Abstract
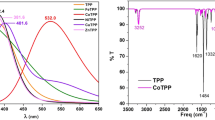
A precursor of non-aqueous porphyrins such as tetraphenyl porphyrin was synthesized and metallated with Ag, Mn and Sn to get respective metalloporphyrins. Further, they were characterized by UV–Vis spectroscopy, IR spectroscopy and proton NMR. Thermal studies of the above compounds were carried out using thermal analyser in synthetic air from room temperature to 800 °C. This analysis revealed that these compounds have thermal stabilities up to 400 °C and therefore can be successfully employed as heterogeneous catalysts as well as in semiconductor devices. In tin metalloporphyrin two Cl ligands, one above and other below the porphinato plane is comparatively stabilizing the structure to more symmetrical one which is responsible for more thermal stability. Further, residues of these compounds obtained after heating up to 800 °C, showed the presence of metal oxides and coal, respectively.








Similar content being viewed by others
References
Salker AV, Gokakakar SD. Solar assisted photo-catalytic degradation of Amido Black 10B over Cobalt, Nickel and Zinc metalloporphyrins. Int J Phys Sci. 2009;4:377–84.
Gokakakar SD, Salker AV. Solar assisted photocatalytic degradation of methyl orange over synthesized copper, silver and tin metalloporphyrins. Indian J Chem Technol. 2009;16:492–8.
Golubchikov OA, Berezin BD. Applied aspects of the chemistry of the porphyrins. Russian Chem Rev. 1986;55(8):768–85.
Meunier B. Metalloporphyrins as versatile catalysts for oxidation reactions and oxidative DNA cleavage. Chem Rev. 1992;92(6):1411–56.
Borovkov VV, Evstigneeva RP, Strekova LN, Filippovich EI. Porphyrin–quinone compounds as synthetic models of the reaction centre in photosynthesis. Russian Chem Rev. 1989;58(6):602–19.
Keller KE, Foster F. Relaxation enhancement of water by manganese(III) porphyrins. Inorg Chem. 1992;31:1353–9.
Suslick KS, Chen CT, Meredith GR, Cheng LT. Push–pull porphyrins as nonlinear optical materials. J Am Chem Soc. 1992;114:6928–30.
Marzilli LG. Medical aspects of DNA-porphyrin interactions. New J Chem. 1990;14:409–20.
Mukundan NE, Petho G, Dixon DW, Marzilli LG. Interactions of an electron-rich tetracationic tentacle porphyrin with Calf Thymus DNA. Inorg Chem. 1995;34:3677–87.
Ali H, Vanlier JE. Metal complexes as photo- and radiosensitizers. Chem Rev. 1999;99:2379–450.
Adler AD, Longo FR, Finarelli JD, Goldmacher J, Assour J, Korsakoff L. A simplified synthesis for meso-tetraphenyl porphin. J Org Chem. 1967;32:476.
Adler AD, Longo FR, Kampas F, Kim J. The preparation of metalloporphyrins. J Inorg Nucl Chem. 1970;32:2443–5.
Thomas DW, Martell AE. Metal chelates of tetraphenylporphine and of some p-substituted derivatives. J Am Chem Soc. 1959;81:5111–9.
Janson TR, Katz JJ. The Porphyrins. In: Dolphin D, editor. Vol. 4, Chapter 1, New York: Academic; 1979. p. 35.
Gokakakar SD, Salker AV. Thermal studies of some metal porphins. In: Proceedings of the 15th National Symposium on Thermal Analysis, Thermans. 2006:394–395.
Gokakakar SD, Salker AV. Thermal studies of cobalt, iron and tin metalloporphyrins. J Therm Anal Calorim. 2010;101:809–13.
Gokakakar SD, Salker AV. Synthesis, purification and thermal behavior of sulfonated metalloporphyrins. J Therm Anal Calorim. 2012;109:1487–92.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Gokakakar, S.D., Salker, A.V. Thermal studies of metalloporphyrins with metals in different oxidation states. J Therm Anal Calorim 112, 11–15 (2013). https://doi.org/10.1007/s10973-012-2860-y
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10973-012-2860-y