Abstract
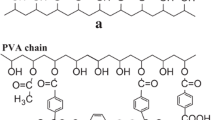
Water borne polyurethane dispersions synthesized using poly caprolactone–isophorone diisocyanate combination and caprolactone–metatetramethyl xylene diisocyanate combination. Thermal stability of the polyurethane dispersions was studied using thermogravimetric technique. The activation energy of decomposition of these polyurethane dispersions was determined by applying classical kinetic models and isoconversional method. Though the classical kinetic models assume constant activation energy for any reaction, the activation energy determined is in good agreement with that obtained via isoconversional analysis. However, the advantage of isoconversional analysis is highlighted compared to classical kinetic models based on the mechanism delineating capacity of isoconversional analysis. The present work compares the thermal stability of the aqueous polyurethane dispersion formed from poly caprolactone–isophorone diisocyanate with caprolactone–metatetramethyl xylene diisocyanate.








Similar content being viewed by others
References
Gao XY, Zhou R, Guo YP, Zhu YC, Chen X, Zheng YH, Gao W, Ma XY, Wang ZC. Synthesis and characterization of well-dispersed polyurethane/CaCO3 nanocomposites. Colloid Surf A Physicochem Eng Asp. 2010;371:1–7.
Vrsaljko D, Leskovac M, Blagojevic SI, Kovacevic V. Interphase phenomena in nanoparticulate filled polyurethane/poly(vinyl acetate) polymer systems. Polym Eng Sci. 2008;48:1931–8.
Xia H, Song M. Preparation and characterization of polyurethane–carbon nanotube composites. Soft Matter. 2005;1:386–94.
Wu Y, Natansohn A, Rochon P. Photoinduced birefringence and surface relief gratings in polyurethane elastomers with azobenzene chromophore in the hard segment. Macromolecules. 2004;37:6090–5.
Wu GM, Kong ZW, Chen CF, Chen J, Huo SP, Jiang JC. Kinetics of the cross-linking reaction of nonionic polyol dispersion from terpene-maleic ester type epoxy resin. J Therm Anal Calorim. 2013;111:735–41.
Byczynski Lukasz. Effect of different polyethers on surface and thermal properties of poly(urethane-siloxane) copolymers modified with side chain siloxane. J Therm Anal Calorim. 2013;114:397–408.
Vunain E, Mishra AK, Krause RW. Ethylene-vinyl acetate (EVA)/polycaprolactone (PCL)—Fe3O4 composites. J Therm Anal Calorim. 2013;114:791–7.
Dieterich D, Keberle W, Witt H. Polyurethane ionomers, a new class of block polymers. Angew Chem Int Eng. 1970;9:40–50.
Dieterich D. Aqueous emulsions, dispersions and solutions of polyurethanes; synthesis and properties. Prog Org Coat. 1981;9:281–340.
Frisch KC, Klempner D. Advances in urethane science and technology, vol. 10. Westport: Technomic Publishing Co.; 1987.
Ruscall R, Corner T, Stageman JF. Polymer colloids. New York: Elsevier; 1985.
Apburn CH. Polyurethane elastomers. 2nd ed. Oxford: Elsevier; 1991.
Oertel G. Polyurethane handbook. 2nd ed. Munich: Carl Hanser Verlag; 1993.
Vincent BJ, Natarajan B, available in http://dx.doi.org/10.4236/ojopm.2014.41006. Accessed 5 Feb 2014.
Brown ME, Dollimore D, Galwey AK. Comprehensive chemical kinetics, vol. 22. Amsterdam: Elsevier; 1988. p. 99.
Kissinger HE. Reaction kinetics in differential thermal analysis. Anal Chem. 1957;29:1702–6.
Augis JA, Bennett JE. Calculation of the Avrami parameters for heterogeneous solid state reactions using a modified the Kissinger method. J Therm Anal. 1978;13:283–92.
Mahadevan S, Giridhar A, Singh AK. Calorimetric measurements on As-Sb-Se glasses. J Non-Cryst Solids. 1986;88:11–34.
Marjorie E. Ducote, Huntsville, Ala. TMXDI, curing agent for hydroxy terminated propellent binders, U. S. Patent 4913753, April 3, 1990.
Abu-Sehly AA. Variation of the activation energy of crystallization in Se81.5Te16Sb2.5 chalcogenide glass: isoconversional analysis. Thermochim Acta. 2009;485:14–9.
Joraid AA. Estimating the activation energy for the non-isothermal crystallization of an amorphous Sb9.1Te20.1Se70.8 alloy. Thermochim Acta. 2007;456:1–6.
Lu MG, Lee JY, Shim MJ, Kim SW. Thermal degradation of film cast from aqueous polyurethane dispersions. J Appl Polym Sci. 2002;85:2552–8.
Mirjana Jovičić, Radmila Radičević, Jelena Pavličević, Oskar Bera. Isoconversional kinetic analysis of the alkyd/melamine resins curing. Chem Ind Chem Eng Q. 2013;19:253–62.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Vincent, B.J., Natarajan, B. Kinetics of thermal degradation of water borne polyurethane dispersion containing polycaprolactone with either isophorone diisocyanate or metatetramethyl xylene diisocyanate. J Therm Anal Calorim 119, 1373–1379 (2015). https://doi.org/10.1007/s10973-014-4264-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10973-014-4264-7