10.1161/CIRCULATIONAHA.105.595231
Demion M, Thireau J, Gueffier M et al (2014) Trpm4 gene invalidation leads to cardiac hypertrophy and electrophysiological alterations. PLoS ONE 9:e115256. doi:10.1371/journal.pone.0115256
Diedrichs H, Chi M, Boelck B et al (2004) Increased regulatory activity of the calcineurin/NFAT pathway in human heart failure. Eur J Heart Fail 6:3–9. doi:10.1016/j.ejheart.2003.07.007
Earley S (2013) TRPM4 channels in smooth muscle function. Pflugers Arch 465:1223–1231. doi:10.1007/s00424-013-1250-z
Eghbali M, Deva R, Alioua A et al (2005) Molecular and functional signature of heart hypertrophy during pregnancy. Circ Res 96:1208–1216. doi:10.1161/01.RES.0000170652.71414.16
Grand T, Demion M, Norez C et al (2008) 9-phenanthrol inhibits human TRPM4 but not TRPM5 cationic channels. Br J Pharmacol 153:1697–1705. doi:10.1038/bjp.2008.38
Guinamard R, Rahmati M, Lenfant J, Bois P (2002) Characterization of a Ca2+-activated nonselective cation channel during dedifferentiation of cultured rat ventricular cardiomyocytes. J Membr Biol 188:127–135. doi:10.1007/s00232-001-0180-4
Guinamard R, Demion M, Magaud C et al (2006) Functional expression of the TRPM4 cationic current in ventricular cardiomyocytes from spontaneously hypertensive rats. Hypertension 48:587–594. doi:10.1161/01.HYP.0000237864.65019.a5
Guinamard R, Demion M, Launay P (2010) Physiological roles of the TRPM4 channel extracted from background currents. Physiology 25:155–164. doi:10.1152/physiol.00004.2010
Haq S, Choukroun G, Lim H et al (2001) Differential activation of signal transduction pathways in human hearts with hypertrophy versus advanced heart failure. Circulation 103:670–677
Hof T, Sallé L, Coulbault L et al (2016) TRPM4 non-selective cation channels influence action potentials in rabbit Purkinje fibres. J Physiol 594:295–306. doi:10.1113/JP271347
Ikeda H, Shiojima I, Ozasa Y et al (2009) Interaction of myocardial insulin receptor and IGF receptor signaling in exercise-induced cardiac hypertrophy. J Mol Cell Cardiol 47:664–675. doi:10.1016/j.yjmcc.2009.08.028
Jacobs G, Oosterlinck W, Dresselaers T et al (2015) Enhanced β-adrenergic cardiac reserve in Trpm4−/− mice with ischaemic heart failure. Cardiovasc Res 105:330–339. doi:10.1093/cvr/cvv009
Kecskés M, Jacobs G, Kerselaers S et al (2015) The Ca(2+)-activated cation channel TRPM4 is a negative regulator of angiotensin II-induced cardiac hypertrophy. Basic Res Cardiol 110:43. doi:10.1007/s00395-015-0501-x
Kemi OJ, Ceci M, Wisloff U et al (2008) Activation or inactivation of cardiac Akt/mTOR signaling diverges physiological from pathological hypertrophy. J Cell Physiol 214:316–321. doi:10.1002/jcp.21197
Kim J, Wende AR, Sena S et al (2008) Insulin-like growth factor I receptor signaling is required for exercise-induced cardiac hypertrophy. Mol Endocrinol 22:2531–2543. doi:10.1210/me.2008-0265
Köster OF, Szigeti GP, Beuckelmann DJ (1999) Characterization of a [Ca2+]i-dependent current in human atrial and ventricular cardiomyocytes in the absence of Na+ and K+. Cardiovasc Res 41:175–187
Kuwahara K, Wang Y, McAnally J et al (2006) TRPC6 fulfills a calcineurin signaling circuit during pathologic cardiac remodeling. J Clin Invest 116:3114–3126. doi:10.1172/JCI27702
Launay P, Fleig A, Perraud AL et al (2002) TRPM4 is a Ca2+-activated nonselective cation channel mediating cell membrane depolarization. Cell 109:397–407
Launay P, Cheng H, Srivatsan S et al (2004) TRPM4 regulates calcium oscillations after T cell activation. Science 306:1374–1377. doi:10.1126/science.1098845
Li R, Zheng W, Pi R et al (2007) Activation of peroxisome proliferator-activated receptor-alpha prevents glycogen synthase 3beta phosphorylation and inhibits cardiac hypertrophy. FEBS Lett 581:3311–3316. doi:10.1016/j.febslet.2007.06.017
Li Y, Baylie RL, Tavares MJ, Brayden JE (2014) TRPM4 channels couple purinergic receptor mechanoactivation and myogenic tone development in cerebral parenchymal arterioles. J Cereb Blood Flow Metab 34:1706–1714. doi:10.1038/jcbfm.2014.139
Liu H, El Zein L, Kruse M et al (2010) Gain-of-function mutations in TRPM4 cause autosomal dominant isolated cardiac conduction disease. Circ Cardiovasc Genet 3:374–385. doi:10.1161/CIRCGENETICS.109.930867
Liu H, Chatel S, Simard C et al (2013) Molecular genetics and functional anomalies in a series of 248 Brugada cases with 11 mutations in the TRPM4 channel. PLoS ONE 8:e54131. doi:10.1371/journal.pone.0054131
Makarewich CA, Zhang H, Davis J et al (2014) Transient receptor potential channels contribute to pathological structural and functional remodeling after myocardial infarction. Circ Res 115:567–580. doi:10.1161/CIRCRESAHA.115.303831
Mann N, Rosenzweig A (2012) Can exercise teach us how to treat heart disease? Circulation 126:2625–2635. doi:10.1161/CIRCULATIONAHA.111.060376
Mathar I, Kecskes M, Van der Mieren G et al (2014) Increased β-adrenergic inotropy in ventricular myocardium from Trpm4−/− mice. Circ Res 114:283–294. doi:10.1161/CIRCRESAHA.114.302835
Molkentin JD, Lu JR, Antos CL et al (1998) A calcineurin-dependent transcriptional pathway for cardiac hypertrophy. Cell 93:215–228
Nakayama H, Wilkin BJ, Bodi I, Molkentin JD (2006) Calcineurin-dependent cardiomyopathy is activated by TRPC in the adult mouse heart. FASEB J 20:1660–1670. doi:10.1096/fj.05-5560com
Nilius B, Prenen J, Droogmans G et al (2003) Voltage dependence of the Ca2+-activated cation channel TRPM4. J Biol Chem 278:30813–30820. doi:10.1074/jbc.M305127200
Nilius B, Prenen J, Janssens A et al (2005) The selectivity filter of the cation channel TRPM4. J Biol Chem 280:22899–22906. doi:10.1074/jbc.M501686200
Oliveira RSF, Ferreira JCB, Gomes ERM et al (2009) Cardiac anti-remodelling effect of aerobic training is associated with a reduction in the calcineurin/NFAT signalling pathway in heart failure mice. J Physiol 587:3899–3910. doi:10.1113/jphysiol.2009.173948
Onohara N, Nishida M, Inoue R et al (2006) TRPC3 and TRPC6 are essential for angiotensin II-induced cardiac hypertrophy. EMBO J 25:5305–5316. doi:10.1038/sj.emboj.7601417
Palkar R, Lippoldt EK, McKemy DD (2015) The molecular and cellular basis of thermosensation in mammals. Curr Opin Neurobiol 34:14–19. doi:10.1016/j.conb.2015.01.010
Park J-Y, Hwang EM, Yarishkin O et al (2008) TRPM4b channel suppresses store-operated Ca2+ entry by a novel protein-protein interaction with the TRPC3 channel. Biochem Biophys Res Commun 368:677–683. doi:10.1016/j.bbrc.2008.01.153
Qi J-Y, Xu M, Lu Z-Z, Zhang Y-Y (2010) 14-3-3 inhibits insulin-like growth factor-I-induced proliferation of cardiac fibroblasts via a phosphatidylinositol 3-kinase-dependent pathway. Clin Exp Pharmacol Physiol 37:296–302. doi:10.1111/j.1440-1681.2009.05282.x
Runnels LW (2011) TRPM6 and TRPM7: a Mul-TRP-PLIK-cation of channel functions. Curr Pharm Biotechnol 12:42–53
Runnels LW, Yue L, Clapham DE (2001) TRP-PLIK, a bifunctional protein with kinase and ion channel activities. Science 291:1043–1047. doi:10.1126/science.1058519
Sah R, Mesirca P, Mason X et al (2013a) Timing of myocardial trpm7 deletion during cardiogenesis variably disrupts adult ventricular function, conduction, and repolarization. Circulation 128:101–114. doi:10.1161/CIRCULATIONAHA.112.000768
Sah R, Mesirca P, Van den Boogert M et al (2013b) Ion channel-kinase TRPM7 is required for maintaining cardiac automaticity. Proc Natl Acad Sci USA 110:E3037–E3046. doi:10.1073/pnas.1311865110
Serafini N, Dahdah A, Barbet G et al (2012) The TRPM4 channel controls monocyte and macrophage, but not neutrophil, function for survival in sepsis. J Immunol 189:3689–3699. doi:10.4049/jimmunol.1102969
Shimizu I, Minamino T (2016) Physiological and pathological cardiac hypertrophy. J Mol Cell Cardiol 97:245–262. doi:10.1016/j.yjmcc.2016.06.001
Simard C, Sallé L, Rouet R, Guinamard R (2012) Transient receptor potential melastatin 4 inhibitor 9-phenanthrol abolishes arrhythmias induced by hypoxia and re-oxygenation in mouse ventricle. Br J Pharmacol 165:2354–2364. doi:10.1111/j.1476-5381.2011.01715.x
Simard C, Hof T, Keddache Z et al (2013) The TRPM4 non-selective cation channel contributes to the mammalian atrial action potential. J Mol Cell Cardiol 59:11–19. doi:10.1016/j.yjmcc.2013.01.019
Tao L, Bei Y, Zhang H et al (2015) Exercise for the heart: signaling pathways. Oncotarget 6:20773–20784. doi:10.18632/oncotarget.4770
Ullrich ND, Voets T, Prenen J et al (2005) Comparison of functional properties of the Ca2+-activated cation channels TRPM4 and TRPM5 from mice. Cell Calcium 37:267–278. doi:10.1016/j.ceca.2004.11.001
Umar S, Nadadur R, Iorga A et al (2012) Cardiac structural and hemodynamic changes associated with physiological heart hypertrophy of pregnancy are reversed postpartum. J Appl Physiol 113:1253–1259. doi:10.1152/japplphysiol.00549.2012
Vennekens R, Olausson J, Meissner M et al (2007) Increased IgE-dependent mast cell activation and anaphylactic responses in mice lacking the calcium-activated nonselective cation channel TRPM4. Nat Immunol 8:312–320. doi:10.1038/ni1441
Wilkins BJ, Molkentin JD (2004) Calcium-calcineurin signaling in the regulation of cardiac hypertrophy. Biochem Biophys Res Commun 322:1178–1191. doi:10.1016/j.bbrc.2004.07.121
Wilkins BJ, Dai Y-S, Bueno OF et al (2004) Calcineurin/NFAT coupling participates in pathological, but not physiological, cardiac hypertrophy. Circ Res 94:110–118. doi:10.1161/01.RES.0000109415.17511.18
Xie J, Cha S-K, An S-W et al (2012) Cardioprotection by Klotho through downregulation of TRPC6 channels in the mouse heart. Nat Commun 3:1238. doi:10.1038/ncomms2240
Zhang Y-J, Ma N, Su F et al (2015) Increased TRPM6 expression in atrial fibrillation patients contribute to atrial fibrosis. Exp Mol Pathol 98:486–490. doi:10.1016/j.yexmp.2015.03.025
Acknowledgements
This work was supported by École Doctorale Sciences du Mouvement Humain (SMH) ED463 and the “Agence Nationale pour Recherche” -Target channel (RPV09046F5A).
Author information
Authors and Affiliations
Corresponding author
Additional information
Mélanie Gueffier and Justin Zintz have contributed equally to this work.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Gueffier, M., Zintz, J., Lambert, K. et al. The TRPM4 channel is functionally important for the beneficial cardiac remodeling induced by endurance training. J Muscle Res Cell Motil 38, 3–16 (2017). https://doi.org/10.1007/s10974-017-9466-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10974-017-9466-8