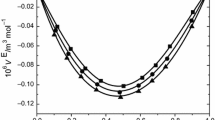
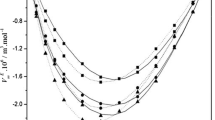
Abstract
Experimental values of the density (ρ), speed of sound (u), and viscosity (η) have been measured for binary mixtures of 2-chloroaniline with toluene and xylenes (o-xylene, m-xylene, and p-xylene) at temperatures of 303.15, 308.15, 313.15, and 318.15 K over the entire mole fraction range. By using this data, the excess molar volumes (V E), deviation in isentropic compressibility (Δκ s), and viscosity deviation (Δη) for the binary systems at the above mentioned temperatures were calculated and fitted to Redlich–Kister equation to determine the fitting parameters and the root-mean-square deviations. The excess molar volumes, deviation in isentropic compressibility, and viscosity deviation and excess Gibbs energy of activation of viscous flow have been analyzed in terms of n–π interactions, charge-transfer complexes, and dipole–induced dipole interaction between unlike molecules. The results obtained for viscosities of binary mixtures were used to test the semi-empirical relations of Grunberg–Nissan, Katti–Chaudhri, and Hind et al., equations.
















Similar content being viewed by others
References
Shaik J, Gowri sankar M, Ramachandran D, Rambabu C. Orientation effect on sign and magnitude of excess thermodynamic functions of non electrolyte solutions at different temperatures (303.15 K, 308.15 K, and 313.15 K) Korean. J Chem Eng 2014; 31(8):1460–9.
Gowri sankar M, Venkateswarlu P, Siva kumar K, Sivarambabu S. Ultrasonic studies on molecular interactions in binary mixtures of N- methyl aniline with methyl isobutylketone, + 3- pentanone, + cycloalkanones at 303.15 K. J Solut Chem 2013; 42(5):916–35.
Gowri sankar M, Venkateswarlu P, Siva kumar K, Sivarambabu S. Density, ultrasonic velocity, viscosity and their excess parameters of the binary mixtures of N,N-dimethylaniline with 1-Alkanols (C3–C5), +2-Alkanols (C3–C4), + 2-methyl-1-propanol, + 2-methyl-2- propanol at 303.15 K. Korean J Chem Eng 2013;30(5):1131–41.
Gowri sankar M, Sivarambabu S, Venkateswarlu P, Siva kumar K. Molecular interactions between amine and cyclic ketones at different temperatures. J. Therm Anal Calorim. 2014;115:1821–7.
Gowri sankar M, Sivarambabu S, Venkateswarlu P, Siva kumar K. Volumetric, speed of sound data and viscosity at (303.15 and 308.15) K for the binary mixtures of N,N-dimethylaniline + aliphatic ketones (C3–C5), +4-methyl-2-pentanone, +acetophenone + cyclicketones. J Ind Eng Chem. 2014;20:406–18.
Bhupesh GN, Namdeo V K, Potaji LK. Dielectric behaviour of binary mixture of 2-Chloroaniline with 2-Methoxyethanol and 2-Ethoxyethanol, Orbital Elec., J Chem. 2013;5:1–6.
Deshwal BR, Sharma A, Singh KC. Speeds of sounds and excess isentropic compressibilities of butyl acetate with aromatic hydrocarbons. Chin J Chem Eng. 2008;16:599–604.
Al-Kanadary JA, Al-Jimaz AS, Abdul-Latif MA. Excess molar volumes and refractive indices of (methoxy benzene + benzene or toluene, or o-xylene, or m-xylene, or p-xylene, or mesitylene) binary mixtures between T = (288.5–303.15) K. J. Chem. Thermodyn. 2006;38:1351–61.
Jeevanandham P, Kumar S, Periyasamy P. Densities, viscosities, refractive indices and excess properties of ortho-and meta-chloroaniline with 2-alkoxyethanols at 303.15 K. J Mol Liq. 2013;188:203–9.
Schaaffs W, In: Hellwege KH editor, Molekularakustik. Springer-verlag, Berlin, 1975.
Nain AK, Chandra P, Pandey JD, Gopal S. Densities, refractive indices, and excess properties of binary mixtures of 1, 4-dioxane with benzene, toluene, o-xylene, m-xylene, p-xylene and mesitylene at temperatures from (288.15–308.15) K. J Chem Eng Data. 2008;53:2654–65.
Yang C, Liu Z, Lai H, Ma P. Thermodynamic properties of binary mixtures of N-methyl-2-pyrrolidinone with cyclohexane, benzene, toluene at (303.15–353.15) K and atmospheric pressure, J Chem Thermodyn. 2007;39:28–38.
Sharma S, Thakkar K, Patel P, Makavana, M. Volumetric, viscometric and excess properties of binary mixtures of 1-iodobutane with benzene, toluene, o-xylene, m-xylene, p-xylene and mestitylene at temperatures from 303.15 to 313.15 K. Adv Phys Chem. 2013; ID 932103:1–12.
George J, sastry NV. Densities, excess molar volumes, viscosities, speeds of sound, excess isentropic compressibilities, and relative permittivities for CmH2m+1 (OCH2CH2)n OH (m = 1 or 2 or 4 and n = 1) +benzene, +toluene, + (o-,-m,-p) xylenes, +ethylbenzene, and + cyclohexane. J Chem Eng Data 2003;48:977–89.
Aminabhavi TM, Joshi SS. Excess molar volumes and viscosities of ten binary and four ternary liquid mixtures. Indian J Technol. 1992;30:197–208.
Brocos P, Pineiro A, Bravo R, Amigo A. Refractive indices, molar volumes and molar refractions of binary liquid mixtures, concepts and correlations. Phys Chem Chem Phys. 2003;5:550–7.
Pineiro A, Brocos P, Amigo A, Pintos M, Bravo R. Prediction of excess volumes and excess surface tension from experimental refractive indices. Phys Chem Liq. 2000;38:251–60.
Agarwal D, Singh M. Viscometric studies of molecular interactions in binary liquid mixtures of nitromethane with some polar and non polar solvents at 298.15 K. J Indian Chem Soc. 2004;8:850–9.
Redlich O, Kister AT. Thermodynamics of non electrolytic solutions. Algebraic representation of Thermodynamic properties and the classification of solutions. Ind Eng Chem 1948;40:345–8.
Grunberg L, Nissan AH. Mixture law for viscosity. Nature. 1949;164:799–800.
Katti PK, Chaudhri MH. Viscosities of binary mixtures benzyl acetate with dioxane, aniline and m-cresol. J Chem Eng Data. 1964;9:442–3.
Hind RK, McLaughlin E, Ubbelohde. A. Structure and viscosity of liquid camphor and pyrene mixtures. Trans Faraday Soc. 1960;56:328–30.
Tamura M, Kurata M. Viscosity of a binary mixture of liquids. Bull Chem Soc Japan. 1952;25:32–7.
Fort RJ, Moore WR. Viscosities of binary liquids mixtures. Trans Faraday Soc. 1966;62:1112–9.
Author information
Authors and Affiliations
Corresponding authors
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Praveen Chand, G., Gowri Sankar, M., Ramachandran, D. et al. Orientation effect on sign and magnitude of excess thermodynamic and transport properties of binary liquid mixtures at different temperatures. J Therm Anal Calorim 119, 2069–2078 (2015). https://doi.org/10.1007/s10973-014-4285-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10973-014-4285-2